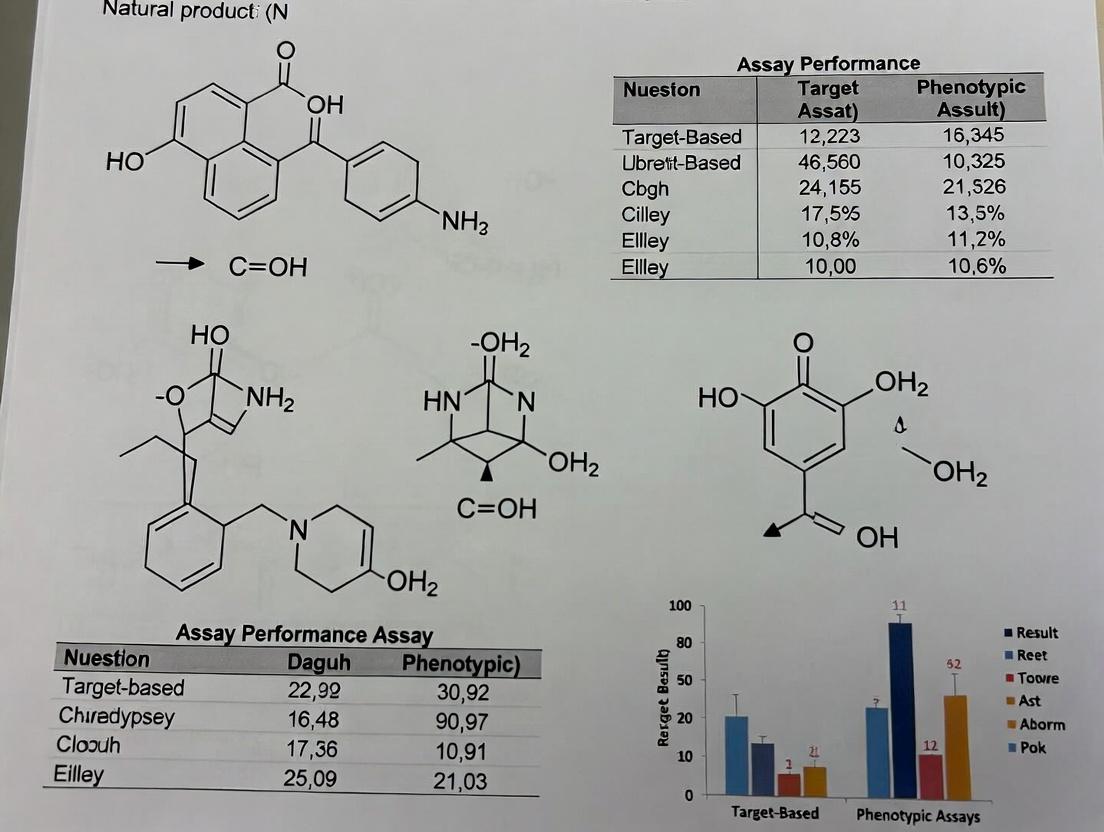
Target-Based vs. Phenotypic Assays in Natural Product Discovery: 2025 Strategic Guide
This article provides a comprehensive analysis for researchers navigating the evolving landscape of natural product-based drug discovery.
Target-Based vs. Phenotypic Assays in Natural Product Discovery: 2025 Strategic Guide
Abstract
This article provides a comprehensive analysis for researchers navigating the evolving landscape of natural product-based drug discovery. It explores the foundational principles, comparative strengths, and inherent challenges of target-based and phenotypic screening paradigms. The discussion is grounded in current methodological advances, including AI-integrated phenotypic profiling and cutting-edge target deconvolution techniques. A critical evaluation of validation strategies and performance metrics is presented, synthesizing recent evidence to offer actionable insights for optimizing assay selection and workflow design. The conclusion posits that a synergistic, integrated approach, leveraging the biological relevance of phenotypic screens and the mechanistic clarity of target-based methods, represents the most promising path forward for translating the therapeutic potential of natural products into clinical successes.
Decoding the Paradigms: Core Principles and Evolution of Assay Strategies in NP Discovery
The discovery of new therapeutics from natural products (NPs) operates at the intersection of two dominant screening philosophies: the hypothesis-driven target-based approach and the observation-driven phenotypic approach. The former begins with a known disease-associated molecular target, while the latter starts with a desired change in cellular or organismal biology, agnostic to the specific mechanism [1] [2]. For NP research, characterized by structurally complex compounds with potentially polypharmacological effects, this philosophical divide has profound implications. Phenotypic screening offers an unbiased path to discover novel biology from NPs, but leaves the challenging task of target deconvolution [3] [4]. Target-based screening accelerates optimization but may overlook the multifaceted, systems-level activities that make many NPs therapeutically valuable [5] [6]. This guide objectively compares the performance, experimental frameworks, and technological integrations of both strategies within contemporary NP-based drug discovery.
Foundational Philosophies and Strategic Objectives
The core distinction between the two paradigms lies in their starting point and primary objective.
Target-Based Screening is a deductive, hypothesis-driven process. It commences with the selection and validation of a specific protein, nucleic acid, or pathway believed to be critically involved in a disease pathology. The primary objective is to identify molecules that potently and selectively modulate the activity of this predefined target. This approach is built on a deep understanding of disease biology and allows for rational drug design. Its success is exemplified by drugs like imatinib (targeting BCR-Abl kinase) and HIV integrase inhibitors [2].
Phenotypic Screening is an inductive, observation-driven process. It begins by defining a clinically relevant phenotypic endpoint—such as inhibition of pathogen growth, reduction of a toxic protein aggregate, or restoration of normal cell morphology—in a biologically complex system (cell, organoid, or whole organism). The primary objective is to discover compounds that elicit this beneficial phenotype without any prior assumption about the molecular mechanism involved. This approach is particularly powerful for diseases with complex or poorly understood etiologies and has been instrumental in discovering first-in-class medicines, including the antimalarial artemisinin [1] [2].
Performance and Output Comparison
Historical and contemporary data reveal distinct success patterns for each strategy, particularly in the context of first-in-class drug discovery. A seminal 2013 analysis of new molecular entities provides a clear quantitative comparison [1].
Table 1: Comparative Analysis of Screening Strategies for First-in-Class Medicines (1999-2008)
| Metric | Phenotypic Screening | Target-Based Screening | Implications for NP Research |
|---|---|---|---|
| Number of First-in-Class Drugs Discovered | 28 | 17 | Phenotypic approaches have been more successful at discovering novel therapeutic mechanisms [1]. |
| Percentage of Total First-in-Class Drugs | 62.2% | 37.8% | Highlights the value of unbiased discovery for novel disease biology [1]. |
| Typical Molecular Mechanism | Often novel, previously unknown | Known, hypothesis-derived | Phenotypic screening of NPs is a key source of novel target discovery [1] [4]. |
| Key Challenge | Target identification/deconvolution | Target validation & relevance | For NPs, the "target ID" challenge is significant but is being addressed by new technologies [3] [5]. |
The resurgence of phenotypic screening is supported by technological advances. High-resolution phenotypic profiling, which uses multiplexed imaging to generate cytological "fingerprints" of compound effects, can both identify bioactive NPs and predict their mechanism of action by comparing their profiles to those of compounds with known targets [4].
Experimental Protocols and Methodologies
Target-Based Screening Protocol
A standard biochemical target-based screen for an enzyme inhibitor involves:
- Target & Assay Development: A purified, recombinant target protein (e.g., a kinase) is prepared. A biochemical assay is configured to measure its activity, often using a change in fluorescence, luminescence, or absorbance. A key metric like Z'-factor (>0.5) is validated to ensure assay robustness for high-throughput screening (HTS) [7].
- Library Screening: A diverse compound library, which can include purified natural products or NP-inspired synthetic derivatives, is dispensed into microplates (384- or 1536-well format). The target and substrates are added robotically, and the reaction is allowed to proceed [7].
- Primary Hit Detection: Plate readers measure the assay signal. Compounds causing a significant deviation from control activity (e.g., >50% inhibition) are flagged as "hits."
- Counter-Screening & Selectivity: Hits are tested against unrelated enzymes to rule out non-specific interference or aggregation-based artifacts (PAINS). They may also be screened against related protein family members to assess initial selectivity [7].
- Secondary Validation: Confirmed hits are titrated to generate dose-response curves (IC50 values). Their binding and mechanism are further validated using orthogonal techniques like surface plasmon resonance (SPR) or isothermal titration calorimetry (ITC) [7].
Phenotypic Screening Protocol (Imaging-Based High-Content Screening)
A phenotypic screen for compounds that alter specific cellular structures or pathways involves:
- Model System & Assay Development: A disease-relevant cell line is selected or engineered. A panel of fluorescent dyes or antibodies is chosen to stain key cellular components (e.g., nuclei, cytoskeleton, lysosomes, specific phosphorylated proteins) [4].
- Cell Seeding & Compound Treatment: Cells are seeded into multi-well imaging plates. After adherence, they are treated with the NP library for a defined period.
- Fixation, Staining, and Imaging: Cells are fixed, permeabilized, and stained with the fluorescent panel. Automated high-content microscopes capture high-resolution images from multiple channels in each well [4].
- Image & Data Analysis: Software algorithms segment individual cells and quantify hundreds of morphological and intensity features (e.g., nuclear size, lysosomal count, filament density). These features are normalized to control treatments to generate a cytological profile for each NP [4].
- Hit Identification & MoA Prediction: Compounds inducing the desired phenotype (e.g., reduced pathogenic protein aggregation) are identified. Their multidimensional cytological profiles can be computationally compared to a reference database of profiles for compounds with known mechanisms to generate testable hypotheses about the NP's molecular target(s) [4].
Diagram 1: Foundational Workflow of Target-Based vs. Phenotypic Screening
The Critical Challenge: Target Identification for Phenotypic Hits
The major bottleneck in phenotypic NP discovery is target deconvolution—identifying the specific biomolecule(s) through which a hit compound exerts its effect. Modern "target fishing" strategies are mitigating this challenge [3] [5].
1. Affinity-Based Proteomics: A bioactive NP is chemically modified with a linker to create a "pull-down" probe without destroying its activity. This probe is incubated with a cell lysate or live cells, allowing it to bind its protein targets. The probe-protein complexes are then immobilized on beads, purified, and the bound proteins are identified using mass spectrometry [3] [5].
2. Label-Free Techniques: * Drug Affinity Responsive Target Stability (DARTS): Exploits the principle that a protein's susceptibility to proteolysis often decreases when bound to a ligand. Treated and untreated lysates are digested with a protease; proteins protected in the treated sample are identified by proteomics [3]. * Cellular Thermal Shift Assay (CETSA): Measures the thermal stabilization of a target protein upon ligand binding in a cellular context. Protein melting curves are generated with and without compound, and stabilized proteins are identified [3].
3. Computational & Integrative Approaches: Emerging strategies like the NP-VIP (Virtual-Interact-Phenotypic) framework combine virtual screening, chemical proteomics, and phenotypic metabolomics to triangulate high-confidence targets. For example, this approach identified PARP1 and STAT3 as key targets for Salvia miltiorrhiza in treating ischemic stroke [8].
Diagram 2: Target Deconvolution Pathways for Phenotypic Natural Product Hits
The Scientist's Toolkit: Essential Research Reagents and Platforms
The execution of both screening paradigms relies on specialized tools and reagents.
Table 2: Key Research Reagent Solutions for Screening and Target ID
| Tool/Reagent | Primary Function | Typical Application | Considerations for NP Research |
|---|---|---|---|
| HTS Biochemical Assay Kits (e.g., Transcreener) | Universal, homogenous assays to measure enzyme activity (kinase, ATPase, etc.) via fluorescence polarization (FP) or TR-FRET [7]. | Target-based primary screening and hit validation. | Must ensure NP auto-fluorescence or interference does not create false signals. |
| Fluorescent Cell Staining Dyes & Antibodies | Label specific cellular compartments (nuclei, lysosomes) or post-translational modifications (phospho-proteins) [4]. | Generating multiparametric cytological profiles in phenotypic HCS. | NP-induced autofluorescence must be controlled for using appropriate filter sets. |
| Activity-Based NP Probes | Chemically modified NPs with linkers (biotin, alkyne) for immobilization or click chemistry [3] [5]. | Affinity purification pull-down experiments for target fishing. | Synthetic modification must not abolish the NP's biological activity. |
| CRISPR-Cas9 Libraries | Enable genome-wide knockout or activation screens to identify genes essential for a phenotype or compound sensitivity [9]. | Functional validation of putative targets from deconvolution. | Can confirm if a hypothesized target is genetically required for the NP's effect. |
| AI-Powered Target Prediction Servers | Use QSAR, pharmacophore modeling, and deep learning to predict protein targets based on compound structure [5] [8]. | Generating initial target hypotheses for computational triage. | Accuracy is highly dependent on training data; novel NP scaffolds may be challenging. |
Synthesis and Strategic Integration
The dichotomy between target-based and phenotypic screening is not absolute, and the most effective modern NP research integrates both. A combined targeted-phenotypic approach is increasingly common, where a cellular assay is designed to report on a specific pathway or target activity within its native physiological context [9]. Furthermore, phenotypic hits can be reverse-engineered via target deconvolution to fuel new target-based discovery campaigns. Conversely, NP structures identified in target-based screens can be subjected to broad phenotypic profiling to uncover additional, potentially therapeutic off-target activities or to predict toxicity [4] [6].
The choice of strategy depends on the research goal: phenotypic screening for novel biology and first-in-class mechanisms, and target-based screening for optimizing selectivity and developing best-in-class drugs against validated targets [1] [9]. For the unique challenges and opportunities presented by natural products, leveraging both philosophies in a complementary cycle represents the most robust path from complex mixtures to novel therapeutics.
The dominant paradigm in drug discovery has cycled between phenotypic and target-based approaches. Historically, most medicines, including natural products (NPs), were discovered by observing their effects on whole organisms or tissues—a phenotypic approach [10] [11]. The late 20th century saw a decisive shift toward target-based drug discovery (TDD), driven by advances in genomics and molecular biology that promised rational design and high-throughput efficiency [11] [9]. However, analyses revealing that a majority of first-in-class drugs (1999-2008) originated from phenotypic drug discovery (PDD) have fueled a significant resurgence of this approach over the past decade [10] [11]. This resurgence is particularly pronounced in natural products research, where the complex chemistry and polypharmacology of NPs often defy reductionist target-based screening. Modern PDD is now characterized by high-resolution profiling technologies, advanced disease models, and sophisticated target deconvolution methods, creating a synergistic interplay with target-based strategies [4] [12] [6]. This guide compares the performance of contemporary phenotypic and target-based assay paradigms within NP research, supported by experimental data and protocols.
Historical Context and Performance Comparison
The shift between paradigms is rooted in their fundamental strategies. PDD identifies compounds based on their modulation of a disease-relevant phenotype in a cellular or organismal system, without preconceived notions of the molecular target [10]. TDD, in contrast, begins with a hypothesized protein target implicated in a disease and screens for compounds that modulate its activity in a purified or engineered system [11] [9].
A landmark analysis by Swinney and Anthony (2011) demonstrated that between 1999 and 2008, 28 of 50 (56%) first-in-class small-molecule drugs were discovered through phenotypic screening, compared to 17 (34%) through target-based approaches [10] [11]. This disproportionate contribution of PDD to innovative therapeutics is attributed to its target-agnostic nature, which can reveal novel biology and unexpected mechanisms of action (MOA), such as modulators of protein folding, splicing, or multi-protein complexes [10]. Notable NP-derived examples include the immunosuppressant rapamycin (sirolimus), whose target (mTOR) was identified years after its phenotypic discovery, and the anti-malarial artemisinin [11] [6].
The following table compares the core characteristics and outputs of the two paradigms, particularly in the context of NP research:
Table: Comparative Analysis of Phenotypic vs. Target-Based Drug Discovery for Natural Products
| Aspect | Phenotypic Drug Discovery (PDD) | Target-Based Drug Discovery (TDD) |
|---|---|---|
| Starting Point | Disease phenotype in a biologically complex system (cell, tissue, organism) [10]. | Hypothesis about a specific protein target's role in disease [11] [9]. |
| Primary Screening Readout | Holistic measurement of phenotype reversal (e.g., cell viability, morphology, functional recovery) [4] [10]. | Biochemical activity on an isolated target (e.g., enzyme inhibition, receptor binding) [9]. |
| Advantages in NP Research | Unbiased discovery of novel targets/MOAs; captures polypharmacology and systems-level effects; suitable for NPs with unknown targets [10] [6]. | Straightforward structure-activity relationship (SAR) and hit optimization; high throughput; clear mechanistic hypothesis [11] [9]. |
| Key Challenges | Target deconvolution can be difficult; assays may be lower throughput and more complex; hit chemistry may be challenging [10] [13]. | May fail due to poor target validation or lack of cellular activity; misses complex, multi-target mechanisms common to NPs [10] [11]. |
| Contribution to First-in-Class Drugs (1999-2008) | 56% (28 of 50 drugs) [10] [11]. | 34% (17 of 50 drugs) [10] [11]. |
| Target Identification Necessity | Required after hit discovery (downstream) [13] [14]. | Defined before screening (upstream) [9]. |
The Modern Phenotypic Assay Toolkit
The resurgence of PDD is powered by technological advances that address its historical limitations. Modern phenotypic screening employs high-resolution, multi-parameter profiling to generate rich data far beyond simple viability readouts.
High-Content Imaging and Cytological Profiling: As demonstrated by a 2017 study, high-content screening (HCS) can profile NP-induced effects using a panel of 14 fluorescent markers targeting major organelles and pathways [4]. This generates cytological profiles (CPs)—unique phenotypic fingerprints—for each compound. Testing 124 NPs revealed that small structural changes could cause profound phenotypic shifts, enabling cell-based structure-activity relationship studies and prediction of MOA by comparing NP profiles to a library of reference compounds with known targets [4].
Cell Painting and Predictive Profiling: The Cell Painting assay, a standardized morphological profiling technique, stains eight cellular components to create a high-dimensional phenotypic profile [15]. A 2023 large-scale study evaluated the power of chemical structure (CS), gene expression (GE from L1000), and morphological profiles (MO from Cell Painting) to predict bioactivity in 270 unrelated assays. Morphological profiling alone predicted the highest number of assays (28) with high accuracy (AUROC > 0.9). Critically, combining morphological profiles with chemical structure data nearly doubled the number of predictable assays compared to chemical structure alone (31 vs. 16), demonstrating powerful complementarity [15].
Table: Key Phenotypic Profiling Technologies and Performance Data
| Technology | Key Metrics/Output | Experimental Findings in NP/Compound Screening | Reference |
|---|---|---|---|
| High-Content Cytological Profiling | 134 cellular features distilled to 20 core features; profiles 14 cellular markers [4]. | Screened 124 NPs; identified sub-classes (e.g., topoisomerase inhibitors) via profile matching; enabled SAR for 17 podophyllotoxin derivatives [4]. | [4] |
| Cell Painting (Morphological Profiling) | 5-channel fluorescence imaging capturing ~1,500 morphological features [15]. | MO profiles predicted 28/270 assays (AUROC>0.9); combined with chemical structure (CS+MO), predicted 31 assays, showing strong synergy [15]. | [15] |
| Gene Expression Profiling (L1000) | Measures expression of 978 landmark genes; infers whole transcriptome [15]. | GE profiles predicted 19/270 assays (AUROC>0.9); provided complementary information to MO and CS [15]. | [15] |
Target Identification and Validation for Phenotypic Hits
A major challenge in PDD is identifying the molecular target(s) underlying an observed phenotype, a process known as target deconvolution. For NPs with complex structures, traditional chemical proteomics methods requiring compound modification can be prohibitively difficult [13] [14]. This has driven the development and adoption of label-free target identification methods.
Key Label-Free Methodologies:
- Cellular Thermal Shift Assay (CETSA): This method detects target engagement by measuring ligand-induced changes in a protein's thermal stability within cells or lysates. When a drug binds, the protein typically becomes more stable and resists heat-induced unfolding and aggregation. The proportion of remaining soluble protein is quantified at different temperatures (melting curve) or at a single temperature (isothermal dose-response), often using mass spectrometry [13] [16]. CETSA confirms binding in a physiologically relevant cellular context.
- Drug Affinity Responsive Target Stability (DARTS): DARTS exploits the principle that a protein is less susceptible to proteolysis when bound to a ligand. Cell lysates treated with or without the compound are subjected to limited proteolysis, and the resulting protein fragments are compared. Proteins protected from degradation in the compound-treated sample are potential targets [13] [14].
- Stability of Proteins from Rates of Oxidation (SPROX): SPROX measures the rate of methionine oxidation in proteins as a function of chemical denaturant concentration. Ligand binding can alter a protein's thermodynamic stability, shifting its denaturation curve, which is detected via mass spectrometry [13].
Integrated Multi-Omics Strategy (NP-VIP): A 2024 study on Salvia miltiorrhiza introduced a Natural Product Virtual screening-Interaction-Phenotype (NP-VIP) strategy that synergistically combines target-based and phenotypic concepts [12]. The workflow involves: 1) Virtual Screening (VS) of NP constituents against protein databases to predict potential targets; 2) CETSA to experimentally validate direct protein binding in cells; and 3) Metabolomics to observe phenotypic changes in cellular metabolism and identify functionally relevant pathways [12]. Applying this to Salvia miltiorrhiza extract identified 29, 100, and 78 potential targets from VS, CETSA, and metabolomics, respectively. Integration pinpointed five high-confidence targets (e.g., PARP1, STAT3), demonstrating how multi-modal integration overcomes the limitations of any single approach [12].
Experimental Protocols for Key Assays
Objective: To generate high-resolution cytological profiles (CPs) of NP-induced effects for MOA prediction and SAR analysis. Workflow:
- Cell Culture and Treatment: Seed U2OS cells in 384-well plates. Treat with a library of NPs (e.g., 124 compounds) and reference pharmacologically active compounds across a range of concentrations (e.g., 0.1-30 µM) for 24 hours. Include DMSO vehicle controls.
- Multiplexed Staining: Fix cells and stain with a panel of fluorescent dyes targeting:
- Nucleus: DNA (Hoechst).
- Nucleolus: RNA (SYTO RNASelect).
- Endoplasmic Reticulum: Concanavalin A.
- Golgi Apparatus: BODIPY TR ceramide.
- Lysosomes: LysoTracker Deep Red.
- Actin & Microtubules: Phalloidin and anti-α-tubulin antibody.
- Additional Markers: For cell health, plasma membrane, NF-κB translocation, etc.
- Image Acquisition: Acquire images using a high-content imaging system (e.g., ImageXpress Micro) with a 20x objective. Capture 4-9 fields per well to analyze 500-1000 cells per condition.
- Image and Data Analysis: Use image analysis software (e.g., MetaXpress) to segment cells and extract ~134 morphological and intensity-based features (e.g., organelle count, size, intensity, texture). Normalize data to vehicle control.
- Profile Generation and Analysis: Reduce features to a core set of 20 descriptors. Generate CPs as heatmaps. Use hierarchical clustering to compare NP profiles to reference compound libraries for MOA prediction. Perform principal component analysis to visualize compound clustering and identify SAR trends within chemical series.
Objective: To detect direct binding of an NP to its cellular protein targets by measuring thermal stabilization. Workflow (MS-based CETSA):
- Compound Treatment: Treat intact cells (e.g., HeLa, 1-2 million per sample) with the NP of interest or vehicle control for a predetermined time (e.g., 1 hour).
- Heat Challenge: Aliquot cell suspensions into PCR tubes. Heat each aliquot at a distinct temperature across a gradient (e.g., 37°C to 67°C) for 3 minutes using a thermal cycler.
- Cell Lysis and Clarification: Lyse heated cells, freeze-thaw, and centrifuge at high speed (e.g., 20,000 x g) to separate soluble protein from aggregated precipitates.
- Protein Digestion and TMT Labeling: Quantify protein in supernatants. Digest proteins with trypsin. Label peptides from different temperature points with tandem mass tag (TMT) reagents.
- LC-MS/MS Analysis and Data Processing: Pool labeled samples and analyze by liquid chromatography-tandem mass spectrometry (LC-MS/MS). Identify and quantify proteins.
- Melting Curve Analysis: For each protein, plot the relative soluble amount (log2(compound/control)) against temperature. A rightward shift in the melting curve (increased Tm) in the compound-treated sample indicates thermal stabilization and direct target engagement.
Objective: To identify high-confidence target ensembles for complex natural product extracts. Workflow:
- Virtual Screening (Interaction Prediction):
- Establish the chemical profile of the NP extract using UPLC-MS/MS.
- Dock identified constituent molecules against human protein targets using software like LeDock.
- Prioritize predicted targets based on docking scores and literature association with the disease of interest.
- CETSA (Interaction Validation):
- Perform MS-based CETSA (as in Protocol 4.2) on cells treated with the whole NP extract.
- Identify proteins with significantly altered thermal stability (Tm shifts) upon extract treatment.
- Metabolomics (Phenotype Analysis):
- Treat cells with the NP extract and perform untargeted metabolomics via LC-MS.
- Identify significantly altered metabolites and map them to affected biochemical pathways using KEGG or MetaboAnalyst.
- Infer the proteins/enzymes regulating those pathways as phenotypically relevant targets.
- Data Integration:
- Intersect the target lists from VS, CETSA, and metabolomics analyses.
- Proteins appearing in multiple datasets constitute a high-confidence target ensemble for the NP extract.
- Validate key targets through orthogonal methods like western blot, siRNA knockdown, or functional assays.
The Scientist's Toolkit: Key Research Reagent Solutions
Table: Essential Reagents and Materials for Featured Assays
| Item | Function/Description | Typical Application |
|---|---|---|
| Fluorescent Dye Panel (Hoechst, LysoTracker, ConA, etc.) | A multiplexed set of dyes for staining specific organelles and cellular structures to create cytological profiles [4]. | High-content phenotypic screening (HCS). |
| LOPAC1280 or Similar Library | A library of pharmacologically active compounds with known mechanisms of action, used as a reference for phenotypic profile matching [4]. | MOA prediction and annotation in phenotypic screens. |
| Tandem Mass Tag (TMT) Reagents | Isobaric chemical labels for multiplexed quantitative proteomics. Allows simultaneous quantification of proteins from multiple samples (e.g., different temperatures in CETSA) [12]. | MS-based CETSA and other quantitative proteomics workflows. |
| Chemical Denaturants (Urea, GdmCl) | Chaotropic agents that disrupt protein non-covalent structure, used to measure protein folding stability [13]. | SPROX, Pulse Proteolysis, CPP experiments. |
| Non-ionic Detergent (e.g., NP-40) | Used in cell lysis buffers to solubilize membranes while maintaining protein-protein interactions and complex integrity [13]. | Preparation of cell lysates for DARTS, CETSA (lysate mode). |
| Thermostable Protease (e.g., Pronase) | A broad-spectrum protease used for limited proteolysis in DARTS experiments [13]. | DARTS target identification. |
| Silica Gel for Column Chromatography | Stationary phase for fractionating complex natural product extracts based on polarity [12]. | Pre-fractionation of NP extracts prior to screening or analysis. |
| CETSA-Compatible Cell Line | A robust, adherent cell line (e.g., HeLa, U2OS) suitable for the heating and processing steps of CETSA [16] [12]. | CETSA target engagement studies. |
The discovery and development of therapeutics from natural products present a unique paradox. These compounds, derived from plants, microbes, and marine organisms, have been the source of numerous first-in-class medicines and possess inherent bioactivity and structural complexity often unmatched by synthetic libraries [3]. However, their very advantages constitute the core challenges for modern, mechanism-driven drug development. This article frames these challenges within the long-standing strategic dichotomy in pharmaceutical research: target-based versus phenotypic screening approaches [17].
Target-based discovery begins with a well-characterized molecular target, leveraging structural biology and rational design to develop highly specific inhibitors or modulators [17]. In contrast, phenotypic discovery identifies compounds based on a measurable biological response in cells or whole organisms, often without prior knowledge of the specific molecular target [17]. For natural products, this dichotomy is critical. Their frequent polypharmacology (action on multiple targets) and unknown mechanisms of action (MoAs) align more naturally with the holistic view of phenotypic screening. Yet, the demand for mechanistic understanding and safety validation pushes research toward target deconvolution, a process that remains notoriously difficult [18]. This guide objectively compares the performance of research strategies for natural products, focusing on their ability to navigate complexity, elucidate polypharmacology, and reveal unknown MoAs, supported by experimental data and protocols.
Comparative Analysis: Phenotypic vs. Target-Based Screening for Natural Products
The choice between phenotypic and target-based screening paradigms significantly impacts the trajectory of natural product research. The table below summarizes the performance of each approach against key criteria relevant to natural products' unique challenges.
Table 1: Performance Comparison of Phenotypic vs. Target-Based Screening for Natural Product Research
| Evaluation Criterion | Phenotypic Screening Approach | Target-Based Screening Approach | Supporting Data & Evidence |
|---|---|---|---|
| Ability to Discover Novel Mechanisms | High. Unbiased by prior target hypotheses; historically responsible for most first-in-class drugs [17]. | Low. Constrained by pre-selected, known targets; cannot identify novel biology outside the target hypothesis. | Discovery of immunomodulatory drugs like thalidomide and its analogs (lenalidomide) via phenotypic effects on TNF-α inhibition, with target (cereblon) identified years later [17]. |
| Handling of Polypharmacology | High. Captures net functional outcome of multi-target interactions; polypharmacology is an inherent advantage. | Low. Designed for single-target specificity; polypharmacology is typically seen as an off-target liability to be eliminated. | Artemisinin's antimalarial action may involve multiple mechanisms (alkylation, oxidative stress); a phenotypic screen identified its activity while target identification remains complex [18]. |
| Target Identification / MoA Deconvolution | Major Challenge. Requires extensive, often difficult follow-up work (affinity purification, chemoproteomics). | Not Applicable. Target is known from the outset, though full MoA may still require elaboration. | A 2025 review notes that target ID for natural products is a "significant challenge," driving innovation in chemical proteomics methods [3]. |
| Hit Rate for Bioactive Natural Products | Moderate to High. Filters for compounds that can penetrate cells and induce a relevant biological effect. | Very Low. Requires the natural product to be a potent, specific ligand for a single, pre-chosen protein target. | Phenotypic screens of natural product libraries consistently yield bioactive hits affecting complex processes like immune cell activation or cancer cell death [17]. |
| Optimization & Medicinal Chemistry | Complex. Requires iterative cycling between phenotypic optimization and target identification. Can be guided by structure-activity relationships (SAR). | Straightforward. SAR is directly informed by the structure of the target binding site, enabling rational design. | Optimization of thalidomide to lenalidomide was guided by phenotypic SAR (increased potency for TNF-α downregulation, reduced sedation) [17]. |
| Risk of Clinical Attrition | Potentially Lower. Compounds have demonstrated efficacy in a complex, disease-relevant system early on. | Potentially Higher. High target specificity may not translate to clinical efficacy if the target hypothesis is flawed or compensatory pathways exist [17]. | Analysis shows targeted approaches often fail due to lack of clinical efficacy stemming from incomplete disease biology understanding [17]. |
Experimental Protocols for Mode of Action Deconvolution
Overcoming the "unknown MoA" challenge requires a suite of sophisticated experimental techniques. Below are detailed protocols for two cornerstone methodologies.
Protocol 1: Affinity Purification (Target Fishing) with Quantitative Proteomics
This classic strategy remains a mainstay for identifying direct protein binders of a natural product [3].
1. Probe Design & Synthesis:
- Procedure: A functionalized derivative of the natural product is synthesized. This typically involves chemically adding a linker (like a polyethylene glycol chain) terminated with a handle (e.g., an alkyne for "click chemistry," or biotin for strong affinity capture). Critical Control: A structurally similar but inactive analog should be modified identically to serve as a negative control for non-specific binding.
- Validation: The modified probe must be validated in the original phenotypic assay to ensure it retains bioactivity comparable to the parent natural product.
2. Cell Lysis and Probe Incubation:
- Procedure: Prepare whole-cell lysates from relevant cell lines or primary tissues under non-denaturing conditions to preserve native protein structures. Incubate the lysates with the bioactive probe (experimental) or the inactive control probe. Competition experiments can be performed by co-incubating with an excess of unmodified natural product to identify binding that is specifically displaced.
- Materials: Lysis buffer (e.g., 50 mM Tris-HCl pH 7.5, 150 mM NaCl, 1% NP-40, protease inhibitors), rotator for gentle mixing.
3. Affinity Capture & Wash:
- Procedure: If using a biotinylated probe, incubate the lysate-probe mixture with streptavidin-conjugated beads. For "click chemistry" probes, perform a copper-catalyzed azide-alkyne cycloaddition (CuAAC) reaction to conjugate the probe-bound proteins to azide- or cyclooctyne-functionalized beads.
- Wash: Wash the beads stringently with lysis buffer followed by high-salt buffers (e.g., 1 M NaCl) and possibly mild detergent buffers to remove non-specifically adsorbed proteins.
4. Protein Elution & Identification:
- Procedure: Elute bound proteins using Laemmli buffer for western blot analysis, or by on-bead trypsin digestion for mass spectrometry (MS).
- MS Analysis: Subject digested peptides to liquid chromatography-tandem MS (LC-MS/MS). Identify and quantify proteins in the experimental sample relative to the negative control.
- Key Reagents: Streptavidin/NeutrAvidin beads, CuAAC kit reagents (if needed), sequencing-grade trypsin, LC-MS/MS system.
5. Hit Validation:
- Procedure: Candidate target proteins require orthogonal validation. Techniques include Cellular Thermal Shift Assay (CETSA) to demonstrate ligand-induced thermal stabilization of the target protein in intact cells, surface plasmon resonance (SPR) to measure binding kinetics in vitro, or gene knockdown/knockout to see if it phenocopies or rescues the natural product's effect [3] [18].
Protocol 2: Photoaffinity Labeling (PAL) for Capturing Transient Interactions
PAL is crucial for identifying low-abundance or transient protein-ligand interactions by covalently "capturing" the binding event upon UV irradiation [3].
1. Photoaffinity Probe Design:
- Procedure: Synthesize a derivative of the natural product incorporating a photoactivatable group (e.g., diazirine, benzophenone) and a reporter tag (biotin or alkyne). The photoactivatable group is inert until exposed to UV light of a specific wavelength, generating a highly reactive carbene or radical that forms a covalent bond with nearby proteins.
- Key Consideration: The linker and photoaffinity group must be positioned to minimize interference with the compound's bioactivity.
2. Live-Cell or Lysate Labeling:
- Procedure (Live-Cell): Incubate live cells with the photoaffinity probe. Wash cells to remove excess probe. Irradiate the cell culture with UV light (e.g., 365 nm for diazirines) to activate crosslinking. Harvest and lyse cells.
- Procedure (Lysate): Incubate cell lysates with the probe, then irradiate the mixture.
- Materials: UV crosslinker with appropriate wavelength control.
3. Capture and Analysis:
- Procedure: Following crosslinking, proceed with affinity capture using the reporter tag (e.g., streptavidin pull-down for biotin) as described in Protocol 1. The covalent bond formed during PAL allows for even more stringent washing conditions, reducing background.
- Analysis: Elute and identify proteins by MS. The covalent labeling also allows for mapping the binding site by digesting the captured protein and identifying the specific peptide(s) carrying the probe modification via MS.
4. Data Analysis:
- Procedure: Use bioinformatics software to compare protein abundance in probe-labeled samples vs. negative control samples (no UV, no probe, or inactive probe). Proteins significantly enriched in the experimental sample are high-confidence candidate targets [3].
Diagram 1: Target Deconvolution Workflow for Natural Products (Max. Width: 760px)
Navigating Polypharmacology: From Challenge to Therapeutic Strategy
Polypharmacology—the action of a single compound on multiple molecular targets—is not a bug but a fundamental feature of many effective natural products [19]. This multi-target action can be harnessed for superior efficacy, especially in complex diseases like cancer and autoimmune disorders, but requires precise characterization to avoid unwanted side effects.
Table 2: Documented Polypharmacology Mechanisms of Natural Products & Drugs
| Compound | Primary Known Target/Pathway | Additional Identified Targets/Effects | Therapeutic Consequence | Identification Method |
|---|---|---|---|---|
| Thalidomide / Lenalidomide | Cereblon (CRL4 E3 ubiquitin ligase substrate receptor), leading to degradation of Ikaros (IKZF1) and Aiolos (IKZF3) [17]. | Binds cereblon to induce degradation of multiple other "neosubstrates" (e.g., CK1α, SALL4). Also modulates TNF-α production and COX-2 expression [17]. | Efficacy in multiple myeloma and myelodysplastic syndromes; also causes teratogenicity (via SALL4 degradation) and other side effects. | Biochemical purification, phenotypic screening, and later structural biology [17]. |
| Artemisinin | Heme activation leading to alkylation and oxidative stress in malaria parasite [18]. | Shown to bind to multiple human proteins in a chemoproteomic screen, suggesting potential host-directed effects. Also has reported anti-cancer and anti-viral activity [18]. | Potent, rapid antimalarial action; potential for drug repurposing. | Reverse chemical proteomics, phenotypic screening in other diseases [18]. |
| Curcumin | Pleiotropic effects, but identified targets include KEAP1 (NRF2 pathway), IKK (NF-κB pathway), and various enzymes [3]. | Interacts with a wide network of signaling proteins, transcription factors, and enzymes (e.g., amyloid-β, STAT3). Poor bioavailability complicates analysis. | Broad anti-inflammatory and antioxidant effects claimed; but clinical efficacy is debated due to pharmacokinetics. | Affinity purification, chemoproteomics, computational docking [3]. |
| Kinase Inhibitors (e.g., Staurosporine - natural product origin) | Originally identified as a potent inhibitor of Protein Kinase C (PKC). | Profiling shows potent inhibition of a broad spectrum of kinases (e.g., PKA, CAMK, CK1) [19]. | Excellent research tool, but too promiscuous for clinical use as an anticancer drug. Led to development of more selective analogs. | Kinase activity profiling panels, chemoproteomics [19]. |
Diagram 2: Polypharmacology Network of a Natural Product (Max. Width: 760px)
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Natural Product MoA Studies
| Tool/Reagent | Function in Experiment | Key Consideration for Natural Products |
|---|---|---|
| Functionalized Natural Product Probes (Biotin-, Alkyne-, Photoaffinity-tagged) | Serve as molecular bait to fish out target proteins from complex biological mixtures for affinity purification or photoaffinity labeling [3]. | Synthetic derivatization must not abolish bioactivity. An inactive analog probe is a critical negative control. |
| Streptavidin/Avidin Beads | High-affinity capture matrix for biotinylated probes and their bound protein complexes. | High binding capacity is needed for low-abundance targets. Non-specific binding can be high; requires stringent wash optimization. |
| "Click Chemistry" Kits (CuAAC or Copper-free) | Enable bioorthogonal conjugation of an alkyne-bearing probe to an azide-bead (or vice versa) after the binding event occurs in cells [3]. | Useful for probes where direct biotin conjugation harms activity. Copper-catalyzed (CuAAC) reactions can be toxic to some proteins. |
| Photoaffinity Groups (Diazirine, Benzophenone) | Upon UV irradiation, form highly reactive intermediates that covalently crosslink the probe to its binding site on the target protein [3]. | Allows capture of transient or low-affinity interactions. Placement on the natural product scaffold is critical for successful labeling. |
| Cell Lysate/Membrane Protein Prep Kits | Generate functional protein extracts from cells or tissues under native conditions for in vitro binding assays. | Natural products often target membrane proteins or multiprotein complexes; lysis conditions must preserve these structures. |
| Thermal Shift Assay Dyes (e.g., SYPRO Orange) | Detect ligand-induced stabilization of a target protein during a Cellular Thermal Shift Assay (CETSA), indicating direct binding. | An orthogonal, cell-based validation method. Works best with purified recombinant protein or simple lysates for target validation. |
| CRISPR/Cas9 Knockout Libraries or siRNA Pools | Enable genome-wide or targeted gene knockdown to identify genes essential for the natural product's phenotypic effect (genetic MoA studies). | Complementary to biochemical methods. Can reveal synthetic lethal interactions or pathway dependencies, even if not a direct target. |
The unique challenges posed by natural products—structural complexity, polypharmacology, and unknown MoAs—necessitate a move beyond the rigid dichotomy of phenotypic versus target-based screening. The future lies in integrated hybrid approaches [17]. This involves initiating discovery with complex phenotypic screens to leverage the holistic bioactivity of natural products, followed by rapid target deconvolution using the advanced chemical proteomics and computational methods outlined here. The resulting multi-target profiles must then be understood not as a list of off-target effects, but as polypharmacology networks that can be mapped and optimized. By systematically applying this comparative framework, researchers can transform the inherent challenges of natural products into a structured strategy for discovering novel, effective, and mechanistically rich therapeutics.
Natural products (NPs) have been a cornerstone of drug discovery, historically providing a significant percentage of new therapeutics. Their structural complexity and evolutionary optimization allow them to engage with challenging biological targets—such as protein-protein interactions, nucleic acid complexes, and spliceosomes—that often remain intractable to conventional synthetic, "drug-like" libraries [20]. However, the path from a bioactive natural extract to a characterized therapeutic candidate is fraught with complexity. This journey is navigated using two primary, and often philosophically opposed, methodological roadmaps: target-based assays and phenotypic assays.
Target-based screening operates on a reductionist principle. It involves testing compounds against a purified protein or a well-defined molecular target in a controlled biochemical environment. Success is measured by the compound's ability to modulate the specific target's activity [21]. In contrast, phenotypic screening adopts a holistic approach. It assesses the effects of compounds on whole cells or organisms, measuring complex, multifaceted outputs like cell morphology, viability, or reporter gene expression without pre-supposing the mechanism of action (MOA) [22] [4]. The resulting "phenotypic fingerprint" can reveal bioactivity against any biomolecular component within the living system.
The central thesis of modern NP research is that an exclusive commitment to either paradigm is suboptimal. The "imperative for integration" arises from the need to leverage the precision of target-based methods with the biological relevance and de novo discovery power of phenotypic approaches. This guide provides a comparative analysis of these strategies, supported by experimental data and protocols, to inform researchers and drug development professionals on building a synergistic discovery pipeline.
Methodological Comparison: Core Principles, Applications, and Data
The following tables provide a structured comparison of the two assay paradigms, summarizing their defining characteristics, strengths, limitations, and representative experimental outcomes.
Table 1: Foundational Comparison of Target-Based vs. Phenotypic Assay Paradigms
| Aspect | Target-Based Assays | Phenotypic Assays |
|---|---|---|
| Core Principle | Tests interaction with or modulation of a predefined, isolated molecular target (e.g., enzyme, receptor) [21]. | Measures observable change in cell or organism phenotype without assumption of a specific target [22] [4]. |
| Typical Readout | Biochemical signal (e.g., fluorescence, luminescence, binding affinity). | Multidimensional imaging features, cell viability, morphological changes, gene expression profiles [22] [4]. |
| Primary Strength | High precision, mechanistic clarity, amenable to high-throughput screening (HTS), direct structure-activity relationship (SAR) studies. | Biologically relevant context, discovers novel targets/pathways, identifies polypharmacology, captures complex phenotypes like mitotic arrest [22]. |
| Key Limitation | May not translate to cellular activity; misses off-target effects; requires a prior, validated target hypothesis. | Mechanism of action (MOA) is initially unknown; can be lower throughput; data analysis is complex; may identify cytotoxic compounds nonspecifically [22]. |
| Ideal Application | Optimizing leads for a known target, fragment-based screening, selectivity profiling. | De novo drug discovery, investigating complex diseases, natural product MOA exploration, toxicology assessment [4]. |
| Target Identification | Built into the assay design (the target is known). | Requires follow-up techniques (e.g., chemical proteomics, genetic screens) for deconvolution [8] [3]. |
Table 2: Comparative Performance Data from Representative Studies
| Study Focus | Target-Based Approach (Data) | Phenotypic Approach (Data) | Comparative Insight |
|---|---|---|---|
| Hit Discovery Rate | In screens for challenging targets (e.g., protein-protein interactions), hit rates from synthetic libraries can be very low [20]. | Screening 5,304 microbial extracts via cytological profiling identified 41 discrete bioactivity clusters, including a specific antimitotic cluster [22]. | Phenotypic screening of NP libraries can yield richer, more diverse hit clusters for complex biology, as NPs sample broader chemical space [20] [22]. |
| Mechanism Elucidation | Affinity chromatography (e.g., Cell Membrane Chromatography) directly isolates receptor ligands from complex mixtures [21]. | Cytological profiles of extracts clustered with known microtubule poisons; subsequent isolation confirmed diketopiperazine XR334 as the antimitotic agent [22]. | Target-based methods directly link compound to target. Phenotypic methods predict MOA by profile matching, requiring confirmation but enabling discovery of unexpected mechanisms. |
| Complexity & Throughput | Affinity selection mass spectrometry (ASMS) can screen complex mixtures against a single target in a semi-high-throughput manner [21]. | High-content screening (HCS) of 124 NPs generated 134-dimensional phenotypic profiles per compound, but requires sophisticated image analysis [4]. | Target-based assays are generally higher in throughput. Phenotypic assays generate vastly more information per well but at a slower rate and with greater computational demand. |
| Data Output | Quantitative binding constants (IC50, Kd), enzyme kinetic parameters. | Quantitative multiparametric "fingerprints" (e.g., nuclear size, lysosomal count, tubulin intensity) that can be clustered [4]. | Target-based data is unidimensional and direct. Phenotypic data is multidimensional and integrative, revealing system-wide effects. |
Experimental Protocols
Protocol for Phenotypic Screening: Cytological Profiling (CP)
This protocol, adapted from high-content phenotypic screening studies, is used to generate multiparametric fingerprints of natural product effects [22] [4].
- Cell Preparation & Plating: Seed adherent cells (e.g., HeLa, U2OS) into multi-well imaging plates at a density ensuring ~70% confluence at the time of assay. Allow cells to adhere overnight in standard culture conditions.
- Compound Treatment: Treat cells with natural product extracts or pure compounds at a range of concentrations. Include positive controls (e.g., paclitaxel for mitotic arrest, nocodazole for microtubule destabilization) and vehicle (DMSO) controls. Incubate for a defined period (typically 6-24 hours).
- Fixation and Staining: Fix cells with paraformaldehyde (e.g., 4% in PBS). Permeabilize with Triton X-100. Block with a suitable protein (e.g., BSA). Apply a multiplexed fluorescent stain panel to mark key cellular components. A typical panel includes:
- DAPI/Hoechst: Nuclear DNA (cell count, nuclear morphology).
- Phalloidin: Actin cytoskeleton.
- Anti-α-tubulin antibody: Microtubule network.
- LysoTracker/LAMP1 antibody: Lysosomes.
- Antibodies for markers like phosphorylated histone H3 (pHH3, mitosis), cleaved caspase-3 (apoptosis), or NF-κB localization.
- High-Content Imaging: Image each well using an automated high-content microscope with environments for each fluorescence channel. Acquire multiple fields per well to capture a statistically significant cell population (500-1000 cells).
- Image & Data Analysis:
- Use image analysis software (e.g., CellProfiler, IN Carta) to segment cells and identify subcellular compartments.
- Extract quantitative features for each cell: intensities, textures, morphological measurements (size, shape), and counts for each channel.
- Calculate population means for each feature per well, normalized to vehicle controls.
- Reduce dimensionality (e.g., to 20 core features) and perform hierarchical clustering. Compare compound profiles to a reference database of profiles from compounds with known MOAs to predict biological activity [22] [4].
Protocol for Target-Based Screening: Cell Membrane Chromatography (CMC) with MS
This protocol describes an online, affinity-based method to fish out active components from complex NP mixtures targeting specific membrane receptors [21].
- Preparation of Cell Membrane Stationary Phase (CMSP): Harvest cells expressing the target receptor of interest. Isolate cell membranes via homogenization and differential centrifugation. Immobilize the membrane fragments onto activated silica particles using a phospholipid-assisted method. Pack the CMSP into a liquid chromatography column.
- On-line Screening Setup: Connect the CMC column as the first dimension in an LC system. Connect a reverse-phase analytical column coupled to a mass spectrometer as the second dimension via a multi-port switching valve.
- Screening Run: Inject the crude natural product extract onto the CMC column. Use a mild mobile phase (e.g., isotonic PBS) to maintain receptor integrity. Compounds with no affinity for the receptor elute in the void volume and are wasted.
- Target Compound Capture & Identification: Switch the valve to trap the retained fraction (containing receptor-bound ligands) on a trapping column. Elute the bound ligands from the CMC column onto the trapping column using a denaturing organic solvent. Then, switch the valve to forward the trapped ligands to the second-dimension RP column for separation. Detect and identify the individual ligands using tandem mass spectrometry (MS/MS).
- Validation: Synthesize or purify the identified compound and validate its activity and binding affinity for the target receptor in a secondary biochemical or cellular assay.
Visualizing the Pathways and Workflows
Diagram 1: Dual Pathways in Natural Product Drug Discovery
Diagram 2: Target Deconvolution Strategies Post-Phenotypic Screen
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for Integrated NP Screening
| Reagent/Material | Primary Function | Application Context |
|---|---|---|
| Prefractionated NP Libraries | Chemically simplified extracts that reduce complexity while preserving natural chemical diversity. | Primary screening input for both phenotypic and target-based assays to improve hit resolution and deconvolution [22]. |
| Multiplex Fluorescent Stain Kits (e.g., DAPI, Phalloidin, LysoTracker, Antibody Panels) | Simultaneously label multiple organelles and cellular states for high-content imaging. | Generating multiparametric cytological profiles in phenotypic screening [22] [4]. |
| Cell Lines with Engineered Reporters | Express fluorescent or luminescent proteins under pathway-specific control (e.g., NF-κB response element). | Enabling targeted phenotypic readouts or reporter-gene assays within a cellular context. |
| Immobilized Target Proteins / CMSP Columns | Purified protein or cell membrane fragments fixed to a solid support for affinity capture. | Target-based screening via affinity chromatography, SPR, or ASMS to "fish out" ligands from mixtures [21]. |
| Biotin or Photoaffinity Tags (e.g., Diazirine, Benzophenone) | Chemical handles for conjugating a small molecule to a solid support or enabling UV-induced covalent crosslinking. | Creating chemical probes for affinity-based pull-down and target identification (chemoproteomics) [3] [23]. |
| Streptavidin-Coated Magnetic Beads | High-affinity solid support for capturing biotin-tagged chemical probes and their bound target proteins. | Isolating protein-compound complexes from lysates after affinity pull-down experiments [23]. |
| LC-MS/MS Systems | High-sensitivity analytical instrumentation for separating compounds and determining their structure or identifying proteins. | NP Research: Dereplication, compound identification. Target ID: Protein identification from pull-downs [21]. |
| Bioinformatic Software (e.g., CellProfiler, MetaboAnalyst, Clustering Algorithms) | Automated image analysis, multivariate statistical analysis, and pattern recognition for complex datasets. | Extracting quantitative features from HCS images and clustering phenotypic or metabolomic profiles to predict MOA [22] [4]. |
The dichotomy between target-based and phenotypic screening is a false crossroads. As the data and protocols illustrate, each has irreplaceable strengths and inherent blind spots. The modern imperative is for integration.
A forward-looking strategy begins with phenotypic screening of diverse NP libraries to identify extracts that produce a desirable, biologically relevant phenotype. Advanced cytological profiling can then prioritize hits and suggest a MOA [22] [4]. Following this, target deconvolution techniques—leveraging affinity pull-downs with chemical probes [3] [23] or label-free methods like thermal proteome profiling—are employed to identify the molecular target(s). Finally, target-based assays are used to characterize the compound-target interaction with precision, enabling medicinal chemistry optimization.
This synergistic cycle, leveraging phenotypic assays for discovery and target-based methods for mechanistic elucidation and optimization, bridges the gap between biological complexity and molecular precision. It represents the most robust path forward for unlocking the full therapeutic potential of natural products in the development of novel drugs for challenging diseases.
Modern Toolkits: Deploying Phenotypic Profiling and Target Engagement Assays for NPs
The discovery of bioactive molecules from natural sources is undergoing a transformative shift, moving beyond single-target assays toward system-level phenotypic profiling. This evolution addresses a core challenge in natural products research: the frequent mismatch between a compound's in vitro target affinity and its in vivo efficacy or unexpected toxicity [24] [25]. Traditional target-based screening, while precise, operates within a predefined biological understanding, potentially missing novel mechanisms and polypharmacology—a hallmark of many natural products [4]. Phenotypic screening, in contrast, begins with a measurable cellular or organismal change, agnostic to the specific molecular target, making it exceptionally powerful for discovering first-in-class therapies and novel biology [24] [26].
High-content phenotypic profiling technologies, such as Cell Painting and L1000, have matured to bridge this gap. They offer a middle ground, providing deep, multi-parametric data on compound effects that is richer than a single readout but more tractable than whole-organism studies [27] [28]. Cell Painting captures hundreds of morphological features from microscopy images, creating a visual "fingerprint" of cellular state [27]. The L1000 assay quantifies the expression of 978 "landmark" genes, from which the majority of the transcriptome can be computationally inferred, offering a complementary molecular signature of perturbation [28]. The integration of AI-driven analysis is now unlocking the full potential of these rich datasets, enabling the prediction of mechanism of action (MOA), toxicity, and the identification of promising candidates from complex natural product libraries [29] [30]. This guide provides a detailed, data-driven comparison of these two pivotal profiling platforms within the context of modern, systems-level natural products discovery.
Platform Deep Dive: Mechanisms and Methodologies
Cell Painting: Morphological Profiling at Single-Cell Resolution
Cell Painting is a high-content, image-based assay designed to provide an unbiased, comprehensive view of cellular morphology. Its protocol involves staining cultured cells with a cocktail of six fluorescent dyes to highlight eight major cellular components or organelles, which are then imaged across five fluorescence channels [27].
- Core Staining Panel: The assay uses:
- Hoechst 33342 / DAPI: Labels DNA (nucleus).
- Concanavalin A, Alexa Fluor 488 conjugate: Labels the endoplasmic reticulum and Golgi apparatus.
- Wheat Germ Agglutinin, Alexa Fluor 555 conjugate: Labels the plasma membrane and Golgi.
- Phalloidin, Alexa Fluor 568 conjugate: Labels polymerized actin (cytoskeleton).
- SYTO 14: Labels nucleoli and cytoplasmic RNA.
- MitoTracker Deep Red: Labels mitochondria [27].
- Feature Extraction: Automated image analysis software (e.g., CellProfiler) identifies individual cells and measures approximately 1,500 morphological features per cell. These features include size, shape, texture, intensity, and correlations between channels, capturing subtle phenotypic changes [27] [31].
- Key Advantage: It provides single-cell resolution, allowing researchers to detect heterogeneity in cell populations and identify phenotypic effects in specific subpopulations, which is critical for understanding complex natural product effects [27] [4].
L1000: A High-Throughput Gene Expression Profiling Platform
The L1000 platform, developed as part of the NIH LINCS Consortium, is a cost-effective, high-throughput method for gene expression profiling. It is based on a "reduced representation" strategy that measures a carefully selected set of 978 informative "landmark" transcripts, from which the expression levels of ~81% of non-measured transcripts can be accurately inferred using computational models [28].
- Assay Mechanism: The technology uses ligation-mediated amplification (LMA) followed by detection on fluorescently addressed microspheres (beads). Briefly, mRNA is captured, converted to cDNA, and amplified with barcoded, gene-specific primers. These products are hybridized to color-coded beads, and expression is quantified via a phycoerythrin signal [28].
- Scale and Cost: This bead-based approach enables massive scale; the published Connectivity Map (CMap) contains over 1.3 million L1000 profiles. The reagent cost is remarkably low, at approximately $2 per sample, making large-scale screening of natural product libraries feasible [28].
- Key Advantage: It delivers a direct molecular readout of cellular response (transcriptional changes) at a very high throughput and low cost, facilitating the creation of massive reference databases for connectivity analysis [32] [28].
The AI-Driven Analysis Layer
AI and machine learning are not standalone assays but a critical analytical layer that maximizes the value of data from both Cell Painting and L1000. Modern approaches move beyond simple clustering to predictive and generative models.
- For Image-Based Data: Advanced workflows use deep learning (e.g., convolutional autoencoders) to learn compact, informative representations from raw Cell Painting images or extracted features. These representations improve performance in key tasks like Mechanism of Action (MOA) classification and batch effect correction [26] [30]. Self-supervised anomaly detection models, trained on control well data, can identify subtle, unexpected phenotypes that might be missed by standard analysis [30].
- For Transcriptomic Data: AI models integrate L1000 data with other biological networks. For example, the Pathway and Transcriptome-Driven Drug Efficacy Predictor (PTD-DEP) uses a dual-modality architecture combining pathway prediction with graph convolutional networks (GCNs) on L1000 transcriptomic profiles to identify multi-target therapeutics, as demonstrated in the discovery of melatonin's dual anti-aging and anti-Alzheimer's activity [29].
- Integrated Analysis: The most powerful applications involve multi-modal AI that jointly learns from both morphological and gene expression profiles, along with chemical structure data, to build more robust predictors of bioactivity, toxicity, and MOA [32] [29].
Comparative Performance Analysis
A systematic, head-to-head comparison of Cell Painting and L1000 reveals distinct strengths, guiding platform selection for specific research goals in natural products discovery [32].
Technical and Performance Metrics
Table 1: Core Technical Specifications and Performance Metrics [27] [32] [28]
| Feature | Cell Painting Assay | L1000 Assay |
|---|---|---|
| Primary Readout | Cellular morphology (image-based) | Gene expression (bead-based luminescence) |
| Profiling Dimension | ~1,500 morphological features per cell | 978 directly measured landmark transcripts |
| Resolution | Single-cell | Population-averaged |
| Key Advantage | Detects spatial/organelle-level phenotypes & heterogeneity | Direct molecular signature; massive scalability |
| Typical Assay Cost | Low (dye-based) | Very Low (~$2/sample) |
| Throughput | High (384-well plate) | Very High (optimized for 384-well) |
| Data Reproducibility | Higher (Median Pairwise Correlation) | High, but slightly lower than Cell Painting |
Information Content and Predictive Utility
A landmark study profiling 1,327 compounds from the Drug Repurposing Hub in A549 cells with both assays provided quantitative insights into their information content and utility for drug discovery tasks [32].
Table 2: Comparative Information Content & Predictive Performance (Based on 1,327 Compound Study) [32]
| Metric | Cell Painting | L1000 | Interpretation for Natural Products Research |
|---|---|---|---|
| Profile Reproducibility | Higher | High | Cell Painting profiles are more consistent across replicates, crucial for reliable phenotyping of complex extracts. |
| Signal Diversity | Higher | High | Cell Painting captures a wider variety of distinct phenotypic states, better for novel MOA discovery. |
| # of Independent Feature Groups | Lower | Higher | L1000 measures more orthogonal biological axes, potentially capturing more distinct pathways. |
| MOA Classification Accuracy | Complementary | Complementary | Each assay excels for different MOA classes; combined use yields best overall prediction. |
| Sensitivity to Batch/Position Effects | Higher (requires correction) | Lower | Cell Painting requires careful normalization (e.g., spherize transform) for plate-edge effects [32]. |
Application to Natural Products Research
Phenotypic profiling is particularly suited to natural products, which often have complex, unknown, or multiple mechanisms of action [4]. A broad-spectrum cytological profiling platform using 14 cellular markers successfully classified natural products, predicted MOA (e.g., identifying topoisomerase inhibitors), and elucidated structure-activity relationships (SAR) by clustering compounds with similar phenotypic fingerprints [4]. This approach moves beyond simple cytotoxicity to a multi-parameter assessment of physiological impact, distinguishing between selective agents and broadly toxic compounds [4].
Experimental Protocols and Data Analysis
- Cell Seeding & Perturbation: Plate cells in 384-well plates. Treat with natural product compounds/extracts (typically for 48 hours).
- Staining & Fixation: Fix cells, then stain with the 6-dye cocktail as described in Section 2.1.
- Image Acquisition: Automatically image plates using a high-throughput microscope with 5 fluorescence channels.
- Image Analysis & Feature Extraction: Use CellProfiler software to identify cells and measure ~1,500 features/cell.
- Data Normalization: Apply correction for plate positional effects (e.g., spherize transform using DMSO control wells) [32] [31].
- Profiling & Analysis: Generate median profiles per well, then use similarity metrics (e.g., correlation) for clustering, MOA prediction, or anomaly detection via AI models.
- Cell Perturbation: Treat cells in 384-well plates with compounds (typically for 24 hours).
- Lysate Preparation: Lyse cells and capture mRNA on oligo-dT-coated plates.
- cDNA Synthesis & Amplification: Synthesize cDNA and perform Ligation-Mediated Amplification (LMA) with barcoded gene-specific primers.
- Bead-Based Detection: Hybridize amplified products to fluorescently color-coded beads. Quantify expression via phycoerythrin signal on a Luminex bead reader.
- Data Processing: Normalize data and infer the expression of ~81% of the transcriptome from the 978 landmark genes using a pre-trained model.
- Signature Generation & Connectivity Analysis: Generate differential expression signatures and compare them to the CMap database to identify similar profiles and hypothesize MOA.
Visualizing Workflows and Integration
Diagram 1: Cell Painting generates morphological profiles from images for AI-driven analysis.
Diagram 2: Integrated AI models combine multimodal data for enhanced prediction.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions for Phenotypic Profiling
| Item | Primary Function | Example/Note |
|---|---|---|
| Cell Painting Dye Cocktail | Multiplexed staining of organelles for morphological profiling. | Hoechst 33342, Concanavalin A-AF488, WGA-AF555, Phalloidin-AF568, SYTO 14, MitoTracker Deep Red [27]. |
| L1000 Detection Beads & Primers | Bead-based detection of 978 landmark transcripts. | Color-coded Luminex beads coupled to barcode-specific oligonucleotides [28]. |
| Reference Compound Libraries | Assay controls & training data for AI models. | Drug Repurposing Hub, LOPAC library for MOA annotation [32] [4]. |
| Normalization Controls | Corrects technical variation (plate, batch effects). | DMSO controls distributed across plates for spherize transform [32] [31]. |
| Validated Chemical Probes | Establishes disease relevance & pathway modulation in phenotypic assays [25]. | Used for assay validation and connecting targets to phenotypes. |
| AI/ML Software Platforms | Analyzes high-dimensional data for prediction & clustering. | Includes tools for deep learning on images (TensorFlow, PyTorch) and transcriptomic analysis (CMap tools) [29] [30]. |
Cell Painting and L1000 are not competing technologies but powerful orthogonal pillars of modern phenotypic profiling. For natural products research, Cell Painting offers unparalleled insight into direct cellular morphology and heterogeneity, while L1000 provides a cost-effective, scalable window into the transcriptional landscape. The choice depends on the primary research question: phenotypic characterization and novel MOA discovery favor Cell Painting, whereas large-scale library screening and connectivity mapping leverage L1000's strengths.
The future lies in their strategic integration, powered by AI-driven analysis. Combining morphological, transcriptomic, and chemical data within multi-modal AI frameworks creates a system more predictive of in vivo outcomes than any single modality [32] [29]. This integrated approach is perfectly poised to decode the complex mechanisms of natural products, accelerating the transition from hit identification to validated lead with a known phenotypic signature and a deconvoluted mechanism, ultimately enriching the pipeline for safer and more effective therapeutics.
The landscape of drug discovery has undergone a significant strategic evolution, marked by a renaissance in phenotypic screening approaches that demand sophisticated deconvolution methodologies. Historically, the pharmaceutical industry heavily favored target-based screening, where compounds were tested against isolated, purified proteins with known disease relevance. While this approach benefits from straightforward chemistry optimization and clear intellectual property pathways, analyses reveal its limitations in generating first-in-class medicines [1]. The fundamental challenge lies in the reductionist nature of target-based methods, which often fail to capture the complex pathophysiology of disease as it manifests in living systems.
In contrast, phenotypic screening observes compound effects in cells, tissues, or whole organisms, producing hits that modulate a disease-relevant phenotype without prior bias toward a specific molecular target [33]. This approach operates within a physiologically relevant context, accounting for compound permeability, metabolism, and off-target effects early in discovery. For natural products research—where compounds often possess complex structures and unknown mechanisms—phenotypic screening is particularly valuable as it allows biological activity to guide discovery without requiring target hypotheses [34].
However, the major challenge following a phenotypic hit is target deconvolution: identifying the specific biomolecule(s) through which the compound exerts its effect. This process transforms a phenotypic observation into mechanistic understanding, enabling medicinal chemistry optimization, predictive toxicology, and intellectual property protection [35]. The "deconvolution revolution" refers to the expanding toolkit of chemical, proteomic, genetic, and computational strategies that have matured to address this critical bottleneck, making phenotypic screening a more powerful and reliable discovery engine [36].
Core Deconvolution Strategies: A Comparative Framework
Target deconvolution strategies can be broadly categorized into affinity-based methods, which rely on the direct physical interaction between compound and target, and functional inference methods, which deduce targets through analysis of downstream biological effects [34]. The choice of strategy depends on compound properties, available instrumentation, and the biological system.
Table 1: Comparison of Major Target Deconvolution Strategies
| Method | Core Principle | Typical Timeframe | Key Advantages | Major Limitations | Best Suited For |
|---|---|---|---|---|---|
| Affinity Chromatography [34] | Immobilized compound pulls down binding proteins from lysate. | 2-4 weeks | Direct, conceptually simple; can detect weak binders with cross-linking. | Requires compound derivatization which may alter activity/selectivity; high background common. | Stable, potent compounds with known site for linker attachment. |
| Activity-Based Protein Profiling (ABPP) [34] | Reactive probe labels active-site nucleophiles of enzyme families. | 1-3 weeks | Reports on enzyme activity (not just abundance); can profile entire enzyme families. | Limited to enzymes with susceptible nucleophiles (e.g., Ser, Cys hydrolases); requires probe design. | Covalent inhibitors or modulators of specific enzyme classes (proteases, lipases). |
| Photoaffinity Labeling (PAL) [36] | Photoreactive compound crosslinks to proximal proteins upon UV irradiation. | 3-6 weeks | Captures transient, low-affinity interactions in live cells; can map binding sites. | Synthesis of bifunctional (photoreactive + handle) probes is challenging; potential for non-specific labeling. | Compounds where binding site is tolerant to modification; studying membrane proteins. |
| Cellular Thermal Shift Assay (CETSA) | Target protein stabilization upon compound binding measured via thermostability. | 1-2 weeks | Label-free; works in cells and tissues; can monitor target engagement. | Does not identify novel/unknown targets; requires antibody or MS readout. | Validation of suspected targets and engagement studies. |
| Genomic Profiling (CRISPR, RNAi) [36] | Identification of genetic alterations that confer resistance or sensitivity to the compound. | 4-8 weeks | Unbiased, genome-wide; can identify pathways, not just single proteins. | Labor-intensive; hits may be indirect; resistance mutations can be rare. | Compounds with strong, selective phenotype in proliferating cells. |
| Transcriptomic/Proteomic Profiling [35] | Comparison of gene or protein expression signatures to reference databases. | 2-3 weeks | Label-free; provides MoA context and pathway information. | Identifies downstream consequences, not direct binders; requires robust signature. | Elucidating pathway-level mechanism of action (MoA). |
Detailed Experimental Protocols for Key Methods
This protocol merges affinity purification with photoaffinity labeling to capture lower-affinity interactions.
- Probe Synthesis: Derivatize the hit compound to incorporate both a photoreactive group (e.g., diazirine, benzophenone) and a bio-orthogonal handle (e.g., alkyne). The modification site is informed by structure-activity relationship (SAR) data to minimize activity loss.
- Cell Treatment and Crosslinking: Treat live cells or use cell lysates with the probe (1-10 µM). For live cells, incubate to allow cellular uptake (1-6 hours). Irradiate sample with UV light (~365 nm for diazirine) to induce crosslinking.
- Click Chemistry Conjugation: Lyse cells if using live cells. React the alkyne on the crosslinked probe with an azide-bearing affinity tag (e.g., azide-biotin) via copper-catalyzed azide-alkyne cycloaddition (CuAAC).
- Affinity Enrichment: Incubate the lysate with streptavidin-coated magnetic beads to capture biotinylated protein complexes. Wash stringently to remove non-specific binders.
- Elution and Identification: Elute proteins via boiling in SDS-PAGE buffer or competitive biotin elution. Resolve by gel electrophoresis, stain, and excise bands for in-gel tryptic digestion. Analyze resulting peptides by liquid chromatography-tandem mass spectrometry (LC-MS/MS).
Multiplexed Proteomic Profiling Using Tandem Mass Tag (TMT) Labeling
This label-free quantitative proteomics protocol identifies proteins whose abundance or state changes in response to compound treatment.
- Sample Preparation: Treat multiple cell populations (e.g., vehicle, hit compound, inactive analog) in biological triplicate. Harvest cells and lyse.
- Protein Digestion and Labeling: Reduce, alkylate, and digest lysates with trypsin. Label the resulting peptides from each sample with a unique isobaric TMT reagent.
- Pooling and Fractionation: Combine all TMT-labeled samples into one tube. Fractionate the pooled sample via high-pH reverse-phase chromatography to reduce complexity.
- LC-MS/MS Analysis: Analyze each fraction by nano-flow LC-MS/MS on an Orbitrap mass spectrometer. Quantify peptide abundance by measuring the intensity of reporter ions released during MS2 fragmentation.
- Data Analysis and Target Inference: Use bioinformatics to identify proteins significantly upregulated or downregulated by the active compound versus controls. Compare the resulting protein signature to databases of known drug profiles or genetic perturbations to infer potential targets and pathways.
Performance Benchmarking: Success Rates and Operational Metrics
The effectiveness of a deconvolution strategy is measured by its success rate, throughput, and resource requirements. Data aggregated from published campaigns and service providers like CDI Labs (offering HuProt microarray services) reveal distinct profiles for each method [37].
Table 2: Performance Benchmarking of Deconvolution Methods
| Metric / Method | Affinity Chromatography | Photoaffinity Labeling | Genomic Screening | Omics Profiling | HuProt Microarray [37] |
|---|---|---|---|---|---|
| Reported Success Rate | ~30-40% | ~40-60% | ~20-30% | ~15-25% (for direct target ID) | >70% (for antibody targets) |
| Primary Output | Direct binding protein(s) | Direct binding protein(s) | Gene(s) whose loss alters compound sensitivity | Pathway/expression signature | Direct binding protein(s) |
| Throughput (Samples/Week) | Low (2-5) | Low (2-5) | Medium (10-20) | High (50+) | Very High (100+) |
| Specialized Equipment Needed | MS, HPLC | MS, UV Crosslinker | NGS platform, robotic automation | MS or NGS platform | Microarray scanner |
| Relative Cost per Experiment | High | Very High | Medium-High | Medium | Low-Medium |
| Key Failure Point | Poor probe activity/retention | Non-specific crosslinking | Weak/no resistance phenotype | Indirect signature; noisy data | Limited to soluble folded proteins |
For natural products, which are often chemically complex and difficult to modify, label-free approaches like genomic and transcriptomic profiling offer an attractive starting point, despite their generally lower direct identification rates. The integration of artificial intelligence for pattern recognition in omics data is a developing frontier aimed at improving these success rates [38].
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful target deconvolution relies on specialized reagents and platforms.
Table 3: Key Research Reagent Solutions for Target Deconvolution
| Reagent/Platform | Supplier/Example | Primary Function in Deconvolution | Key Consideration |
|---|---|---|---|
| Alkyne/Azide-Tagged Building Blocks | Click Chemistry Tools (e.g., Alkynyl linkers, Azide-PEG3-Biotin) | Enable bio-orthogonal "click" conjugation of affinity/fluorescent tags to probe molecules for enrichment or visualization. | Choice of linker length and polarity is crucial to maintain probe cell permeability and target affinity. |
| Photoreactive Crosslinkers | Thermo Fisher (e.g., Succinimidyl-diazirine), Sigma-Aldrich | Provide benzophenone, diazirine, or aryl azide groups for incorporation into probes to capture protein-compound interactions. | Diazirines offer smaller size and activation at longer, less damaging UV wavelengths (~350 nm). |
| Activity-Based Probes (ABPs) | Custom synthesis or broad-spectrum probes (e.g., FP-TAMRA for serine hydrolases). | Covalently label active enzymes in complex proteomes, enabling enrichment and identification of compound targets within specific enzyme classes. | Specificity of the reactive "warhead" determines which enzyme family is profiled. |
| HuProt Human Proteome Microarray [37] | CDI Labs | A high-density array containing thousands of purified human proteins for directly screening compound or antibody binding in a non-cellular context. | Excellent for identifying high-affinity binders but may miss targets requiring cellular context (e.g., membrane proteins in native lipid environment). |
| CRISPR Knockout or Activation Libraries | Broad Institute GECCO, Sigma MISSION | Genome-wide pooled libraries to identify genes whose knockout confers resistance or sensitivity to the compound, implicating them in the MoA. | Requires a strong, selectable phenotype (e.g., cell death or proliferation) for effective screening. |
| Isobaric Mass Tagging Kits | Thermo Fisher TMT, SciEx iTRAQ | Enable multiplexed quantitative proteomics by labeling peptides from different conditions for simultaneous MS analysis, improving throughput and accuracy. | The degree of multiplexing (e.g., 6-plex, 11-plex) balances throughput with quantitative depth and cost. |
Integrated Workflows and Future Outlook
The most successful modern deconvolution campaigns rarely rely on a single method. An integrated, iterative workflow is considered best practice. A typical cascade might begin with a label-free, unbiased method like transcriptomic profiling or a genetic screen to generate a shortlist of candidate targets and pathways [24]. This list is then prioritized using bioinformatics and existing biological knowledge. High-priority candidates are subsequently validated using direct binding methods such as affinity chromatography or CETSA, and finally confirmed through phenotypic rescue experiments (e.g., showing that overexpression of the putative target negates the compound's effect).
The future of deconvolution is being shaped by converging technologies. Deep learning models are increasingly adept at predicting drug-target interactions from chemical structure and omics data patterns, helping to prioritize candidates from large screening datasets [38]. Furthermore, the line between phenotypic and target-based screening is blurring with strategies like in-cell fragment-based ligand discovery, where libraries of photo-crosslinkable fragments are applied to cells, combining the unbiased nature of phenotypic screening with direct proteomic readout of binding events [36].
For the natural products researcher, this expanding toolkit is empowering. It provides a structured pathway to move from a fascinating biological activity isolated from a complex extract to a defined molecular mechanism—a journey that is central to validating the relevance of the finding and unlocking its full therapeutic potential.
Natural Products (NPs) remain an indispensable source of novel therapeutics, with nearly half of FDA-approved small-molecule drugs from 1981 to 2019 being derived from or inspired by NPs [39]. The discovery of these drugs has historically been driven by two complementary paradigms: phenotypic screening and target-based screening [33].
Phenotypic screening, which measures a compound's effect in cells, tissues, or whole organisms without prior knowledge of its molecular target, has proven particularly successful for identifying first-in-class medicines [1]. This approach is advantageous for NP research as it identifies bioactive compounds based on a relevant biological effect, accommodating the complex mechanisms and polypharmacology often exhibited by NPs [40]. However, a major challenge following a phenotypic "hit" is target deconvolution—identifying the specific protein target(s) responsible for the observed activity [41]. Without this knowledge, lead optimization and understanding of potential off-target effects are severely hindered.
Target-based screening, in contrast, tests compounds against a predefined, purified protein target. While this allows for rational drug design and high-throughput screening, it risks selecting compounds that are ineffective in a physiologically relevant cellular environment due to issues like poor permeability or metabolic instability [33].
This is where Cellular Thermal Shift Assay (CETSA) and related label-free biophysical methods create a critical bridge. They directly address the core challenge of target engagement validation—providing evidence that a drug candidate physically binds to its intended target within the complex native environment of a living cell [42]. For NPs identified through phenotypic screens, CETSA offers a path to directly identify and validate their molecular targets without requiring chemical modification of the often complex and fragile NP structure, thereby preserving its native bioactivity [39] [40].
Core Principle: Thermal Stability as a Proxy for Target Engagement
The foundational principle underlying CETSA and related assays is ligand-induced thermal stabilization. When a small molecule ligand binds to its target protein, it typically stabilizes the protein's three-dimensional conformation, making it more resistant to heat-induced denaturation and aggregation [42]. This measurable increase in thermal stability (manifested as a shift in the protein's melting temperature, (T_m)) serves as a direct proxy for physical drug-target binding.
The standard CETSA protocol involves four key steps [42]:
- Compound Incubation: Live cells, tissues, or cell lysates are treated with the drug compound or a control vehicle.
- Heat Challenge: Samples are subjected to a gradient of elevated temperatures.
- Separation: Denatured, aggregated proteins are separated from soluble, folded proteins (typically via centrifugation or filtration).
- Detection & Quantification: The remaining soluble target protein is quantified. A higher amount of soluble protein at a given temperature in the drug-treated sample versus the control indicates ligand-induced stabilization and confirms target engagement.
This principle extends to other label-free methods, such as Drug Affinity Responsive Target Stability (DARTS), which measures protection from proteolysis, and Stability of Proteins from Rates of Oxidation (SPROX), which measures protection from methionine oxidation [39]. These methods all detect changes in a protein's biophysical stability upon ligand binding, providing a label-free strategy for target identification.
Comparative Analysis of CETSA Formats and Alternative Methods
The evolution of CETSA from a Western blot-based experiment to a suite of proteome-wide profiling tools offers researchers a range of options tailored to different stages of the drug discovery pipeline. The table below compares the key formats of CETSA and alternative thermal shift methods.
Table 1: Comparison of CETSA Formats and Alternative Label-Free Target Engagement Assays
| Method | Core Principle | Detection Mode | Throughput & Scale | Key Advantages | Primary Limitations | Best Application in NP Research |
|---|---|---|---|---|---|---|
| WB-CETSA [40] [42] | Ligand-induced thermal shift. | Antibody-based (Western Blot). | Low; single target. | Simple, accessible, works in intact cells and lysates. | Requires high-quality antibody; low throughput; hypothesis-driven. | Validation of suspected/predicted targets. |
| MS-CETSA / Thermal Proteome Profiling (TPP) [39] [40] | Ligand-induced thermal shift. | Mass spectrometry (LC-MS/MS). | Medium; proteome-wide (7,000+ proteins). | Unbiased, identifies on/off-targets, no antibody needed. | Expensive, complex data analysis, lower sensitivity for very low-abundance proteins. | Unbiased target deconvolution for NPs of unknown mechanism. |
| High-Throughput CETSA (HT-CETSA) [42] [43] | Ligand-induced thermal shift. | Plate-based (e.g., Split-Luciferase, AlphaLISA). | High; 384-/1536-well format, single target. | Excellent for screening large compound libraries, quantitative EC50. | Requires protein tagging or antibody pairs; not for endogenous proteins in HT format. | Lead optimization and SAR studies for NP-derived compounds. |
| Isothermal Dose-Response CETSA (ITDR-CETSA) [39] | Dose-dependent stabilization at fixed temperature. | WB, MS, or HT. | Medium. | Generates binding affinity (EC50) data in cells. | Typically applied to a single or limited number of targets. | Ranking compound potency and cellular permeability. |
| Drug Affinity Responsive Target Stability (DARTS) [39] | Ligand-induced protection from proteolysis. | WB or MS. | Low to Medium. | Simple, minimal equipment, no compound modification. | Sensitivity depends on protease choice; challenging for low-abundance targets. | Initial target identification in cell lysates. |
| Stability of Proteins from Rates of Oxidation (SPROX) [39] | Ligand-induced protection from methionine oxidation. | Mass spectrometry. | Medium. | Can provide binding site information (domain-level). | Limited to methionine-containing peptides; requires MS expertise. | Studying domain-specific interactions and weak binders. |
For research on complex NP mixtures, MS-CETSA (TPP) is particularly powerful. Its unbiased nature allows for the identification of the full spectrum of protein targets engaged by a mixture, which is crucial for understanding polypharmacology and potential synergistic effects [40]. A notable application is the profiling of the flavonoid quercetin, where CETSA-MS identified 70 putative direct cellular targets, including both stabilized (e.g., CBR1, GSK3A) and destabilized (e.g., MAPK1) proteins, vastly expanding the understanding of its complex mechanism of action [44].
Detailed Experimental Protocol: A Representative MS-CETSA (TPP) Workflow
The following protocol outlines a standard Thermal Proteome Profiling (TPP) experiment for the unbiased identification of NP targets in intact cells [39] [44].
1. Sample Preparation:
- Cell Culture & Treatment: Grow adherent or suspension cells (e.g., HEK293T). Divide into two pools: one treated with the NP (dissolved in DMSO or suitable solvent) and one with vehicle control. Typical treatment involves 1-2 hour incubation at physiologically relevant concentrations [44].
- Heating: Aliquot each pool (drug-treated and control) into multiple PCR tubes. Use a thermal cycler to heat each aliquot to a different precise temperature across a gradient (e.g., from 37°C to 67°C in 3-4°C increments). Include an unheated control (37°C).
2. Protein Solubilization and Digestion:
- Lysis: Lyse heated cells using a combination of freeze-thaw cycles and detergent (e.g., NP-40) to liberate soluble proteins. Remove aggregated proteins by high-speed centrifugation.
- Protein Precipitation and Digestion: Precipitate soluble proteins from the supernatant (e.g., using acetone). Redissolve and digest the protein pellet into peptides using a protease like trypsin.
3. Mass Spectrometric Analysis:
- Peptide Labeling (Optional): For multiplexed TMT (Tandem Mass Tag) TPP, label peptides from different temperature points with isobaric tags, pool them, and fractionate by high-pH reverse-phase chromatography [40].
- LC-MS/MS: Analyze peptides via liquid chromatography coupled to a tandem mass spectrometer (LC-MS/MS). For label-free TPP, samples from each temperature are run separately [44].
4. Data Processing and Target Identification:
- Protein Quantification: Use software (e.g., MaxQuant, Proteome Discoverer) to identify and quantify proteins/peptides.
- Melting Curve Fitting: For each protein, plot the normalized amount of soluble protein remaining versus temperature for both drug-treated and control samples. Fit a sigmoidal melting curve to the data.
- Hit Selection: Calculate the melting point ((Tm)) for each protein. Proteins showing a statistically significant shift in (Tm) ((ΔT_m)) between drug and control conditions are considered putative direct binding targets. Software packages like the R package
Inflectare designed specifically for this analysis [44].
Integrating CETSA into the NP Drug Discovery Pipeline: A Strategic Workflow
CETSA is not a standalone technique but a powerful connector within a broader discovery strategy. The following diagram illustrates how different CETSA formats integrate with phenotypic and target-based approaches to form a cohesive pipeline for NP drug discovery.
CETSA Bridges Phenotypic Screening with Target-Based Validation
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagent Solutions for CETSA Experiments
| Reagent / Material | Function in CETSA | Key Considerations & Examples |
|---|---|---|
| Cell Lines | Provide the physiologically relevant native environment for target engagement. Can be wild-type, engineered, or primary cells. | Choice depends on target expression and relevance to disease (e.g., HEK293T for general studies, cancer lines for oncology NPs) [44]. |
| Natural Product Compound | The molecule of interest whose target is being investigated. | Critical to use a pure, well-characterized compound. Solubility in DMSO or buffer must be optimized to avoid non-specific effects [40]. |
| Lysis Buffer | Disrupts cell membranes after heating to release soluble, non-denatured proteins. | Typically contains detergent (e.g., NP-40, IGEPAL) and protease/phosphatase inhibitors. Must be compatible with downstream detection [43]. |
| Protein Detection System | Quantifies the soluble target protein remaining post-heat challenge. | WB: Specific antibodies [42]. MS: Trypsin for digestion, LC-MS/MS system [44]. HT: Split-Luciferase tags or antibody pairs (AlphaLISA) [43]. |
| Thermal Cycler | Provides accurate and controlled heating of multiple samples across a temperature gradient. | Essential for generating precise melting curves. Must have a heated lid to prevent condensation [39]. |
| Centrifuge | Separates aggregated (denatured) proteins from the soluble protein fraction after lysis. | Requires high speed (e.g., 20,000 x g) and temperature control to maintain sample integrity [39]. |
| Data Analysis Software | Processes raw data to calculate protein abundance and melting curves ((T_m)). | MS Data: MaxQuant, Proteome Discoverer. Curve Fitting & (T_m) Calculation: R packages (TPP, Inflect), dedicated commercial software [44]. |
CETSA and related thermal shift assays have fundamentally changed the approach to target engagement validation in natural product research. By providing a label-free, physiologically relevant, and scalable method to directly observe drug-target interactions, CETSA effectively bridges the gap between the phenotypic discovery of bioactive NPs and the target-based rationalization of their mechanism.
The integration of MS-CETSA (TPP) allows for the unbiased deconvolution of targets for NPs with unknown mechanisms, revealing their often complex polypharmacology [44]. Meanwhile, HT-CETSA formats accelerate the optimization of NP-derived leads by providing cellular target engagement data as a key parameter in structure-activity relationships [42] [43].
The future of CETSA in NP research lies in continued technological refinement—such as improved sensitivity for low-abundance targets and streamlined data analysis pipelines [45]—and its deeper integration with other 'omics' technologies. As part of a holistic strategy, CETSA strengthens the critical link between the observed phenotypic effect of a natural product and its underlying molecular targets, de-risking the development of novel therapeutics derived from nature's chemical treasury [40] [46].
Leveraging AI and Multi-Omics for Predictive Bioactivity and MoA Elucidation
The discovery of first-in-class medicines has historically been propelled by two divergent strategic philosophies: target-based and phenotypic drug discovery [1]. A seminal analysis revealed that between 1999 and 2008, phenotypic screening strategies were responsible for the discovery of a greater proportion of first-in-class small-molecule medicines compared to target-based approaches [1]. The principal rationale for this success is the unbiased identification of the molecular mechanism of action (MMOA). Phenotypic assays, which measure compound effects in cells, tissues, or whole organisms without preconceived molecular targets, allow for the discovery of novel biology and unexpected therapeutic mechanisms [1]. In contrast, target-based approaches begin with a hypothesis-driven selection of a specific protein or pathway believed to be central to a disease, screening for compounds that modulate its activity in isolation [1].
This historical context frames a central thesis in modern natural products research: while natural products are unparalleled in their structural complexity and proven therapeutic value [6], their study is hampered by the very challenges each screening paradigm seeks to address. Target-based screening of complex natural extracts is often confounded by mixture complexity and unknown interfering compounds, whereas phenotypic screening with natural products frequently leads to a "target deconvolution bottleneck"—the difficult and time-consuming process of identifying the precise molecular target responsible for the observed phenotype [6] [47].
The integration of Artificial Intelligence (AI) and multi-omics technologies promises to resolve this historical dichotomy. By providing a systems-level, data-rich framework, these tools can bridge the gap between the unbiased discovery power of phenotypic assays and the mechanistic clarity of target-based approaches, creating a new, synergistic paradigm for elucidating the bioactivity and mechanisms of action (MoA) of natural products [48] [49] [50].
Comparative Analysis: Target-Based vs. Phenotypic Screening in Natural Product Research
The following table provides a systematic comparison of the two primary screening strategies, highlighting their respective advantages, limitations, and suitability within natural product discovery campaigns.
Table 1: Comparative Analysis of Target-Based and Phenotypic Screening Paradigms
| Aspect | Target-Based Screening | Phenotypic Screening | Impact on Natural Products Research |
|---|---|---|---|
| Primary Approach | Hypothesis-driven; assays designed around a purified protein or known pathway [1]. | Empirical; measures a holistic cellular or organismal response (e.g., cell death, morphology change) [1] [47]. | Target-based is challenged by extract complexity; Phenotypic is ideal for discovering novel bioactivity from mixtures [6]. |
| Mechanistic Insight | High at the outset; target and MoA are predefined [1]. | Low initial insight; requires subsequent target deconvolution [1]. | Major bottleneck for natural products; deconvoluting the active component and its target is non-trivial [6]. |
| Success Rate for First-in-Class Drugs | Historically lower compared to phenotypic approaches for novel mechanisms [1]. | Historically higher for discovering first-in-class medicines with novel MoAs [1]. | Aligns with the historical role of natural products as sources of novel therapeutics with unique mechanisms [6]. |
| Throughput & Cost | Typically high-throughput and automatable with recombinant proteins. | Can be lower throughput, more complex, and costly due to cell/tissue culture. | High-throughput target screening of fractionated libraries is common; phenotypic screening often used for prioritized extracts [6]. |
| Risk of Artifacts | Prone to identifying hits that are non-bioactive in cells (e.g., assay interference compounds). | Identifies compounds active in a physiological context, filtering for cell permeability and toxicity. | Critical for natural products, which may contain promiscuous binders or fluorescent compounds that disrupt target assays. |
| Data Integration Potential | Generates simple, quantitative data (e.g., IC50, Ki) suitable for cheminformatics. | Generates complex, multidimensional data (e.g., imaging, biomarker changes) suitable for multi-omics. | Phenotypic data is rich fodder for AI/ML models that can predict MoA from complex response patterns [49] [50]. |
| Thesis Context | Represents the molecular, reductionist pole of the discovery spectrum. | Represents the systems-level, holistic pole of the discovery spectrum. | AI and multi-omics serve as the integrator, extracting target hypotheses from phenotypic data and validating them in a systems context [48] [51]. |
AI and Multi-Omics as an Integrative Solution
The convergence of AI and multi-omics creates a powerful scaffold to overcome the limitations of both traditional screening approaches, particularly for complex natural products. Multi-omics provides the layered molecular description of a system's response to a perturbation, while AI offers the computational tools to integrate and extract meaning from these vast, heterogeneous datasets [49] [51].
Table 2: AI and Multi-Omics Technologies for Bridging Screening Paradigms
| Technology | Core Function | Application in Natural Product MoA Elucidation | Supporting Data/Performance |
|---|---|---|---|
| Network-Based Integration | Integrates multi-omics data (genomics, proteomics, etc.) onto biological interaction networks (PPI, metabolic) [50]. | Places natural product-induced changes within the context of cellular pathways. Identifies key network nodes (proteins/genes) as putative targets [50]. | Methods like graph neural networks prioritize dysregulated network modules. Case studies show use in target identification and drug repurposing [50]. |
| Deep Learning for Pattern Recognition | Uses neural networks to identify complex, non-linear patterns in high-dimensional data [48] [49]. | Analyzes phenotypic screening data (e.g., high-content imaging) or multi-omics profiles to predict MoA classes or specific targets. | AI models can classify unknown compounds by MoA based on transcriptional or proteomic signatures with high accuracy [49]. |
| Generative AI & In Silico Chemistry | Generates novel molecular structures or predicts the properties of natural product analogues [48]. | Expands upon discovered natural product scaffolds; predicts bioavailability or toxicity; designs optimized derivatives. | Used for de novo molecular design and predicting ADMET properties, accelerating lead optimization [48]. |
| Explainable AI (XAI) | Makes AI model decisions interpretable to humans (e.g., highlighting which omics features drove a prediction) [49]. | Critical for building scientific trust. Reveals which genes, proteins, or pathways the model associates with a natural product's activity. | Techniques like SHAP (SHapley Additive exPlanations) quantify feature importance, providing a hypothesis for experimental validation [49]. |
| Genome Mining & CRISPR-Cas | Identifies biosynthetic gene clusters (BGCs) in microbial genomes and uses CRISPR to activate silent BGCs [52]. | Predicts the chemical potential of a microbial strain and enables the discovery of new natural products by activating silent pathways. | Key strategy in modern microbial NP research to overcome "rediscovery" and access cryptic metabolites [52]. |
Core Experimental Protocols for Integrated Workflows
The effective integration of these technologies requires standardized methodological workflows. Below are detailed protocols for two key experiments that sit at the intersection of phenotypic screening and multi-omics/AI analysis.
Protocol 1: Multi-Omics Profiling for Target Hypothesis Generation from Phenotypic Hits
This protocol details the steps to transition from a natural product showing phenotypic activity in a cell-based assay to generating testable hypotheses about its molecular target(s) using an unbiased multi-omics approach.
Phenotypic Screening & Hit Selection:
- Perform a high-content phenotypic screen (e.g., measuring cell viability, morphology, or a specific reporter) against a library of purified natural products or fractionated extracts [47].
- Select confirmed hits based on robust, dose-dependent activity in the phenotypic assay.
Sample Preparation for Multi-Omics:
- Treat isogenic cell cultures with the natural product hit at its IC50/EC50 and a vehicle control (DMSO) for a relevant time point (e.g., 6, 12, 24 hours).
- Harvest cells in triplicate. Split the cell pellet into aliquots for parallel omics analyses:
- Transcriptomics: Isolate total RNA for RNA-seq library preparation.
- Proteomics & Phosphoproteomics: Lyse cells in appropriate buffer for mass spectrometry (MS)-based analysis. Enrich for phosphopeptides if pathway activation is of interest.
- Metabolomics: Quench metabolism and extract polar and non-polar metabolites for LC-MS analysis.
Multi-Omics Data Acquisition:
- Transcriptomics: Perform paired-end sequencing on a next-generation sequencer (e.g., Illumina). Align reads to the reference genome and quantify gene expression.
- Proteomics: Analyze peptides via liquid chromatography-tandem mass spectrometry (LC-MS/MS). Use data-dependent acquisition (DDA) or data-independent acquisition (DIA) modes for protein quantification.
- Metabolomics: Run samples on high-resolution LC-MS platforms. Identify and quantify metabolites by matching to spectral libraries.
Data Integration & Network Analysis:
- Perform differential analysis for each omics layer to identify genes, proteins, and metabolites significantly altered by treatment.
- Integrate signatures using a network-based method [50]. For example, map differentially expressed genes and proteins onto a Protein-Protein Interaction (PPI) network.
- Use network propagation algorithms to identify densely connected subnetworks or "modules" most perturbed by the natural product. The hub genes/proteins within these modules represent high-priority candidate targets or critical effector nodes in the MoA.
AI-Powered MoA Prediction & Prioritization:
- Input the differential multi-omics signature (a vector of gene/protein/metabolite changes) into a pre-trained deep learning model designed for MoA classification [49].
- Use Explainable AI (XAI) tools on the model's output to identify the top features (e.g., specific pathway genes) contributing to the prediction.
- Combine network analysis results with AI predictions to generate a final, prioritized list of 3-5 candidate molecular targets for experimental validation.
Protocol 2: AI-Guided Target Validation and Mechanism Mapping
This protocol follows the generation of target hypotheses, detailing how to use AI and focused experiments to validate the target and map the downstream mechanism.
In Silico Molecular Docking & Binding Site Prediction:
- Obtain or generate a 3D structure of the prioritized candidate target protein (e.g., from AlphaFold or PDB) [48].
- Perform molecular docking simulations using the known or predicted structure of the natural product. Use AI-enhanced docking programs to score binding poses and predict interaction affinity.
- Identify the most promising protein-ligand complex for further analysis.
Cellular Target Engagement Assays:
- Design a Cellular Thermal Shift Assay (CETSA) or Drug Affinity Responsive Target Stability (DARTS) experiment.
- Treat cells with the natural product, lyse them, and either heat (CETSA) or digest with protease (DARTS). A shift in protein stability or digestibility indicates direct target engagement.
- Detect the candidate target protein via western blot or MS. Successful engagement supports the target hypothesis.
Functional Validation via CRISPR-Cas9 or RNAi:
- Use CRISPR-Cas9 to generate a knockout (KO) cell line for the candidate target gene or RNAi for knockdown (KD) [52].
- Test the natural product's phenotypic effect in the KO/KD cells versus wild-type controls. Significant attenuation of activity in the KO/KD line provides strong functional evidence for the target's role in the MoA.
Mechanistic Mapping via Targeted Proteomics/Phosphoproteomics:
- Based on the initial multi-omics data and validated target, hypothesize the downstream signaling pathway.
- Design a targeted MS (MRM/PRM) assay or a phospho-specific antibody array to quantitatively monitor key pathway nodes (e.g., kinase phosphorylation states) over a time course of natural product treatment.
- This data refines the mechanistic model, confirming pathway inhibition or activation.
Results Integration and Model Refinement:
- Integrate all validation data (docking pose, CETSA/DARTS results, KO/KD phenotype, targeted pathway data) into a unified mechanistic model.
- Use this model to refine the AI classifiers by adding the newly generated high-confidence data to their training sets, improving future predictions for similar compounds.
Visualization of Integrated Workflows and Relationships
The following diagrams, generated using Graphviz DOT language, illustrate the core logical relationships and experimental workflows described in this guide.
Diagram 1: The AI-Multi-Omics Bridge Between Screening Paradigms
Title: AI-Multi-Omics Connects Phenotypic and Target-Based Screening
Diagram 2: Integrated MoA Elucidation Workflow
Title: Stepwise Workflow for Natural Product MoA Discovery
The Scientist's Toolkit: Essential Reagents & Solutions
Successful execution of the integrated workflows described above relies on a suite of specialized reagents and platforms. The following table details key solutions for researchers embarking on AI and multi-omics-enabled natural product discovery.
Table 3: Essential Research Reagent Solutions for Integrated Workflows
| Tool Category | Specific Item/Kit | Primary Function | Relevance to Thesis |
|---|---|---|---|
| Phenotypic Screening | High-Content Imaging (HCI) Reagents (e.g., fluorescent viability, apoptosis, organelle dyes). | Enable multiplexed, quantitative readouts of cell state in response to natural product treatment. | Generates the complex, multidimensional data that is the starting point for AI/ML analysis and MoA prediction [47]. |
| Multi-Omics Sample Prep | TriZol or equivalent monophasic phenol-guanidine reagent. | Simultaneous extraction of RNA, DNA, and protein from a single sample, preserving compatibility for multi-omics. | Ensures all omics layers are analyzed from the same biological sample, reducing variability for integration [49]. |
| Magnetic bead-based kits for phosphopeptide enrichment. | Isolates phosphorylated peptides from complex lysates for phosphoproteomics by LC-MS. | Critical for mapping signaling pathway perturbations, a key part of mechanism elucidation for many natural products. | |
| Omics Data Acquisition | Next-Generation Sequencing (NGS) library prep kits (e.g., for RNA-seq). | Convert RNA into sequencer-ready libraries to generate transcriptomic profiles. | Provides the foundational genomics/transcriptomics layer for integration and network analysis [50]. |
| Tandem Mass Tag (TMT) or isobaric labeling kits for proteomics. | Multiplex samples for quantitative proteomics, increasing throughput and reducing run-to-run variation. | Enables precise quantification of protein abundance changes across multiple treatment conditions or time points. | |
| Bioinformatics & AI | Commercial or open-source software platforms (e.g., GenePattern, Galaxy, KNIME). | Provide user-friendly interfaces and workflows for multi-omics data processing, normalization, and basic integration. | Lowers the computational barrier for researchers to begin integrating disparate data types [51]. |
| Access to pre-trained AI/ML models for MoA prediction (e.g., via repositories like GitHub or ModelHub). | Allow researchers to input their omics signatures and obtain predictions without building models from scratch. | Accelerates the hypothesis generation step, directly linking phenotypic/omics data to potential mechanisms [48] [49]. | |
| Target Validation | Cellular Thermal Shift Assay (CETSA) kits. | Provide optimized buffers and protocols to detect drug-target engagement in intact cells. | Offers direct experimental evidence linking a natural product to its hypothesized protein target, validating AI predictions. |
| CRISPR-Cas9 gene editing kits (e.g., synthetic gRNAs, Cas9 protein/expression plasmids). | Enable rapid generation of knockout cell lines for candidate target genes. | Provides the most definitive functional validation of a target's role in the observed phenotype [52]. |
The search for bioactive compounds within traditional medicine repositories presents a fundamental methodological choice: target-based screening versus phenotypic screening. Target-based approaches, focused on isolated molecular targets like G protein-coupled receptors (GPCRs), offer mechanistic clarity and high-throughput potential. In contrast, phenotypic assays, which observe effects in whole cells or organisms, better capture the complex systems biology and multi-target synergy often inherent to traditional remedies but may obscure the precise mechanisms of action [53].
This comparison guide evaluates the genome-wide pan-GPCR screening platform as a powerful hybrid strategy within this thesis. By enabling the systematic profiling of complex natural product mixtures against the entire repertoire of human GPCRs (the "GPCRome"), this platform merges the specificity of target-based methods with a breadth capable of illuminating the polypharmacology of traditional medicines [53] [54]. Approximately one-third of all marketed drugs target GPCRs, yet only about 15% of the over 800 human GPCRs are modulated by existing therapeutics, leaving a vast untapped resource for drug discovery [53] [55]. This guide provides an objective comparison of this platform's performance against alternative screening paradigms, supported by experimental data and protocols.
Comparative Analysis: Target-Based, Phenotypic, and Pan-GPCR Screening
The following table summarizes the core characteristics, advantages, and limitations of the three primary screening philosophies in natural products research.
Table: Comparison of Screening Paradigms for Traditional Medicine Research
| Aspect | Target-Based (Single GPCR) | Phenotypic (Untargeted) | Genome-Wide Pan-GPCR Screening |
|---|---|---|---|
| Primary Screening Objective | Identify ligands for a pre-defined, therapeutically relevant GPCR target. | Identify extracts/complex mixtures that produce a desired phenotypic change (e.g., cell death, differentiation). | Deconvolute the polypharmacology of mixtures by identifying interactions across the entire GPCRome. |
| Throughput & Scale | Very high for a single target. Scalability to many targets is linear and resource-intensive. | Typically moderate, limited by complexity of phenotypic readout. | Ultra-high-throughput once the unified cell library is established; screens all ~800 GPCRs in parallel [55]. |
| Mechanistic Insight | High for the specific target; provides immediate structure-activity relationship (SAR) data. | Low initially; target identification requires extensive downstream deconvolution (a major bottleneck). | High and immediate. Identifies specific receptor targets upon primary hit detection [53] [54]. |
| Suitability for Complex Mixtures | Low. Activity may be missed if not mediated by the single chosen target. Signal may be an aggregate of multiple weak interactions. | High. Captures integrated biological activity regardless of the number of targets involved. | Very High. Designed to dissect multi-target effects by mapping component activity to specific GPCRs [53]. |
| Key Limitation | Requires strong prior hypothesis. Misses off-target effects and synergistic polypharmacology. | Target deconvolution is slow, difficult, and often fails. Hit may be a known nuisance compound. | High initial investment to construct and validate the comprehensive cell library. Data analysis is computationally intensive [55]. |
| Representative Experimental Data Output | IC₅₀/EC₅₀ for a single receptor (e.g., "Compound X: β2-AR agonist, EC₅₀ = 150 nM"). | Phenotypic score (e.g., "Extract Y: inhibits cell migration by 70% at 10 μg/mL"). | GPCR activity signature (e.g., "Extract Y: agonist for CB2, GPR55; antagonist for 5-HT₂A, A₂A"). |
Core Experimental Protocols for GPCRome-Wide Screening
The genome-wide pan-GPCR platform relies on standardized cell-based assays. Below are detailed protocols for the two primary assay types employed.
Protocol 1: Competitive Ligand-Binding Assay (CLBA)
This primary screen identifies components that bind directly to a GPCR's orthosteric or allosteric site [53].
Detailed Methodology:
- Membrane Preparation: Harvest cells from the pan-GPCR library (e.g., a single GPCR overexpressed per cell line). Lyse cells and isolate crude membrane fractions via differential centrifugation.
- Radioligand Incubation: In a 96- or 384-well plate, incubate membrane preparations (5-20 μg protein/well) with a fixed, low concentration of a radioisotope-labeled reference ligand (e.g., [³H]-labeled antagonist) specific to the GPCR of each well.
- Test Compound Addition: Co-incubate with serially diluted traditional medicine extract fractions or purified compounds. A control well with excess unlabeled ligand defines non-specific binding.
- Separation and Detection: Terminate the reaction by rapid filtration through glass-fiber filter plates to separate bound from free radioligand. Wash filters extensively. Measure bound radioactivity using a microplate scintillation counter.
- Data Analysis: Calculate specific binding for each test concentration. Fit data to a one-site competition model to determine the inhibition constant (Ki) for each hit against each GPCR.
Protocol 2: Functional Assay Using PRESTO-Tango
This assay detects receptor activation by measuring downstream transcriptional response, identifying agonists, inverse agonists, and allosteric modulators [55].
Detailed Methodology:
- Cell Library Construction: Generate the screening library by stably integrating the PRESTO-Tango construct into a uniform host cell line (e.g., HEK293). This construct places a tetracycline transactivator (tTA) downstream of the GPCR, separated by a viral protease cleavage site. The GPCR's C-terminus is fused to a protease. A separate reporter plasmid contains a luciferase gene under a tTA-responsive promoter.
- Screening Procedure: Seed cells from the pan-GPCR library into assay plates. Treat with traditional medicine samples for a defined period (e.g., 16-24 hours).
- Mechanism of Signal Generation: Upon GPCR activation and subsequent β-arrestin recruitment, the tethered protease cleaves and releases tTA, which translocates to the nucleus and drives luciferase expression.
- Detection: Lyse cells and add luciferin substrate. Measure bioluminescence as a quantitative proxy for GPCR activation.
- Hit Triage: Compare luminescence to vehicle (basal) and reference agonist (maximal) controls. Hits are normalized and expressed as % activation. Dose-response curves are generated for confirmed hits to determine potency (EC₅₀) and efficacy (Emax).
Signaling Pathway and Screening Workflow
The following diagrams illustrate the GPCR signaling pathway and the integrated screening workflow.
Diagram 1: GPCR Signaling Cascade Initiated by a Bioactive Ligand
Diagram 2: Genome-Wide GPCR Screening and Data Analysis Workflow
Cheminformatics & Data Analysis for Complex Mixtures
The chemical complexity of traditional medicine extracts requires specialized cheminformatics tools for hit prioritization and pattern recognition.
Key Challenge: The structural complexity of natural products—characterized by more stereocenters, sp³ carbons, and unique scaffolds compared to synthetic compounds—makes standard similarity search methods less reliable [56]. Solution: Implementing biosynthetically-informed algorithms like GRAPE/GARLIC, which perform in silico retrobiosynthesis and align compounds based on their likely building blocks, has been shown to outperform conventional 2D fingerprint methods for classifying modular natural products (e.g., non-ribosomal peptides, polyketides) [56].
Table: Comparison of Cheminformatics Methods for Natural Product Analysis
| Method Type | Example Algorithms | Advantages for Natural Products | Key Limitations |
|---|---|---|---|
| 2D Circular Fingerprints | ECFP4, ECFP6 | Fast, widely used, good for broad scaffold hopping. | May miss subtle stereochemical and macrocyclic differences critical for bioactivity [56]. |
| Retrobiosynthesis & Alignment | GRAPE/GARLIC | High accuracy for modular NP classes; aligns based on biosynthetic logic. | Requires knowledge of biosynthetic rules; limited to well-characterized NP families [56]. |
| Multiparameter Optimization | Principal Component Analysis (PCA) of physicochemical properties | Visualizes extract libraries in chemical space; identifies chemical outliers. | Does not directly predict target engagement or biological activity. |
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table: Key Reagents for GPCRome-Wide Screening of Traditional Medicines
| Reagent/Solution | Function in Screening | Example & Notes |
|---|---|---|
| Genome-Wide GPCR Cell Library | Provides a uniform cellular background expressing individual human GPCRs for standardized screening. | Libraries constructed via overexpression, PRESTO-Tango, or CRISPRa/i technologies [55]. |
| Fluorescent/Radioactive Ligands | Serve as tracers for competitive binding assays to measure direct receptor occupancy. | Example: [³H]-Naloxone for opioid receptors. Fluorescent ligands (e.g., for adrenergic receptors) enable non-radioactive assays [53]. |
| β-Arrestin Recruitment Assay Kits | Enable functional high-throughput screening by detecting receptor activation via β-arrestin coupling. | PRESTO-Tango system is a genetically encoded example; commercial kits (e.g., PathHunter) are also available [55]. |
| Second Messenger Detection Kits | Quantify downstream signaling events (cAMP, Ca²⁺, IP1) to confirm functional activity and pathway bias. | HTRF (Homogeneous Time-Resolved Fluorescence) based assays are common for cAMP and IP1 detection. |
| Standardized Traditional Medicine Extract Libraries | Provide chemically characterized, reproducible starting material for screening. | Libraries should be fractionated to reduce complexity and annotated with source, extraction method, and preliminary chemistry data. |
| Integrated Data Analysis Software | Manages, analyzes, and visualizes high-dimensional screening data from millions of data points. | Platforms like Genedata Screener streamline data processing, hit calling, and dose-response analysis [57]. |
Discussion: Advantages, Limitations & Future Directions
The genome-wide pan-GPCR platform addresses critical gaps in both pure target-based and phenotypic approaches. Its primary advantage is the ability to simultaneously deconvolute mechanism and polypharmacology. For example, the anti-inflammatory terpenoid celastrol was identified as a selective CB2 agonist through targeted screening, but a pan-GPCR screen could reveal its full receptor interaction profile, explaining its broader effects [53]. This platform is particularly aligned with the multi-component, multi-target paradigm of traditional medicine [53].
Current Limitations include the significant upfront investment required to build and validate the unified cell library, potential artifacts from GPCR overexpression, and the challenge of detecting very weak but therapeutically relevant interactions that might be significant in a polypharmaceutical context.
Future Directions point toward even more integrative systems:
- Advanced Cell Models: Implementing screening in primary cells or iPSC-derived cells expressing endogenous levels of GPCRs for more physiologically relevant pharmacology.
- Multi-Omic Integration: Correlating GPCR activity signatures with transcriptomic or proteomic response data from phenotypic assays on the same extracts.
- AI-Powered Prediction: Using primary screening data to train models that predict the GPCRome activity of new extracts based on their chemical fingerprints, dramatically accelerating the discovery process.
This platform does not render phenotypic screening obsolete but rather creates a powerful synergistic loop. Phenotypic assays can identify the most therapeutically promising extracts, which are then rapidly mechanistically deconvoluted via pan-GPCR screening. This combined strategy effectively bridges the gap between the holistic observations of traditional medicine and the molecular precision of modern drug discovery.
Navigating Pitfalls: Overcoming Key Challenges in NP-Centric Assay Workflows
Addressing Data Heterogeneity and Sparsity in Phenotypic Profiling
The choice between target-based and phenotypic screening strategies defines a fundamental dichotomy in modern drug discovery [33]. Target-based approaches, which screen compounds against a specific purified protein or known molecular target, offer clear mechanisms and are generally less costly and simpler to implement [33]. In contrast, phenotypic drug discovery (PDD) measures complex changes in cells, tissues, or whole organisms without prior bias toward a specific target, making it particularly powerful for identifying first-in-class medicines with novel mechanisms of action [1]. This unbiased nature is especially valuable in natural products (NP) research, where the complex, evolved chemistry of NPs often interacts with biological systems in multifaceted and unpredictable ways [4].
However, the power of phenotypic profiling is challenged by two intrinsic data properties: heterogeneity and sparsity. Heterogeneity refers to the biological variation between individual cells within a treated population, which, if ignored, can obscure true phenotypic signatures [58] [59]. Sparsity arises when the vast landscape of possible compound-induced phenotypes is sampled only thinly by experimental data, making robust predictions difficult [60]. Effectively addressing these challenges is critical for accurately interpreting phenotypic data, predicting mechanisms of action (MoA), and prioritizing NPs for development. This guide compares computational and experimental strategies designed to overcome these limitations, placing them within the practical context of advancing NP research from hit identification to lead optimization.
Comparison of Profiling and Data Fusion Strategies
The following tables compare the performance, applications, and requirements of key methodologies that address heterogeneity and sparsity in phenotypic screening.
Table 1: Comparison of Strategies for Addressing Single-Cell Heterogeneity in Profiling
| Method | Core Approach | Key Performance Finding | Advantages for NP Research | Limitations |
|---|---|---|---|---|
| Average Profiling (Baseline) | Uses mean/median of single-cell features [58]. | Standard approach; loses heterogeneity information [58]. | Simple, computationally efficient, established. | Subpopulations with opposing effects may cancel out; misses population variance [58]. |
| Dispersion-Enhanced Profiling | Concatenates median with median absolute deviation (MAD) for each feature [58]. | Provides minor improvement over median alone [58]. | Captures univariate variance; easy to implement. | Poor signal-to-noise if dispersion is noisy; does not capture covariance between features [58]. |
| Data Fusion via SNF | Fuses similarity matrices from median, MAD, and sparse random projections of covariances [58]. | ~20% better performance in predicting compound MoA and gene pathways vs. alternatives [58]. | Captures complex, multivariate heterogeneity; robust. | More computationally complex; requires sufficient replicate number for fusion [58]. |
| Cytological Profiling (CP) with Multiple Markers | Uses 10-20 cellular features from 14+ fluorescent markers profiling major organelles/pathways [4]. | Enables MoA prediction and SAR analysis on single-cell level [4] [22]. | Provides holistic, interpretable view of NP effects; guides targeted isolation [22]. | Lower throughput than standard Cell Painting; requires extensive marker panel. |
Table 2: Comparison of Modalities for Predicting Compound Bioactivity
| Data Modality | Description | Assay Prediction Performance (AUROC >0.9) | Key Contribution | Practical Considerations |
|---|---|---|---|---|
| Chemical Structure (CS) | Molecular representation via graph convolutional nets [15]. | Predicts 16/270 assays (5.9%) alone [15]. | Always available; no wet-lab work required. | May lack biological context; struggles with activity cliffs [15]. |
| Morphological Profile (MO) | Image-based profiles (e.g., Cell Painting) [15]. | Predicts 28/270 assays (10.4%) alone—the most of any single modality [15]. | Captures broad, unbiased phenotypic response. | Requires wet-lab experiment; cost and scale considerations. |
| Gene Expression (GE) | Transcriptional profiles (e.g., L1000 assay) [15]. | Predicts 19/270 assays (7.0%) alone [15]. | Direct readout of pathway activity. | Less scalable than imaging; more expensive [15]. |
| Late Fusion (CS+MO) | Combines prediction probabilities from CS and MO models [15]. | Predicts 31/270 assays (11.5%) [15]. | 2-3x higher success rate than single modalities; leverages complementarity [15]. | Optimal fusion strategies are still an area of research [15]. |
| All Modalities Combined | Retrospective selection of best single or fused predictor per assay [15]. | Could potentially predict 21% of assays with high accuracy [15]. | Maximizes coverage by leveraging unique strengths of each data type. | Simulates an ideal, informed selection scenario. |
Core Experimental Protocols for High-Resolution Phenotypic Profiling
Data Fusion Protocol for Capturing Single-Cell Heterogeneity
This protocol, based on the method from Nature Communications (2019), improves MoA prediction by fusing information from multiple representations of single-cell data [58].
- Data Acquisition & Feature Extraction: Perform a high-throughput image-based assay (e.g., Cell Painting). Extract hundreds of morphological features (e.g., texture, shape, intensity) from every single cell.
- Create Population Profiles: For each treatment well (compound/perturbation), create three separate profile vectors:
- Median Profile: The median value for each feature across all cells.
- Dispersion (MAD) Profile: The median absolute deviation for each feature.
- Covariance Profile: Calculate the covariance matrix between all feature pairs. Use sparse random projections to reduce this high-dimensional matrix to a manageable, lower-dimensional representation [58].
- Calculate Similarity Matrices: For each profile type (Median, MAD, Covariance), calculate a separate similarity matrix (e.g., using correlation) between all treatment pairs.
- Similarity Network Fusion (SNF): Fuse the three similarity matrices using the SNF algorithm. This method iteratively updates each similarity matrix based on information from the others, creating a single, robust fused similarity network that reflects shared information across data types [58].
- Validation & MoA Prediction: Validate the fused network by measuring the enrichment of known MoA categories among the top-most similar treatment pairs. Compare the enrichment score against scores from networks based on single profile types [58].
Cytological Profiling for Natural Products Mechanism Prediction
This protocol, adapted from Scientific Reports (2017), is designed for the in-depth phenotypic characterization of natural product libraries [4].
- Staining & Imaging: Treat cells with NPs or reference compounds. Fix and stain with a comprehensive panel of 14+ fluorescent markers targeting key organelles (nucleus, cytoskeleton, mitochondria, lysosomes, Golgi) and pathway reporters (e.g., NF-κB) [4].
- Image Analysis & Core Feature Selection: Acquire high-resolution images. Segment individual cells and extract ~150 morphological and intensity features. Condense these into a set of 20 core features for interpretability (e.g., cell count, nuclear size, lysosome number, tubulin intensity) [4].
- Profile Database Building: Create a reference database of cytological profiles (CPs) for hundreds of compounds with known, annotated MoAs [4].
- Similarity Matching & Clustering: Calculate the similarity (e.g., correlation) between the CP of a novel NP and all profiles in the reference database. Use hierarchical clustering to group NPs with reference compounds sharing similar CPs [4].
- MoA Prediction & Validation: Assign a putative MoA to the NP based on the known MoAs of its nearest reference compound neighbors. Perform orthogonal, target-specific assays (e.g., a kinase activity assay or a DNA damage marker like γH2AX) to validate the predicted mechanism [4] [22].
Visualizing Workflows and Logical Relationships
Data Fusion Workflow for Heterogeneity-Aware Profiling (79 characters)
Assay Prediction by Complementary Data Modalities (71 characters)
The Scientist's Toolkit: Essential Reagents & Materials
Table 3: Key Research Reagent Solutions for Phenotypic Profiling
| Reagent/Material | Function in Phenotypic Profiling | Example Application |
|---|---|---|
| Cell Painting Assay Kit | A standardized, multiplexed fluorescent staining protocol to label 5-8 cellular components (nucleus, nucleoli, ER, mitochondria, Golgi, cytoskeleton, plasma membrane). Provides the foundational data for image-based morphological profiling [58] [15]. | Generating high-dimensional morphological profiles for thousands of compounds to predict Mechanism of Action (MoA) [15]. |
| L1000 Assay | A high-throughput, low-cost gene expression profiling method that measures ~1,000 landmark transcripts. Provides complementary transcriptomic phenotypic data [15]. | Generating gene expression profiles to combine with morphological data for improved bioactivity prediction [15]. |
| Broad Multiplex Marker Panel | A custom panel of 10-14 fluorescent dyes and antibodies targeting specific organelles (lysosomes, mitochondria) and pathway reporters (NF-κB, DNA damage). Enables deep cytological profiling [4]. | In-depth characterization of natural product effects, enabling toxicity assessment and detailed MoA prediction [4] [22]. |
| Reference Compound Library with Annotated MoA | A collection of 480-720 well-characterized bioactive compounds with known, diverse mechanisms of action. Serves as a training set for similarity-based MoA prediction [4] [22]. | Clustering and comparing novel natural product profiles to predict their putative biological targets [4]. |
| Validated Control Compounds | Compounds with strong, consistent phenotypic signatures (e.g., nocodazole for microtubule disruption, Brefeldin A for Golgi disruption). Essential for assay quality control (Z'-factor calculation) and batch normalization [59]. | Ensuring technical reproducibility and robustness of the phenotypic screening platform across different experimental runs [59]. |
The drug discovery landscape for natural products (NPs) is defined by a fundamental tension between phenotypic and target-based screening paradigms. Phenotypic screening, which measures a compound's effect in cells or tissues without pre-specified molecular targets, has historically been the source of a majority of first-in-class medicines [1]. This empirical approach is particularly advantageous for NPs, whose intricate scaffolds often engage in polypharmacology—modulating multiple targets simultaneously—which can be crucial for treating complex diseases like neurodegeneration and inflammation [61]. Conversely, the target-based approach, dominant in late-20th-century drug design, focuses on modulating a single, well-defined protein with high specificity [61]. While this "one-target-one-disease" philosophy yields follower drugs efficiently, its hyper-selectivity may contribute to high Phase II clinical failure rates due to lack of efficacy, as drugs fail to meaningfully engage the systems biology of disease [61].
The inherent scaffold diversity and multi-target effects of NPs create both an opportunity and a challenge for assay design. Optimizing assays to capture this complexity requires strategies that either embrace polypharmacology through phenotypic systems or deconvolute it through advanced target identification technologies. This guide compares modern approaches—from library creation and screening to hit optimization—within this core thesis, providing researchers with a framework to select and implement the most effective strategies for their NP-based discovery campaigns.
Comparison Guide 1: Foundational Screening Approaches
The choice between phenotypic and target-based screening sets the trajectory for an entire NP discovery project. The table below summarizes their core operational and strategic differences.
Table 1: Comparison of Phenotypic vs. Target-Based Screening Approaches for Natural Products
| Aspect | Phenotypic Screening | Target-Based Screening |
|---|---|---|
| Primary Objective | Identify compounds that induce a relevant biological change in cells, tissues, or whole organisms. | Identify compounds that modulate the activity of a predefined, purified protein target. |
| Advantages | - Unbiased; can discover novel mechanisms and targets [1].- Accounts for cell permeability, metabolism, and polypharmacology upfront [33].- Historically more successful for first-in-class drugs [1]. | - High throughput and generally less costly [33].- Direct mechanism of action (MOA).- Easier to optimize structure-activity relationships (SAR). |
| Disadvantages | - Often slower, more expensive, and lower throughput [33].- Target identification (deconvolution) is a major bottleneck [62].- Can be susceptible to assay interference. | - Requires a deep, validated understanding of disease biology.- Hits may lack cell activity due to poor permeability or off-target effects being necessary for efficacy.- May miss superior polypharmacology profiles. |
| Best Suited for NPs When... | The disease biology is complex or poorly understood, or when the therapeutic value of NP polypharmacology is being explicitly sought. | A specific, druggable target within a pathway is well-validated, and the goal is to find a potent, selective modulator. |
| Key Assay Design Considerations | Must use disease-relevant cell/ tissue models; requires robust, quantifiable phenotypic readouts (e.g., imaging, cell death, cytokine secretion). | Assay must be configured for potential NP interference (e.g., color, fluorescence, aggregation); purity of target is critical. |
Supporting Experimental Context: The success of the phenotypic approach is underscored by analysis showing it as the more productive strategy for discovering first-in-class small molecule medicines [1]. However, the rise of label-free target identification methods (detailed in Section 4) is directly addressing the primary bottleneck of phenotypic screening—target deconvolution—by enabling the unbiased discovery of a compound's protein targets without requiring chemical modification [62]. Modern strategies increasingly advocate for a hybridized approach, where target-based libraries are phenotypically filtered for cell activity, or phenotypic hits are rapidly deconvoluted to guide medicinal chemistry [33].
Comparison Guide 2: Library Design & Profiling Strategies
The quality of the screened library is paramount. For NPs, this involves unique considerations for sourcing, preparation, and computational profiling to navigate their vast chemical space.
Table 2: Strategies for Natural Product Library Creation and Profiling
| Strategy | Description & Protocol Highlights | Key Performance Data & Application |
|---|---|---|
| Prefractionated Library Creation (e.g., NCI Program) | Protocol: Source organisms are collected under access and benefit-sharing agreements. Biomass is extracted (e.g., with accelerated solvent extraction), then prefractionated using semi-preparative HPLC. Fractions are plated into 384-well plates [63].Goal: To move beyond crude extracts, reducing interference and concentrating minor metabolites. | The U.S. NCI's program is generating a library of 1 million partially purified natural product fractions [63]. Prefractionation improves screening performance by concentrating actives and sequestering nuisance compounds, leading to higher-confidence hit rates [63]. |
| In-Situ Build-Up Library (Optimization Strategy) | Protocol: NP scaffolds are divided into a core fragment (with key pharmacophore) and accessory fragments. A clean, high-yield ligation reaction (e.g., hydrazone formation) is performed directly in assay plates to generate an analog library, which is screened without purification [64].Goal: To enable rapid, comprehensive SAR exploration of complex NPs without lengthy individual syntheses. | Applied to MraY inhibitors, a library of 686 analogues was created from 7 cores and 98 fragments [64]. The method identified potent, broad-spectrum antibacterial analogues effective in a mouse infection model, demonstrating streamlined hit-to-lead progression [64]. |
| Computational Target Prediction (e.g., CTAPred) | Protocol: Uses a similarity-based approach. A query NP's fingerprint is compared to a curated reference database of compounds with known targets (e.g., from ChEMBL, NPASS). Targets of the most similar reference compounds are predicted for the query [65].Goal: To prioritize potential macromolecular targets for an NP before experimental validation. | The CTAPred tool focuses on proteins relevant to NPs. Evaluation shows that considering only the top 3 most similar reference compounds optimizes prediction accuracy, balancing the retrieval of true targets against false positives [65]. |
| Generative AI for Scaffold Optimization | Protocol: A generative model (e.g., Variational Autoencoder) is trained on known active structures. It is refined through active learning cycles using physics-based oracles (e.g., docking scores) and chemical filters to propose novel, optimized analogs [66].Goal: To explore novel chemical space around an NP scaffold for improved properties. | For CDK2, this workflow generated novel scaffolds distinct from known inhibitors. Of 9 synthesized molecules, 8 showed in vitro activity, with one reaching nanomolar potency [66]. |
Experimental Protocol Deep Dive: In-Situ Build-Up Library for MraY Inhibitors [64]
- Design & Synthesis: Core aldehyde fragments, containing the essential uridine moiety for MraY binding, were prepared via total synthesis. A diverse set of 98 hydrazine accessory fragments was synthesized or sourced.
- Library Assembly: In a 96-well plate, 10 mM DMSO solutions of a core aldehyde and a hydrazine fragment were mixed in a 1:1 ratio (total volume 31 µL). No catalysts or additives were used.
- Reaction & Preparation: The plate was incubated at room temperature for 30 minutes to allow hydrazone formation. DMSO was removed via centrifugal vacuum concentration, and the residue was re-dissolved in 30 µL DMSO to create a ~5 mM stock solution of the new analog.
- Direct Screening: This library solution was used directly in a biochemical MraY inhibition assay and a cell-based antibacterial susceptibility assay, assuming 100% conversion. LC-MS analysis confirmed yields were typically >80%.
- Hit Identification & Validation: Analysts with potent dual activity (enzyme inhibition and bacterial killing) were identified, synthesized at scale, purified, and validated in vitro and in vivo.
Comparison Guide 3: Target Identification & Deconvolution Technologies
Following a phenotypic screen, identifying the molecular target(s) of an NP hit is critical. Label-free methods have become essential as they do not require difficult chemical modification of the often complex, scarce NP.
Table 3: Label-Free Target Identification Methods for NPs from Phenotypic Screens
| Method | Core Principle | Experimental Workflow Summary | Advantages for NP Research |
|---|---|---|---|
| Cellular Thermal Shift Assay (CETSA) | A ligand binding stabilizes its target protein against heat-induced denaturation. | Cells or lysates are treated with compound or vehicle, heated to a range of temperatures, and the soluble (native) protein is quantified (often by immuno-blot) [62]. | Requires no compound modification. Works in intact cells, providing physiological relevance. Best for validating a suspected target. |
| Thermal Proteome Profiling (TPP) | A proteome-wide extension of CETSA using mass spectrometry. | Compound- and vehicle-treated samples are heated, followed by proteomic analysis of soluble fractions. Proteins showing a thermal stability shift are potential targets [62]. | Fully unbiased, global mapping of target engagement in a single experiment. Identifies both primary targets and off-targets. |
| Drug Affinity Responsive Target Stability (DARTS) | Ligand binding protects a target protein from proteolytic degradation. | Cell lysates are incubated with compound or vehicle, then subjected to limited proteolysis. Protease-resistant proteins are identified via gel electrophoresis or mass spectrometry [62]. | No compound modification needed. Technically simpler and lower cost than TPP. Can use native lysates. |
| Stability of Proteins from Rates of Oxidation (SPROX) | Ligand binding alters the thermodynamic stability of a protein, changing the rate of methionine oxidation under chemical denaturation. | Lysates +/- compound are treated with a denaturant gradient, followed by oxidation of exposed methionines and quantitative proteomic analysis [62]. | Can detect weaker binding events and conformational changes. |
Visualization: Workflow for Integrated Phenotypic Screening & Target Deconvolution
Diagram Title: Integrated workflow for phenotypic NP screening and target deconvolution.
Table 4: Key Research Reagent Solutions for NP Assay Optimization
| Category | Item / Resource | Function & Relevance to NP Research |
|---|---|---|
| Physical Libraries | NCI Natural Products Repository [63] | One of the world's largest, most diverse collections of natural product extracts and fractions, available for screening. |
| Pre-plated Diversity Sets (e.g., from commercial vendors) [33] | Curated, drug-like compound libraries in assay-ready plates, useful for hybrid screening campaigns. | |
| Computational Tools | CTAPred [65] | Open-source, command-line tool for predicting protein targets of NPs based on chemical similarity. |
| Generative AI Models (e.g., VAE-AL workflow) [66] | AI-driven design of novel NP analogs with optimized target affinity and synthetic accessibility. | |
| Assay Reagents | CETSA / TPP Kits & Reagents | Enable label-free target engagement studies in cells or lysates without modifying the NP. |
| Cell-Based Phenotypic Assay Kits (e.g., viability, apoptosis, reporter gene) | Enable functional screening in disease-relevant cellular models. | |
| Chemical Biology | Fragment Libraries for Build-Up Synthesis [64] | Collections of accessory fragments (e.g., acyl hydrazides) for rapid analog generation via clean ligation chemistry. |
Optimizing assay design for the complexity of natural products requires a departure from rigid, single-target thinking. The most promising modern frameworks are integrative, leveraging the unbiased discovery power of phenotypic screening to identify compelling biological activity, followed by advanced label-free deconvolution methods to map polypharmacology. Concurrently, innovations in computational target prediction and generative AI are providing unprecedented guides for navigating NP chemical space, while build-up library strategies dramatically accelerate the SAR of complex scaffolds.
The future of NP-based drug discovery lies in strategically combining these tools. Initiating with a high-quality, prefractionated library in a phenotypic assay maximizes the chance of finding novel biology. Employing TPP or CETSA early for hit deconvolution rapidly focuses the project on tractable mechanisms. Finally, using AI-guided design and in-situ build-up libraries can efficiently optimize validated NP hits into drug leads. This synergistic approach, which respects and harnesses the inherent scaffold diversity and multi-target effects of NPs, is best positioned to deliver the next generation of first-in-class medicines.
The discovery of first-in-class medicines has historically been more successful through phenotypic screening—an unbiased approach that identifies compounds based on a desired biological effect in cells, tissues, or whole organisms—than through target-based methods [1]. This empirical strategy is particularly powerful for natural products (NPs), whose complex chemical scaffolds and evolutionary optimization for bioactivity offer unique opportunities to modulate novel biological pathways [6]. However, a significant bottleneck follows phenotypic discovery: target deconvolution, the process of identifying the precise molecular target(s) responsible for the observed phenotype [67].
This challenge is magnified for many NPs, which are often difficult to label due to complex chemical structures, limited availability from natural sources, or tight structure-activity relationships (SAR) where even minor modification abolishes activity [62]. Furthermore, NPs may exhibit low-affinity or transient interactions with their protein targets, complicating capture and identification [67]. This guide objectively compares modern target deconvolution strategies, focusing on their applicability to NPs, and provides the experimental data and protocols necessary to implement them. The discussion is framed within the enduring strategic tension between phenotypic and target-based drug discovery, assessing how advanced deconvolution tools are reshaping this paradigm by bridging empirical observation with mechanistic understanding [33].
Comparative Analysis of Target Deconvolution Strategies
The following table provides a high-level comparison of the major target deconvolution strategy classes, detailing their core principle, key advantages for NP research, and primary limitations.
Table 1: Core Target Deconvolution Strategy Classes for Natural Products
| Strategy Class | Core Principle | Key Advantage for NPs | Major Limitation(s) |
|---|---|---|---|
| Affinity-Based Chemoproteomics [62] [67] | Immobilized NP derivative ("bait") pulls down binding proteins from lysate for MS identification. | Gold standard for direct binding confirmation; can provide affinity data (Kd). | Requires chemical modification (labeling) of NP, which is often synthetically challenging and can alter bioactivity [62]. |
| Label-Free Stability Profiling [62] | Measures ligand-induced changes in protein thermal or chemical stability across the proteome. | No chemical modification required; works with native, low-availability NPs; detects both high and low-affinity binders. | Less direct than pull-down; may miss membrane proteins; complex data analysis [62]. |
| Genome-Wide CRISPR Screening [68] | Uses pooled gene knockout libraries to identify genes whose loss abolishes compound-induced phenotype. | Completely label-free; identifies targets and pathway dependencies; highly scalable. | Limited to genetically tractable cell models; identifies genetic dependencies, not always direct binders. |
| Photoaffinity Labeling (PAL) [67] | A photoreactive NP derivative forms a covalent bond with its target upon UV irradiation for capture. | Captures transient or weak interactions; excellent for membrane protein targets. | Requires design and synthesis of a bifunctional probe, risking altered pharmacology. |
Performance Comparison: Quantitative Experimental Data
The selection of a deconvolution strategy is guided by the specific NP and biological context. The following tables summarize performance metrics from key studies.
Table 2: Performance of Label-Free Stability Profiling Methods [62]
| Method | Readout | Typical Workflow Duration | Key Application in NP Studies | Notable Success |
|---|---|---|---|---|
| DARTS (Drug Affinity Responsive Target Stability) | Differential resistance to proteolysis (gel-based). | 2-3 days | Initial, low-cost target validation. | Identified direct target of laurifolioside [62]. |
| CETSA (Cellular Thermal Shift Assay) | Protein solubility after heating (antibody-based). | 1-2 days | Validation of target engagement in intact cells. | Validated in-cell target engagement for multiple drug candidates [62]. |
| TPP (Thermal Proteome Profiling) | Proteome-wide solubility via quantitative MS. | 1-2 weeks | Unbiased identification of primary targets and off-targets. | Mapped targets and downstream pathways for anticancer drugs [62]. |
Table 3: Performance of Genomic and High-Throughput Methods
| Method | Reported Success Rate | Scale (Library Size) | Key Strength for NPs | Reference Study |
|---|---|---|---|---|
| Pooled CRISPR/Cas9 Screening | 97% (38/39 antibodies deconvoluted) [68] | Genome-wide (e.g., ~77k sgRNAs) [68] | Label-free; identifies functional genetic dependencies beyond direct binders. | Accelerated target deconvolution for therapeutic antibodies [68]. |
| AI-Expanded Virtual NP Libraries | Generated 67 million NP-like structures (165x expansion) [69] | 67+ million compounds [69] | Enables in silico target prediction and screening for novel scaffolds. | Creation of a vast database for in silico discovery [69]. |
Experimental Protocols for Key Strategies
Protocol: Thermal Proteome Profiling (TPP) for Unbiased Target ID
TPP is a powerful, label-free method to identify drug-target interactions by monitoring ligand-induced shifts in protein thermal stability across the proteome [62].
- Sample Preparation: Treat separate aliquots of live cells or cell lysate with the NP of interest or a vehicle control (e.g., DMSO).
- Heat Denaturation: Divide each aliquot into 10-12 fractions. Heat each fraction at a different temperature (e.g., 37°C to 67°C) for 3 minutes.
- Soluble Protein Harvest: Centrifuge to pellet denatured/aggregated proteins. Collect the soluble supernatant fraction containing heat-stable proteins.
- Proteomic Digestion & Labeling: Digest soluble proteins with trypsin. Use tandem mass tag (TMT) isobaric labeling to multiplex all temperature points from treated and untreated samples.
- LC-MS/MS Analysis: Pool labeled samples and analyze via liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS).
- Data Analysis: For each protein, model the melting curve (amount soluble vs. temperature). A significant rightward shift in the melting curve (increased Tm) in the drug-treated sample indicates a stabilizing interaction between the NP and that protein [62].
Protocol: Pooled CRISPR/Cas9 Screening for Functional Target Deconvolution
This scalable, genetic approach identifies genes essential for a compound's phenotypic effect [68].
- Library Transduction: Transduce a population of report cells with a lentiviral genome-wide CRISPR knockout library (e.g., Brunello with ~77k sgRNAs) [68].
- Selection: Treat the pooled cell population with the bioactive NP at a relevant concentration. Include a DMSO-treated control pool.
- Phenotypic Sorting/Selection: After several population doublings, apply the phenotypic readout (e.g., cell death, fluorescence reporter). Use FACS to isolate the surviving or phenotype-negative cell population.
- Genomic DNA Extraction & Sequencing: Recover genomic DNA from selected and control populations. Amplify and sequence the integrated sgRNA regions via next-generation sequencing (NGS).
- Bioinformatic Analysis: Use algorithms (e.g., MAGeCK) to compare sgRNA abundance between NP-treated and control populations. Significantly depleted sgRNAs in the treated population pinpoint genes whose knockout confers resistance, indicating they are involved in the compound's mechanism of action [68].
Protocol: Affinity-Based Pull-Down with Chemical Probe
The classical approach requires a modified, bioactive derivative of the NP [67].
- Probe Synthesis: Derivative the NP with a functional handle (e.g., alkyne, biotin) via synthetic chemistry. Critical: Validate that the probe retains biological activity.
- Immobilization: Couple the probe to solid support beads (e.g., streptavidin beads for a biotinylated probe).
- Incubation with Lysate: Incubate immobilized probe beads with pre-cleared cell or tissue lysate. Use beads with an inactive analog (e.g., stereoisomer, scrambled version) as a negative control.
- Wash & Elution: Wash beads stringently to remove non-specific binders. Elute bound proteins with SDS buffer or competitive ligand.
- Protein Identification: Separate eluted proteins by SDS-PAGE, digest gel bands, and identify proteins by LC-MS/MS. Compare results from active probe vs. control to identify specific binding partners.
Visualizing Workflows and Strategic Integration
Target Deconvolution Strategy Selection Workflow
Resolving the Bottleneck: From Phenotypic Hit to Optimized Drug
Table 4: Key Research Reagent Solutions for Target Deconvolution
| Reagent / Resource | Function in Deconvolution | Example & Utility for NPs |
|---|---|---|
| Functionalized NP Probe | Serves as "bait" for affinity purification; requires retained bioactivity. | A biotin- or alkyne-conjugated derivative; critical for affinity pull-down and PAL [67]. |
| Genome-Wide CRISPR Library | Enables pooled, loss-of-function screening to identify genetic dependencies. | Brunello or GeCKO library; used in cells to find genes required for NP activity [68]. |
| Tandem Mass Tag (TMT) Reagents | Enables multiplexed, quantitative proteomics in TPP and pull-down/AP-MS. | 10- or 11-plex TMT kits; allows simultaneous analysis of multiple temp/dose points [62]. |
| Thermal Shift Dyes | Report protein unfolding in thermal stability assays (e.g., CETSA). | Dyes like SYPRO Orange; used in plate-based formats for mid-throughput target validation [62]. |
| Open-Access NP Databases | Provides structural and spectral data for dereplication and in silico studies. | Natural Products Atlas (microbial NPs) [70]; NP Database (67M+) for AI-driven exploration [69]. |
Discussion: Integrating Strategies within the Phenotypic-Targeted Spectrum
The dichotomy between phenotypic and target-based discovery is increasingly a false one [33]. Modern deconvolution tools are creating a powerful hybrid paradigm. The journey begins with a phenotypic screen—highly effective for identifying first-in-class NP therapeutics [1]. The subsequent bottleneck is now addressed by a strategic choice: label-free methods (TPP, CETSA) for delicate or scarce NPs, genetic screens (CRISPR) for functional pathway mapping, or advanced chemoproteomics (PAL, ABPP) for challenging target classes like membrane proteins [62] [68] [67].
Once the target is identified, the workflow seamlessly transitions to a target-based optimization phase, leveraging structural biology and medicinal chemistry—a realm where NPs have traditionally been challenging. Here, AI and computational databases play a transformative role. The generation of over 67 million NP-like virtual compounds demonstrates how machine learning can expand the explorable chemical space around an NP scaffold by orders of magnitude, guiding semi-synthesis or total synthesis toward improved properties [69].
The target deconvolution bottleneck for difficult-to-label or low-affinity natural products is being decisively overcome. No single strategy is universally superior; the power lies in a toolkit approach. Label-free stability profiling offers a path for native NPs, CRISPR screening uncovers functional networks, and advanced chemoproteomics captures elusive interactions. These methods, supported by open-access databases and AI, are not merely solving a technical problem. They are fundamentally bridging the phenotypic and target-based worlds. They transform an NP from a phenomenological curiosity into a mechanistic starting point, enabling the rational optimization of nature's intricate compounds into the next generation of high-precision medicines. The future of NP-based drug discovery lies in this integrated cycle: from unbiased phenotypic observation, through sophisticated target identification, to informed molecular design.
Quantitative Comparison of Preclinical Model Systems
The following tables summarize the key performance characteristics of different preclinical models, highlighting the evolution in complexity and translational value.
Table 1: Comparison of Core Characteristics of Preclinical Models
| Feature | 2D Cell Lines | Multicellular Tumor Spheroids (MCTS) | Patient-Derived Organoids (PDOs) | Patient-Derived Xenografts (PDXs) |
|---|---|---|---|---|
| Architectural & Cellular Complexity | Simple monolayer; homogeneous cell population. | 3D structure; some cell-cell contact; can be multi-cellular. | 3D structure with tissue-like architecture; preserves stem cell hierarchy and differentiated cell types [71] [72]. | In vivo architecture within a mouse host; includes human stroma initially replaced by mouse cells. |
| Genetic & Pathological Fidelity | Often highly mutated from long-term culture; lacks original tumor heterogeneity. | Depends on source cells; may not fully capture heterogeneity. | Preserves genetic landscape, mutations, and heterogeneity of parental tumor [71] [73]. | Generally maintains genetic profile and histology of original tumor. |
| Tumor Microenvironment (TME) | Absent. | Limited, can model nutrient/oxygen gradients. | Can be co-cultured with immune/stromal cells; supports reconstituted or innate immune microenvironments [73]. | Complete but murine TME (vessels, immune cells, stroma). |
| Throughput & Scalability | Very high; suitable for large-scale screening. | High; amenable to medium/high-throughput formats. | Moderate to high; living biobanks enable screening [71]. | Very low; expensive, time-consuming (months), low engraftment rates. |
| Typical Timeline for Assay | Days to 1 week. | 1-2 weeks. | 2-4 weeks (from biopsy to result). | 3-8 months. |
| Cost | Low. | Low to moderate. | Moderate. | Very high. |
| Primary Translational Application | Initial target identification, high-throughput hit discovery. | Study of basic tumor biology, drug penetration. | Personalized drug response prediction, biomarker discovery, precision medicine [71] [72]. | Preclinical in vivo efficacy studies, co-clinical trials. |
Table 2: Representative Global Patient-Derived Organoid (PDO) Biobanks for Solid Tumors [71] This table excerpts data from established living PDO biobanks, demonstrating their scale and application in translational research.
| System | Organ | Number of Samples (Tumor / Healthy) | Country | Primary Translational Application Demonstrated |
|---|---|---|---|---|
| Digestive | Colorectal | 151 / 0 | China | Drug response prediction [71] |
| Digestive | Colorectal | 77 / 31 | The Netherlands | High-throughput screening (in vitro/in vivo) [71] |
| Digestive | Stomach | 46 / 17 | China | High-throughput screening, drug response prediction [71] |
| Digestive | Pancreas | 31 / 0 | Switzerland | Disease modeling, high-throughput screening [71] |
| Reproductive | Mammary Gland | 168 / 0 | The Netherlands | Drug response prediction [71] |
| Reproductive | Ovaries | 76 / 0 | United Kingdom | Disease modeling, drug response prediction [71] |
| Urinary | Kidney | 54 / 47 | The Netherlands | Disease modeling, drug response prediction [71] |
Experimental Protocols for Key Applications
Protocol 1: Establishing a Patient-Derived Tumor Organoid (PDTO) Biobank [71] [73]
- Sample Acquisition & Processing: Obtain fresh tumor tissue from surgical resection or biopsy under ethical approval. Mechanically mince and enzymatically digest (e.g., using collagenase) the tissue into small cell clusters or single cells.
- Matrix Embedding: Resuspend the cell mixture in a basement membrane extract (e.g., Matrigel) or a defined synthetic hydrogel.
- Culture Initiation: Plate the matrix-cell suspension as droplets in a pre-warmed culture plate and allow polymerization. Overlay with a specialized, tissue-specific medium.
- Medium Formulation: The medium is critical and typically contains:
- A base medium (e.g., Advanced DMEM/F12).
- Essential growth factors (e.g., EGF, Noggin, R-spondin, Wnt3A for gastrointestinal tissues) [73].
- Tissue-specific additives (e.g., FGF10 for lung, HGF for liver) [73].
- A cAMP agonist (e.g., forskolin) and often a TGF-β pathway inhibitor (e.g., A83-01).
- B27 supplement and antibiotics/antimycotics.
- Expansion & Passaging: Culture at 37°C with 5% CO₂. Organoids are typically passaged every 1-2 weeks by mechanically breaking and re-embedding fragments in fresh matrix.
- Validation: PDO lines are validated via histology (H&E, immunohistochemistry) against the parent tumor, whole-genome/exome sequencing to confirm mutational retention, and RNA sequencing to assess transcriptional fidelity [71].
Protocol 2: Phenotypic Drug Sensitivity Screening in PDOs
- Organoid Preparation: Harvest and dissociate mature PDOs into single cells or small, uniform clusters.
- Assay Plating: Seed a defined number of cells/clusters into a 384-well plate pre-coated with matrix. Allow for re-aggregation and recovery for 24-48 hours.
- Compound Treatment: Treat organoids with a natural product library or single compounds across a multi-log dose range. Include DMSO vehicle controls and reference cytotoxic controls.
- Co-culture Integration (for Immunotherapy Assessment): For immune-oncology applications, add autologous or allogeneic peripheral blood mononuclear cells (PBMCs) or isolated immune cell subsets to the wells in a defined ratio to the tumor organoids [73].
- Incubation & Endpoint Analysis: Incubate for 5-7 days. Assess viability using 3D-optimized ATP-based luminescence (CellTiter-Glo 3D) or imaging-based assays (e.g., calcein AM/propidium iodide staining). For co-cultures, measure tumor cell death and immune cell activation markers via flow cytometry.
- Data Analysis: Generate dose-response curves, calculate IC₅₀/EC₅₀ values, and correlate sensitivity with genomic and transcriptomic data from the PDOs to identify predictive biomarkers.
Visualizing Workflows and Biological Pathways
Progression from 2D to 3D Patient-Derived Models
Key Signaling Pathways Modulated in Organoid Culture Media
The Scientist's Toolkit: Essential Reagents for Patient-Derived Organoid Culture
Table 3: Key Research Reagent Solutions for Organoid Models
| Reagent Category | Specific Example(s) | Primary Function in Organoid Culture |
|---|---|---|
| Extracellular Matrix (ECM) | Matrigel (Corning), Cultrex BME, Synthetic PEG-based hydrogels | Provides a 3D scaffold that mimics the basement membrane; supports cell polarization, organization, and survival [73]. |
| Base Medium | Advanced DMEM/F12, IntestiCult Organoid Growth Medium | Nutrient-rich, serum-free foundation for preparing complete organoid media [73]. |
| Essential Growth Factors | Recombinant Human EGF, R-spondin-1, Noggin, Wnt3A, FGF-10, HGF | Activate stem cell maintenance and proliferation pathways (e.g., Wnt/β-catenin); suppress differentiation signals; tissue-specific factors promote growth [73]. |
| Small Molecule Inhibitors | A83-01 (TGF-β inhibitor), SB202190 (p38 inhibitor), Y-27632 (ROCK inhibitor) | Inhibate pathways that promote differentiation or anoikis; enhance survival of stem/progenitor cells, especially during initial plating and passaging [73]. |
| Supplement | B-27 Supplement (Serum-Free), N-2 Supplement | Provides hormones, vitamins, transferrin, and other essential components for epithelial cell growth and function. |
| Dissociation Enzyme | Collagenase/Dispase, TrypLE Express, Accutase | Gently dissociates tissue specimens or passaged organoids into cell clusters or single cells while maintaining viability. |
| Viability Assay (3D-optimized) | CellTiter-Glo 3D Cell Viability Assay | Luminescent assay designed to penetrate 3D structures and measure ATP content as a correlate of cell viability for drug screening. |
Translational Relevance in Target-Based vs. Phenotypic Drug Discovery with Natural Products
The shift from simple cell lines to complex PDO models represents a parallel evolution from reductionist, target-based drug discovery towards more physiologically relevant, phenotypic drug discovery (PDD). This shift is particularly critical for natural products research, where mechanisms of action are often unknown at the outset [6].
Target-Based Discovery in 2D Models: Traditional screening of natural product libraries against a single molecular target (e.g., an enzyme or receptor) in 2D cell lines is efficient but flawed. It fails to account for compound permeability, metabolism, and activity within a tissue context, leading to high rates of attrition in later stages. The complex chemical space of natural products often involves polypharmacology, which is poorly assessed by single-target assays [6].
Phenotypic Discovery in 3D PDO Models: Screening natural product extracts or compounds in PDOs constitutes a powerful phenotypic approach. The readout—tumor organoid death or growth inhibition—integrates compound effects across multiple cell types and pathways within a native tissue architecture. A PDO model can reveal the functional consequence of a natural product's polypharmacology. When a response is observed, subsequent multi-omic analysis (genomics, transcriptomics) of the responsive vs. non-responsive PDOs can be used to deconvolute the mechanism of action and identify predictive biomarkers [71] [73] [72].
This paradigm directly addresses the historical challenges of natural products research. By using a human disease-relevant model as the primary filter, researchers can simultaneously:
- Identify promising bioactive leads with inherent clinical predictive value.
- Stratify which patient tumors (via their PDOs) are most likely to respond.
- Use the biological data from the screening to guide the isolation and optimization of the active compound(s).
The establishment of living PDO biobanks [71] is the infrastructural key to this approach, enabling the reproducible, large-scale phenotypic screening of natural products across a genetically diverse population of human tumors, thereby directly improving translational relevance and accelerating the path to personalized therapy.
The discovery of therapeutics from natural products (NPs) has long navigated two distinct philosophical and methodological pathways: the target-based approach and the phenotypic screening approach [74]. The target-based paradigm, fueled by advances in molecular biology, focuses on modulating a predefined, well-characterized molecular target implicated in a disease pathway. In contrast, phenotypic screening identifies compounds based on their observable effects on cells, tissues, or whole organisms, agnostic to the specific mechanism of action (MoA) [6]. Historically, phenotypic screening has been a prolific source of first-in-class medicines, particularly from complex natural extracts, but it faces the significant challenge of target deconvolution—identifying the precise molecular target(s) responsible for the observed phenotype [74].
Artificial Intelligence (AI) and Machine Learning (ML) are now transforming both paradigms, offering tools to accelerate discovery and bridge the gap between them [75] [76]. AI enhances target-based workflows by predicting interactions between NP-derived compounds and protein targets through virtual screening and molecular docking at unprecedented scale [77]. It augments phenotypic screening by using pattern recognition in high-content imaging or transcriptomic data to predict MoA and prioritize hits [74]. However, the effective integration of AI into NP research is critically dependent on two interrelated pillars: model interpretability and bias mitigation.
Interpretability is paramount in NP research due to the multi-target, multi-component nature of many natural extracts [77]. Understanding why an AI model predicts a particular bioactivity is essential for validating leads, elucidating complex pharmacological networks, and guiding synthetic optimization. Concurrently, the data used to train these models—often drawn from heterogeneous sources like ChEMBL, PubChem, or in-house libraries—can harbor systematic biases. These biases may stem from the over-representation of certain chemical scaffolds, the under-sampling of specific biological taxa, or the use of non-standardized assay protocols [78] [79]. If unaddressed, such biases can lead to models that perform well only on narrow, non-representative slices of chemical and biological space, ultimately undermining their predictive power and translational potential in drug development [78].
This guide provides an objective, data-driven comparison of AI-enhanced workflows for target-based and phenotypic NP discovery. It focuses on evaluating strategies to improve model interpretability and mitigate data bias, presenting experimental data, methodological protocols, and practical resources to empower researchers in making informed choices for their discovery campaigns.
Comparative Framework: Target-Based vs. Phenotypic AI Workflows
The integration of AI into NP research creates distinct yet occasionally convergent workflows for target-based and phenotypic approaches. The following table summarizes the core applications, interpretability challenges, and primary data bias sources for each paradigm.
Table 1: Core Comparison of AI-Enhanced Target-Based and Phenotypic Workflows in NP Research
| Aspect | Target-Based AI Workflow | Phenotypic AI Workflow |
|---|---|---|
| Primary AI Application | Virtual screening, Molecular docking scoring, Binding affinity prediction, De novo design of target-focused libraries [77] [76]. | High-content image analysis, Phenotypic profile matching (e.g., to reference MOA profiles), Hit prioritization & expansion, Predictive target deconvolution [74]. |
| Key Interpretability Need | Understanding structural determinants of binding (e.g., key interacting residues, pharmacophore features). Mapping multi-target polypharmacology networks for NPs [77]. | Translating a complex phenotypic "fingerprint" (image, gene expression) into a biologically understandable MOA hypothesis. Identifying which features of the input data drove the classification [74]. |
| Dominant Data Sources | Protein structures (experimental, AlphaFold), Bioactivity databases (ChEMBL, BindingDB), Compound libraries with annotated target activities [74] [80]. | Cell painting, transcriptomics (RNA-seq), high-throughput microscopy data. Public repositories like the LINCS L1000 database [74]. |
| Major Bias Sources | Bias towards well-studied "druggable" target families (kinases, GPCRs). Under-representation of novel or difficult-to-assay targets. Skewed chemical space of synthetic libraries used for training [80] [81]. | Bias from specific cell lines used (e.g., over-reliance on cancer lines like NCI-60). Batch effects in imaging or sequencing. Historical bias towards cytotoxic phenotypes in NP screening [78] [82]. |
| Typical Output | A ranked list of NP compounds or derivatives predicted to potently and selectively modulate a specific target. | A ranked list of NP extracts or compounds predicted to induce a desired phenotype, often with a proposed MOA or target class [74]. |
The workflows for both approaches, highlighting where interpretability tools and bias checks are integrated, are illustrated below.
Experimental Data & Performance Comparison
Case Study: AI-Assisted Target Deconvolution from Phenotypic Screening
A pivotal 2025 study demonstrated a data-driven workflow that bridges phenotypic screening and target-based discovery [74]. Researchers mined the ChEMBL database to create a library of 87 highly selective tool compounds, each with well-defined, potent activity against a single human target. This library was screened against the NCI-60 panel of human cancer cell lines at 10 µM.
Table 2: Experimental Results from Phenotypic Screening of Selective Tool Compounds [74]
| Metric | Result | Implication for AI Model Development |
|---|---|---|
| Total Compounds Screened | 87 | Defines the scale of the experimental validation set. |
| Compounds with Relevant\nMammalian Targets | 38 | Highlights the need for precise biological source filtering in training data. |
| Active Compounds (>80% Growth\nInhibition in ≥1 Cell Line) | 10 (26% of 38) | Provides a phenotypic activity benchmark for selective compounds. |
| Selective Compounds (Total\nSelectivity Score >4) | 7 out of 10 actives | Validates that computational selectivity scoring can predict compounds with focused, interpretable phenotypes. |
| Key Targets Identified | RORγ, HSF1, others | Provides ground-truth data for training AI models to link specific phenotypic responses to target modulation. |
- Experimental Protocol: The selectivity score for tool compound selection was calculated by programmatically analyzing ChEMBL data: awarding positive points for active data on the primary target (pChEMBL >6) and inactive data on other targets (pChEMBL <5), and penalizing points for activity on other targets [74]. This method created a labeled dataset ideal for training supervised ML models to predict which targets are likely responsible for an observed phenotype.
Case Study: Interpretability Through Similarity Analysis of NP Scaffolds
A 2023 study provided a framework for improving the interpretability of NP mechanisms by focusing on chemical similarity [77]. The research hypothesized that NPs sharing a core scaffold likely share similar MoAs. To test this, they systematically compared oleanolic acid (OA) and hederagenin (HG)—two structurally similar triterpenoids—against a structurally distinct control, gallic acid (GA).
Table 3: Comparative Analysis of Similar Natural Product Compounds [77]
| Analysis Method | Comparison (OA vs. HG) | Comparison (OA/HG vs. GA) | Interpretability Insight for AI |
|---|---|---|---|
| Descriptor Similarity\n(Euclidean Distance) | Low Distance (Indicating High Similarity) | High Distance | Confirms that simple chemical descriptors can reliably cluster NPs with shared scaffolds, a useful feature for model explanation. |
| Shared Druggable Targets\n(via BATMAN-TCM Platform) | High Overlap | Low Overlap | Validates that structural similarity predicts target profile similarity, supporting the use of similarity-based reasoning in AI explanations. |
| Over-Representation Analysis\nof KEGG Pathways | Significant Shared Pathways (e.g., Lipid & Atherosclerosis) | Divergent Pathways | Demonstrates that similar compounds perturb similar biological networks, allowing AI to explain predictions via pathway mapping. |
- Experimental Protocol: The study combined: 1) Calculation of 1,116 molecular descriptors using the Mordred library, 2) Prediction of druggable targets using the BATMAN-TCM platform (DTI score ≥10), and 3) Large-scale molecular docking of the compounds against a druggable proteome [77]. This multi-pronged approach provides a template for generating rich, interpretable datasets that link NP structure to target and pathway space.
Benchmarking Bias Mitigation Strategies in Phenotypic Modeling
A comprehensive 2024 benchmarking study explicitly evaluated bias and fairness in electronic phenotyping models, offering critical data for developing robust AI workflows [78]. The study assessed nine different phenotyping algorithms (from rule-based to deep learning) and five bias mitigation strategies on tasks involving pneumonia and sepsis identification from electronic health records.
Table 4: Performance of Bias Mitigation Strategies on a Phenotyping Model [78]
| Bias Mitigation Category | Example Strategy | Key Benchmarking Finding | Recommendation for NP Research |
|---|---|---|---|
| Pre-processing | Reweighting, Disparate Impact Remover | Effectively reduced demographic parity difference but could slightly reduce overall accuracy. | Apply during dataset curation to balance representation of NPs from different sources or structural classes before model training. |
| In-processing | Adversarial Debiasing, Meta Fair Classifier | Directly optimized fairness during training; performance varied significantly by model architecture. | Implement when using complex, deep learning-based phenotype classifiers to enforce fairness constraints. |
| Post-processing | Reject Option Classification, Calibrated Equalized Odds | Adjusted predictions after training; effective but requires careful threshold tuning. | Useful as a final correction step on a deployed model to ensure equitable hit rates across different cell lines or assay conditions. |
The study concluded that no single debiasing strategy was universally superior, emphasizing the need for a bespoke, context-aware approach to bias mitigation in AI-driven discovery [78].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 5: Key Reagents, Databases, and Software for Interpretable and Bias-Aware AI Research
| Item Name | Type | Primary Function in AI Workflow | Relevance to Interpretability/Bias |
|---|---|---|---|
| ChEMBL Database [74] | Public Bioactivity Database | Provides millions of curated bioactivity data points for training target prediction and selectivity models. | Source of historical assay bias; requires careful filtering (e.g., by organism, assay type) for robust model building. |
| NCI-60 Human Tumor Cell Lines [74] | Biological Resource | Standardized panel for phenotypic anticancer screening, generating reproducible, comparable data. | Represents a specific, limited biological context; models trained solely on NCI-60 data may not generalize to other tissue or disease types. |
| BATMAN-TCM Platform [77] | Computational Tool/DB | Predicts drug-target interactions and network pharmacology for natural products. | Provides a pre-computed knowledge base for explaining AI predictions via target and pathway enrichment. |
| Mordred Descriptor Calculator [77] | Software Library | Calculates a comprehensive set of 1,826 2D/3D molecular descriptors from chemical structures. | Generates interpretable feature vectors for ML models; allows similarity analysis to justify predictions. |
| Selective Tool Compound Library [74] | Physical Compound Library | A set of chemically diverse compounds with high potency and selectivity for individual targets. | Serves as a gold-standard experimental validation set for testing AI models performing target deconvolution. |
| Adversarial Debiasing Algorithm [78] | AI Software Module | An in-processing technique that removes dependency of predictions on sensitive attributes (e.g., demographic group). | Directly addresses model bias by altering the learning objective during model training. |
| Natural Product Knowledge Graph (Concept) [79] | Data Architecture | A unified, multimodal data structure linking NPs to genomic, spectroscopic, and bioactivity data. | The ideal framework for causal inference and reducing bias by integrating diverse, complementary data sources. |
Integrated Workflow for Bias Mitigation in NP AI Models
Addressing bias is not a single step but an integrated process throughout the AI lifecycle. The following diagram details a recommended workflow, incorporating strategies from the benchmarking study [78], applied to the context of NP discovery.
The convergence of AI with natural products research presents a powerful avenue for revitalizing drug discovery. As evidenced by the experimental data and case studies presented, both target-based and phenotypic workflows benefit significantly from AI, but their success is contingent on rigorous attention to interpretability and bias.
For Target-Based Workflows: Prioritize interpretability methods that elucidate the structural basis of predictions, such as interaction maps from docking studies or saliency maps from graph neural networks. Actively mitigate bias by curating balanced training sets that include data on understudied target classes and diverse NP scaffolds beyond traditional "druggable" chemical space [80] [81].
For Phenotypic Workflows: Employ multi-modal AI models that can integrate image, gene expression, and chemical data to generate richer, more explainable MOA hypotheses [74] [79]. Implement a systematic bias mitigation pipeline as outlined in Section 5, starting with an audit of training data for representativeness across biological models (e.g., cell lines, primary cells) and phenotypic endpoints [78].
A forward-looking strategy involves investing in the development and adoption of a unified Natural Product Knowledge Graph [79]. This multimodal data structure, linking compounds to genes, spectra, assays, and literature, is the most promising infrastructure for enabling causal inference, reducing fragmented data biases, and building AI systems that truly emulate the nuanced decision-making of expert natural product scientists. By adopting these comparative insights and tools, researchers can enhance the reliability, fairness, and translational impact of AI-driven predictive workflows in natural product-based drug development.
Benchmarking Success: Validating, Comparing, and Translating Assay Data for NPs
Drug discovery operates through two principal, complementary strategies: target-based drug discovery (TDD) and phenotypic drug discovery (PDD). The TDD approach is a hypothesis-driven, molecular strategy that begins with the selection of a specific, well-validated protein target believed to have a causal role in disease. In contrast, PDD is an empirical, biology-first approach that identifies compounds based on their ability to modulate a disease-relevant phenotype in cells, tissues, or whole organisms, without preconceived notions of the molecular target [1] [10].
The central thesis of modern drug development posits that the choice between these strategies represents a fundamental trade-off between two critical performance metrics: translational predictivity and mechanistic certainty. Translational predictivity refers to the likelihood that a compound’s activity in a preclinical model will successfully translate to therapeutic efficacy and safety in human patients. Mechanistic certainty refers to the depth of understanding regarding a compound’s precise molecular mechanism of action (MMOA), including its direct protein target(s) and downstream biochemical effects [10].
Historically, the molecular biology revolution shifted the industry overwhelmingly toward TDD, prioritizing mechanistic certainty. However, a seminal analysis revealed that between 1999 and 2008, a majority of first-in-class small molecule medicines were discovered through phenotypic approaches [1] [10]. This resurgence of PDD acknowledges that complex diseases often involve redundant pathways and polygenic contributions, which may be more effectively modulated by compounds identified through phenotypic, systems-level screening. The challenge for researchers, particularly in the complex arena of natural products research, is to strategically select and integrate these approaches to balance the need for robust clinical translation with the desire for clear mechanistic understanding [33].
Comparative Performance Analysis: Quantitative Metrics
The performance of PDD and TDD can be evaluated across several key quantitative and qualitative dimensions, as summarized in the table below. These metrics are derived from historical analyses of approved drugs and the documented experiences of industrial and academic screening campaigns [1] [10] [33].
Table 1: Comparative Performance Metrics for Phenotypic and Target-Based Drug Discovery
| Performance Metric | Phenotypic Discovery (PDD) | Target-Based Discovery (TDD) | Supporting Data & Notes |
|---|---|---|---|
| Success Rate (First-in-Class) | Higher | Lower | Analysis shows PDD more successful for first-in-class medicines [1]. |
| Success Rate (Follower Drugs) | Lower | Higher | TDD excels at developing improved drugs for validated targets/mechanisms [62]. |
| Typical Screening Throughput | Medium to High | Very High | PDD assays (e.g., high-content imaging) can be complex; TDD biochemical assays are typically ultra-HTS [33]. |
| Translational Predictivity | Potentially High (with complex models) | Variable | PDD using physiologically relevant disease models may better capture efficacy; TDD can fail due to poor target validation or compound toxicity [10] [83]. |
| Mechanistic Certainty at Lead ID | Low (Target unknown) | High (Target known) | Target deconvolution is a major bottleneck in PDD [62]. |
| Time/Cost to Lead Compound | Higher | Lower | PDD often involves more complex models and follow-up target ID work [33]. |
| "Druggable" Target Space | Expansive, includes novel/unknown targets | Limited to known, validated targets | PDD has identified modulators of protein folding, splicing, and multi-protein complexes [10]. |
| Risk of Attrition (Clinical) | Shifts risk earlier in pipeline | Shifts risk later in pipeline | PDD front-loads risk via complex biology; TDD back-loads risk if target relevance to human disease is flawed [10]. |
Experimental Paradigms and Protocols
The execution of PDD and TDD relies on distinct experimental workflows, each with its own protocols for screening, validation, and lead characterization.
Phenotypic Screening and Target Deconvolution
A standard PDD workflow begins with the development of a disease-relevant assay that measures a functional phenotype (e.g., cell death, neurite outgrowth, viral replication). After screening a compound library, hit validation involves confirming dose-responsive activity in secondary phenotypic assays. The critical and challenging next step is target deconvolution. Label-free methods have become essential, especially for natural products, which are often difficult to chemically modify [62].
Key Experimental Protocols in Phenotypic Discovery:
Cellular Thermal Shift Assay (CETSA): This protocol detects ligand-induced changes in protein thermal stability.
- Method: Cells or lysates are treated with the compound or vehicle, heated to a range of temperatures, and centrifuged. The remaining soluble protein in the supernatant is quantified via immunoblotting or mass spectrometry.
- Application: Confirms direct target engagement in a cellular context. A shift in the protein's melting curve indicates compound binding [62].
Drug Affinity Responsive Target Stability (DARTS): This method leverages compound-induced protection of the target protein from proteolysis.
- Method: Cell lysates are incubated with the compound or vehicle and then subjected to limited proteolysis with a non-specific protease (e.g., pronase). The proteolyzed samples are analyzed by SDS-PAGE or mass spectrometry.
- Application: Identifies potential target proteins by detecting protein bands/fragments stabilized (i.e., less degraded) in the compound-treated sample [62].
Thermal Proteome Profiling (TPP): A proteome-wide application of the thermal stability principle.
- Method: Compound- and vehicle-treated cells are heated across a temperature gradient. Soluble proteins are digested and analyzed by quantitative mass spectrometry (e.g., TMT or label-free).
- Application: An unbiased method to identify the full spectrum of proteins whose thermal stability is altered by the compound, revealing direct and indirect targets across the proteome [62].
Target-Based Screening and Mechanistic Validation
The TDD workflow starts with the production and purification of a recombinant target protein. A biochemical assay is developed to measure the target's activity (e.g., enzyme kinetics, receptor-ligand binding). High-throughput screening (HTS) identifies inhibitors/activators, which are then validated in orthogonal binding assays (e.g., SPR, ITC) and cellular assays confirming target modulation.
Key Experimental Protocol: Knowledge-Based Mechanistic Modeling (e.g., ISELA Model) This protocol integrates multiscale data to build a predictive model of drug action, exemplified by the In Silico EGFR-mutant LUAD (ISELA) model for gefitinib [83].
- Method:
- Context Definition: Define the clinical question (e.g., predicting tumor progression in EGFR-mutant lung adenocarcinoma patients on TKI therapy).
- Knowledge Model Construction: Synthesize literature and experimental data into a diagram of key pathological interplay (e.g., EGFR signaling, mutation-driven proliferation, apoptosis).
- Computational Implementation: Translate the knowledge model into a system of ordinary differential equations representing biological processes.
- Model Calibration: Fit the model's parameters using in vitro (signaling, cell viability) and in vivo (xenograft tumor volume) data.
- Model Validation: Test the model's predictive power against independent clinical trial data (e.g., time to progression from the LUX-Lung 7 trial) not used in calibration [83].
- Application: Provides high mechanistic certainty by quantitatively linking drug-target engagement to cellular and clinical outcomes, enabling prediction of patient subpopulation responses.
Visualization of Workflows and Pathways
Comparative Drug Discovery Workflow
The following diagram illustrates the divergent starting points and convergent goals of phenotypic and target-based screening strategies.
Signaling Pathway for Mechanistic Modeling
The diagram below depicts a simplified EGFR signaling pathway in Lung Adenocarcinoma (LUAD), which forms the core knowledge for mechanistic models like ISELA [83]. This visual representation aids in understanding the system that target-based approaches aim to modulate with precision.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for PDD and TDD
| Reagent/Material | Primary Function | Relevance to PDD/TDD |
|---|---|---|
| Disease-Relevant Cell Lines | Provide a physiological context for phenotypic screening (PDD) and cellular target validation (TDD). Includes primary cells, iPSC-derived cells, and engineered lines with disease mutations. | Critical for both. PDD uses them as the primary screening platform; TDD uses them for secondary confirmation [10] [83]. |
| Compound Libraries | Collections of small molecules for screening. Diversity libraries are used in PDD; focused/targeted libraries are used in TDD. Natural product libraries are a key subset for novel chemistry. | Foundational for both. Library choice defines the chemical exploration space [33]. |
| Thermostable Protein Standards | Internal controls for mass spectrometry-based proteomics. Used to normalize protein abundance measurements across samples in TPP. | Essential for label-free target ID in PDD (e.g., TPP) [62]. |
| Protease Cocktails (e.g., Pronase) | Enzyme mixtures for limited, non-specific proteolysis. Used to digest unprotected proteins in the DARTS protocol. | Key reagent for the DARTS target deconvolution method in PDD [62]. |
| Tandem Mass Tag (TMT) Reagents | Isobaric chemical labels for multiplexed quantitative proteomics. Allow simultaneous comparison of protein abundance from up to 16 different samples (e.g., different temperatures in TPP). | Enables high-throughput, quantitative thermal proteome profiling in PDD [62]. |
| Recombinant Target Protein | Purified, functional protein for biochemical assay development. | The cornerstone reagent for initiating a TDD campaign [33]. |
| Mechanistic Computational Model | Integrated software platform (e.g., using R, Python, MATLAB) that encodes biological pathways into mathematical equations for simulation. | Core tool for building and executing predictive models like ISELA in TDD, enhancing mechanistic certainty [83]. |
A Framework for Contrast Analysis
The performance debate between PDD and TDD can be rigorously analyzed using a contrast analysis framework [84] [85]. This statistical method tests specific, directional hypotheses about the pattern of outcomes (e.g., success rates, development times) across different discovery approaches, rather than simply asking if any differences exist.
- Defining the Contrast Weights: For a simplified analysis comparing PDD, TDD for novel targets, and TDD for validated targets, researchers might hypothesize a specific success rate pattern based on historical data [1] [10]:
- Hypothesis (H1): PDD success > TDD (Novel) success > TDD (Validated) success. This reflects the strength of PDD for first-in-class drugs and the challenge of novel target validation in TDD.
- Contrast Weights (λ): Assign numerical weights reflecting this predicted order (e.g., +2 for PDD, +1 for TDD-Novel, -3 for TDD-Validated), ensuring they sum to zero.
- Application: By applying these pre-defined weights to observed success rate data from a set of projects, a contrast analysis can test whether H1 is significantly supported. Competing hypotheses (e.g., that TDD for validated targets is superior) can be tested against each other, providing quantitative evidence to guide strategic portfolio decisions [84].
Strategic Application in Natural Products Research
Natural products present unique challenges and opportunities that influence the choice between PDD and TDD.
- Advantage of PDD: The complex, evolved structures of natural products often confer polypharmacology—simultaneous modulation of multiple targets. PDD is ideally suited to capture this synergistic, systems-level efficacy without needing to pre-define the involved targets [10]. Label-free target deconvolution methods (CETSA, TPP) are especially valuable here, as they do not require difficult chemical modification of the natural product scaffold [62].
- Role of TDD: When a natural product's primary therapeutic target is known (e.g., rapamycin and mTOR), TDD approaches can be used to conduct analogue screening or structure-based design to improve potency, selectivity, or pharmacokinetic properties. Mechanistic modeling can also predict how modifications might affect the compound's behavior within a known pathway [83].
- Integrated Strategy: An effective modern approach often involves a PDD-first, TDD-informed pipeline. A natural product library is screened in a phenotypic assay. Active hits are then subjected to label-free target identification. The revealed targets and pathways subsequently inform mechanistic follow-up using target-specific cellular and biochemical assays (TDD principles) to fully characterize the MoA and optimize the lead [33].
The dichotomy between translational predictivity and mechanistic certainty is not a problem to be solved, but a spectrum to be managed. Phenotypic discovery excels at delivering novel biology and first-in-class medicines with high translational potential, albeit with an initial mechanistic black box. Target-based discovery offers precision, efficiency, and clear development pathways for follower drugs, but its success is wholly contingent on the foundational accuracy of the target hypothesis.
The future of drug discovery, particularly with complex natural products, lies in strategic integration. This involves using complex, human-relevant phenotypic models (e.g., organoids, zebrafish) to maximize translational predictivity early on, followed by the application of advanced 'omics technologies and mechanistic modeling to illuminate the black box and build mechanistic certainty. Computational approaches, including AI and multi-scale systems pharmacology models like ISELA, will be pivotal in bridging these two worlds, predicting how phenotypic hits might function mechanistically and how target-focused compounds will behave in integrated biological systems [83] [86]. By framing decisions through the lens of these two core performance metrics, research teams can make more informed, evidence-based choices to advance the next generation of therapeutics.
The discovery of therapeutics from natural products (NPs) stands at a crossroads between two fundamental philosophical and methodological approaches: target-based drug discovery (TDD) and phenotypic drug discovery (PDD). The TDD paradigm, a hallmark of the molecular biology era, begins with the selection and validation of a purified protein target implicated in a disease, followed by screening for compounds that modulate its activity [87]. In contrast, the PDD paradigm interrogates compounds directly in a cellular or organismal system to elicit a desired therapeutic phenotype, deferring target identification until after a bioactive compound is found [87] [10]. This reverse chemical genetics versus forward chemical genetics framework mirrors classic genetic strategies [87].
For NP research, this dichotomy presents unique challenges and opportunities. NPs are evolutionarily optimized for biological interaction, often possessing complex structures and polypharmacological profiles that can be poorly suited to reductionist, single-target screening [6]. Historically, NPs were discovered through phenotypic observations in humans or animals, with their molecular mechanisms elucidated much later [10]. The contemporary resurgence of PDD, driven by analyses showing its disproportionate yield of first-in-class medicines, compels a critical re-evaluation of how to best leverage each approach for unlocking the therapeutic potential of NPs [10] [88]. This guide provides an objective, data-driven comparison of TDD and PDD performance, with a focus on applications and evidence within NP research.
Performance Comparison: A Data-Driven Analysis
The optimal choice between phenotypic and target-based screening is context-dependent, hinging on project goals, biological understanding of the disease, and available tools. The following table summarizes the core strengths, limitations, and ideal use cases for each paradigm, synthesizing evidence from recent studies and applications.
Table: Comparative Performance of Phenotypic and Target-Based Screening in Natural Product Research
| Aspect | Phenotypic Screening (PDD) | Target-Based Screening (TDD) |
|---|---|---|
| Primary Strength | Discovers novel biology and MOAs; "pre-validates" targets in a disease-relevant context; effective for polygenic/complex diseases [87] [10] [89]. | High throughput, efficiency, and mechanistic clarity from the outset; enables rational, structure-based design [87] [90]. |
| Key Weakness | Target deconvolution is complex, costly, and can fail; hit optimization can be challenging without a known target; lower initial throughput [87] [91] [89]. | Relies on a priori, often imperfect target validation; may miss relevant biology or efficacious compounds; poor for identifying multi-target/polypharmacology drugs [87] [10]. |
| Success Rate (First-in-Class) | Historically higher. Analysis (1999-2008) showed more first-in-class small-molecule drugs originated from PDD [10] [89]. | Lower for first-in-class drugs, but highly effective for developing "best-in-class" agents against validated targets [10] [88]. |
| Suitability for Natural Products | High. Unbiased to target, can capture polypharmacology and novel MOAs inherent to many NPs. Ideal for exploring NP mixtures (e.g., herbal extracts) [12] [6]. | Moderate to Low. Requires purified, single components. May miss bioactive NPs that work through novel, unanticipated, or complex targets [6]. |
| Target "Druggable" Space | Expands the space. Discovers drugs for targets with no known function (e.g., NS5A) or complex cellular machines (e.g., spliceosome) [10]. | Limited to known, traditionally "druggable" target classes (e.g., enzymes, GPCRs). |
| Lead Optimization Path | Can proceed empirically via structure-activity relationship (SAR) on the phenotype. Target knowledge accelerates optimization and safety profiling [10]. | Straightforward, driven by target potency and selectivity assays. Medicinal chemistry is highly focused. |
| Key Risk | Clinical translation risk is lower due to disease-relevant models, but late-stage attrition can occur if MOA/toxicity is poorly understood [10]. | High risk of clinical failure if the chosen target is not causally linked to the human disease phenotype [87] [88]. |
Experimental Protocols: Key Methodologies from Recent Studies
An Integrated Phenotypic-to-Target Workflow for Natural Products (NP-VIP Strategy)
A 2024 study developed a novel "Natural Product Virtual screening-Interaction-Phenotype" (NP-VIP) strategy to overcome the major challenge of target deconvolution for complex NP mixtures [12]. Using Salvia miltiorrhiza (Danshen) for ischemic stroke as a case study, the protocol integrates three complementary layers of evidence.
1. Virtual Screening (VS) for Target Hypothesis Generation:
- Objective: Rapidly identify potential protein targets for compounds in the NP extract.
- Procedure: The chemical profile of the S. miltiorrhiza extract is established via UPLC-high-resolution mass spectrometry. The structures of identified compounds are used for molecular docking against protein target libraries using tools like LeDock. Potential targets are ranked by docking scores [12].
2. Cellular Thermal Shift Assay (CETSA) for Direct Binding Validation:
- Objective: Experimentally confirm direct physical interaction between the NP extract and candidate target proteins in a cellular context.
- Procedure: Cells are treated with the NP extract or vehicle control. Cell aliquots are heated to a range of temperatures (e.g., 37°C to 65°C) to denature proteins. The stabilized target proteins (bound by the compound) require higher temperatures to denature. The soluble protein fraction is isolated and quantified via Western blot or quantitative proteomics (e.g., TMT labeling) to identify proteins with shifted thermal stability [12].
3. Phenotypic Metabolomics for Functional Validation:
- Objective: Link compound-target interactions to a functional, phenotypic outcome to identify the most therapeutically relevant targets.
- Procedure: Cells (e.g., PC12 neuron-like cells) under disease-relevant stress (e.g., oxygen-glucose deprivation for stroke model) are treated with the NP extract. Global metabolomic profiling is performed via LC-MS. Significant changes in metabolite pathways (e.g., glutamate metabolism, antioxidant levels) are analyzed. Proteins that are nodes in these altered pathways, and which also appeared in the VS and CETSA datasets, are prioritized as high-confidence therapeutic targets [12].
4. Integration and Validation:
- Objective: Synthesize data to identify high-confidence targets.
- Procedure: Targets identified from VS, CETSA, and metabolomics are intersected. For S. miltiorrhiza, this yielded five high-confidence targets (PARP1, STAT3, APP, GLUL, GAD67). Their roles in the NP's neuroprotective effect were validated via gene knockdown/overexpression and functional rescue experiments [12].
A Target-Based Screening Protocol for Natural Product Derivatives
Objective: To identify and optimize selective inhibitors of a validated enzymatic target from a library of synthetic derivatives based on an NP core scaffold.
Procedure:
- Target and Assay Development: A purified, recombinant form of the target enzyme (e.g., a kinase, protease) is prepared. A biochemical assay is developed, often using a fluorescent or luminescent readout of enzymatic activity (e.g., ATP consumption, substrate cleavage).
- High-Throughput Screening (HTS): A library of semi-synthetic NP analogues is screened against the target at a single concentration (e.g., 10 µM) in a 384- or 1536-well plate format. Primary hits are compounds that inhibit >70% of activity.
- Dose-Response and Selectivity Profiling: Primary hits are re-tested in a dose-response curve to determine IC50 values. Selectivity is assessed against a panel of related enzymes (e.g., the kinome for a kinase inhibitor) to identify potential off-target effects.
- Cellular Target Engagement: Confirmed inhibitors are tested in cells expressing the target. A cellular pharmacodynamics (PD) assay, such as monitoring phosphorylation of a downstream substrate via Western blot, confirms the compound engages the target in a physiologically relevant environment.
- Phenotypic Triaging: Compounds with potent cellular PD activity are then evaluated in a disease-relevant phenotypic assay (e.g., inhibition of cancer cell proliferation, reduction of inflammatory cytokine release). This step is critical to ensure that biochemical inhibition translates to the desired functional outcome [87].
Visualizing Strategies and Workflows
Diagram 1: Conceptual Workflow of Target-Based and Phenotypic Drug Discovery.
Diagram 2: Integrated NP-VIP Strategy for Target Deconvolution.
The Researcher's Toolkit: Essential Solutions for NP Screening
Table: Key Reagents and Technologies for Comparative Screening Studies
| Tool/Reagent | Primary Function | Application Context | Key Considerations |
|---|---|---|---|
| Human iPSC-Derived Cells | Provide disease-relevant, patient-specific cellular models for phenotypic screening [10] [90]. | PDD for neurodegenerative, cardiac, and other complex diseases. | Cost, differentiation protocol maturity, and phenotypic assay robustness can be challenging [91]. |
| 3D Culture Systems (Organoids, Spheroids) | Model tissue-like architecture, cell-cell interactions, and microenvironmental gradients [91] [89]. | PDD for oncology, toxicology, and developmental biology. | Throughput is lower than 2D, and standardization of assays is an active area of development [89] [90]. |
| Chemical Proteomics Kits (e.g., CETSA, Photoaffinity Probes) | Identify direct protein-binding partners of small molecules in complex biological lysates or live cells [87] [12]. | Target deconvolution in PDD; off-target profiling in TDD. | Requires compound modification for some methods; data analysis requires robust proteomics infrastructure [87] [12]. |
| High-Content Imaging (HCI) Systems | Automate the quantification of complex cellular phenotypes (morphology, protein localization, cell counting) [91] [89]. | Essential for sophisticated phenotypic screens (e.g., neurite outgrowth, infection). | High capital cost; requires expertise in image analysis and bioinformatics. |
| Tandem Mass Tag (TMT) Reagents | Enable multiplexed, quantitative proteomics for analyzing hundreds of samples in parallel [12]. | Used in CETSA and global proteomic profiling for target ID and mechanism studies. | High sensitivity mass spectrometer required; ratio compression can be a technical issue. |
| Validated Target-Based Assay Kits (Kinase, Epigenetic, etc.) | Provide optimized, ready-to-use biochemical assays for specific target classes. | TDD screening and selectivity profiling. | Quality and relevance of the enzyme construct (e.g., post-translational modifications, activation state) are critical. |
| Global Natural Products Social Molecular Networking (GNPS) | An open-access online platform for sharing and analyzing mass spectrometry data to dereplicate and annotate NPs [12] [6]. | Essential for characterizing NP libraries and identifying novel analogs. | Relies on community-contributed data; structural annotation from MS/MS data alone can be tentative. |
In natural products research, the journey from identifying a bioactive compound to understanding its mechanism of action presents a significant challenge. The field has long navigated between two primary discovery paradigms: target-based screening, which starts with a known protein, and phenotypic screening, which begins with an observed biological effect in cells or organisms [39]. Phenotypic approaches are particularly valuable for natural products, as they can reveal novel biology without preconceived target biases; however, the subsequent "target deconvolution" phase—identifying the specific molecular protein target(s)—becomes a major bottleneck [40]. Integrative validation frameworks that combine direct target-engagement assays like the Cellular Thermal Shift Assay (CETSA), broad proteomic profiling, and phenotypic readouts are emerging as powerful solutions to bridge this gap, accelerating the development of natural product-derived therapeutics [92] [16].
Comparative Performance of Target Engagement and Deconvolution Methods
Selecting the appropriate method for target validation and identification depends on the research question, stage of discovery, and the properties of the compound and target protein. The following tables provide a detailed comparison of key label-free techniques and advanced proteomic platforms.
Table 1: Comparison of Label-Free Target Engagement and Deconvolution Methods [39] [93] [94]
| Method | Core Principle | Typical Sample Type | Throughput | Key Advantages | Primary Limitations |
|---|---|---|---|---|---|
| CETSA (Cellular Thermal Shift Assay) | Detects ligand-induced thermal stabilization of proteins [39]. | Live cells, cell lysates, tissues [93]. | Medium (WB) to High (MS) [39]. | Works in physiologically relevant intact cells; can quantify engagement (ITDR) [93] [94]. | Requires significant thermal shift; data interpretation requires biophysical understanding [95]. |
| DARTS (Drug Affinity Responsive Target Stability) | Detects protection from proteolysis upon ligand binding [94]. | Cell lysates, purified proteins [94]. | Low to Medium [39]. | No compound modification; detects subtle conformational changes [94]. | Sensitivity depends on protease choice; challenges with low-abundance targets [39]. |
| SPROX (Stability of Proteins from Rates of Oxidation) | Measures methionine oxidation rates under chemical denaturation to detect domain-level stability shifts [39]. | Cell lysates [93]. | Medium to High [39]. | Provides potential binding site information [93]. | Limited to methionine-containing peptides; requires MS expertise [39]. |
| MS-CETSA / TPP (Thermal Proteome Profiling) | CETSA coupled with mass spectrometry for proteome-wide profiling [93]. | Live cells, lysates, tissues [96]. | High (Proteome-wide) [92]. | Unbiased discovery of on- and off-targets; reveals pathway-level effects [92] [96]. | Resource-intensive; complex data processing [39]. |
| Affinity-Based Proteomics | Uses immobilized compound (e.g., biotin-tagged) to "pull down" binding partners [39]. | Cell lysates [39]. | Low [39]. | High specificity when reagents are optimal [39]. | Requires compound modification, which may alter activity/selectivity [39] [40]. |
Table 2: Comparison of High-Throughput Proteomic Platforms for Biomarker and Pathway Analysis [97] This data informs the proteomics component of an integrative framework, highlighting platform selection trade-offs.
| Platform Feature | Olink Explore 3072 (Immunoassay) | SomaScan v4 (Aptamer) | Context for Integrative Frameworks |
|---|---|---|---|
| Assay Principle | Proximity Extension Assay (PEA) with dual antibody recognition [97]. | Single-stranded DNA aptamer binding [97]. | Platform choice affects downstream pathway analysis from phenotypic screens. |
| Median CV (Precision) | 16.5% [97] | 9.9% [97] | SomaScan shows higher technical precision in plasma. |
| Median Correlation Between Matching Assays | 0.33 (Spearman) [97] | 0.33 (Spearman) [97] | Modest correlation underscores complementarity and need for orthogonal validation. |
| Proteins with Detected cis-pQTLs | 72% of assays [97] | 43% of assays [97] | Olink assays show higher proportion with genetic support for on-target measurement. |
Detailed Experimental Protocols for Key Framework Components
Protocol for MS-CETSA (Thermal Proteome Profiling) for Target Deconvolution
This protocol is adapted for identifying the unknown targets of a natural product hit from a phenotypic screen [93] [96].
- Cell Treatment & Heating: Harvest and plate cells in multiple aliquots. Treat aliquots with either the natural product compound (at a concentration eliciting the phenotypic effect) or vehicle control. Incubate under physiological conditions (e.g., 37°C) to allow cellular uptake and engagement. Subject each aliquot to a different temperature in a defined gradient (e.g., from 37°C to 67°C) in a precision thermal cycler [39] [96].
- Cell Lysis and Soluble Protein Harvest: Rapidly cool samples. Lyse cells using freeze-thaw cycles (e.g., liquid nitrogen) or detergent-free buffers to preserve protein complexes. Centrifuge at high speed to separate soluble (folded) proteins from denatured aggregates [39].
- Protein Digestion and TMT Labeling: Digest the soluble protein fraction from each temperature point with trypsin. Label the resulting peptides from different temperature aliquots with isobaric Tandem Mass Tags (TMT). Pool the TMT-labeled samples into a single vial for simultaneous analysis [93] [96].
- LC-MS/MS Analysis and Data Processing: Analyze the pooled sample via liquid chromatography coupled to tandem mass spectrometry (LC-MS/MS). The relative abundance of each peptide at each temperature point is determined by the intensity of its unique TMT reporter ions [96].
- Melting Curve Fitting and Hit Identification: For each protein identified, plot the relative soluble abundance against temperature to generate a melting curve. Fit curves to a sigmoidal model to calculate the melting temperature (Tm). Compare curves from compound-treated and vehicle-treated samples. Proteins with a significant shift in Tm (ΔTm) are considered potential direct targets or part of an affected pathway [39] [96].
Protocol for Isothermal Dose-Response CETSA (ITDR-CETSA) for Affinity Assessment
This protocol follows target deconvolution to quantify the binding affinity of a natural product to its identified target [93].
- Sample Preparation: Prepare identical aliquots of cells or cell lysate containing the target protein.
- Compound Titration: Treat each aliquot with a different concentration of the natural product, spanning a broad range (e.g., from nM to μM). Include a vehicle-only control.
- Isothermal Heating: Heat all samples at a single, fixed temperature. This temperature is pre-determined from a melt curve experiment and should be near the Tm of the unbound target protein (typically where ~50-80% of the protein is denatured in the vehicle sample) [93].
- Detection and Analysis: Process samples as in the MS-CETSA protocol (steps 2-4) using a target-specific detection method (e.g., Western blot, bead-based immunoassay, or targeted MS). Plot the amount of remaining soluble target protein against the compound concentration. Fit the data to a dose-response model to determine the EC₅₀ value, which reflects the cellular potency of engagement [93].
Protocol for Integrating Phenotypic Readouts with CETSA Data
This framework connects target engagement to functional outcome [40] [96].
- Parallel Phenotypic Assay: In a separate but contemporaneous experiment, treat cells with the same matrix of compound concentrations used in ITDR-CETSA. Measure the relevant phenotypic endpoint (e.g., cell viability, caspase activation, a specific phosphorylation signal, or a high-content imaging metric) after an appropriate duration.
- Data Correlation and MoA Elucidation: Plot the phenotypic dose-response curve alongside the target engagement dose-response (ITDR) curve. A close alignment between the EC₅₀ values for target stabilization and phenotypic effect provides strong evidence that the observed phenotype is driven by engagement with that specific target. A disconnect may indicate off-target effects, downstream signaling amplification, or a requirement for engagement of multiple targets [96].
- Pathway Validation: Use the proteome-wide data from MS-CETSA to identify other proteins with significant thermal shifts (e.g., downstream effectors or proteins in the same complex). Validate these pathway nodes with orthogonal methods like immunoblotting for post-translational modifications to build a comprehensive mechanism of action (MoA) model [96].
Workflow and Pathway Visualization
Integrative Framework for NP Drug Discovery
CETSA Principle: Ligand-Induced Thermal Stabilization
Case Study: MS-CETSA Reveals Gemcitabine Resistance Pathways [96]
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for Integrative Validation Frameworks
| Category | Item / Reagent | Function in the Workflow | Key Considerations |
|---|---|---|---|
| Core Assay Components | CETSA / Thermal Shift Assay Kits | Provide optimized buffers and protocols for protein stability assays in cells or lysates. | Useful for standardizing WB-CETSA. For MS-CETSA, components are often prepared in-house. |
| High-Affinity, Validated Antibodies | Detection of specific target proteins in WB-CETSA or bead-based CETSA HT formats [93]. | Availability and specificity for the native protein are critical limiting factors. | |
| HiBiT/LgBiT Split-Luciferase System | Enables antibody-free, high-throughput CETSA (BiTSA) for known targets in engineered cell lines [93]. | Requires CRISPR-Cas9 tagging of the endogenous target gene. | |
| Proteomics & Mass Spectrometry | Tandem Mass Tags (TMT) | Isobaric chemical labels for multiplexed quantitative proteomics in MS-CETSA/TPP [93] [96]. | Allows pooling of up to 18 samples, increasing throughput and reducing MS run variation. |
| Trypsin/Lys-C | Proteolytic enzymes for digesting soluble protein fractions into peptides for MS analysis. | Sequence-grade purity is essential for reproducible digestion. | |
| LC-MS/MS System with High Resolution | Instrumentation for separating and sequencing peptides, quantifying abundance via TMT reporter ions [96]. | Orbitrap-based systems are commonly used for depth and quantitative accuracy. | |
| Cell Biology & Phenotyping | Phenotypic Assay Reagents (e.g., viability dyes, caspase substrates, phospho-specific antibodies) | Measure the functional biological output (phenotype) triggered by the natural product. | Must be compatible with the cell model and time scales used in parallel CETSA experiments. |
| Cell Line(s) Relevant to Disease Phenotype | The biological system for all experiments. Includes sensitive/resistant pairs for MoA studies [96]. | Genetic background and authenticity are crucial for translational relevance. | |
| Data Analysis | TPP / MS-CETSA Data Analysis Software (e.g., TPP-R, MSFragger, IMPRINTS) | Specialized software packages for processing raw MS data, curve fitting, and statistical analysis of thermal shifts [96]. | A major component of the workflow; requires bioinformatics expertise or collaboration. |
Thesis Context: Target-Based vs. Phenotypic Assays in Natural Products Research
The journey of a natural product from traditional remedy to modern drug is fundamentally shaped by the strategy employed to decipher its mechanism of action. This process sits at the core of a persistent dichotomy in drug discovery: target-based versus phenotypic screening approaches [98].
Phenotypic drug discovery begins with observing a compound's effect on a cell or organism, such as the inhibition of cancer cell growth or the reduction of inflammation, without prior knowledge of the specific protein target. For decades, research on Celastrol (CEL) followed this path, meticulously cataloging its potent effects against a vast array of diseases including cancers, inflammatory disorders, and metabolic syndromes [98] [99]. While this approach successfully validated CEL's therapeutic potential, it left a critical gap: ambiguous target information. This ambiguity poses a significant challenge for modern drug development, which relies on understanding precise mechanisms to optimize efficacy, minimize toxicity, and meet regulatory standards [98] [100].
In contrast, target-based drug discovery starts with a defined molecular target implicated in a disease and seeks compounds that modulate its activity. The transition of CEL research into this paradigm has been enabled by advanced "target deconvolution" platforms. These technologies aim to identify the direct protein targets responsible for the observed phenotypes [101]. The integration of these platforms is resolving the initial ambiguity, revealing CEL not as a mysterious panacea, but as a rational, multi-target therapeutic agent. Its validation story, therefore, provides a compelling case study on how modern target-discovery strategies are essential for translating the complex pharmacology of natural products into actionable development pathways [100].
Platform Performance Comparison: Identifying CEL's Targets
The elucidation of CEL's polypharmacology has been achieved through a suite of complementary experimental platforms. Each platform operates on different principles, offering unique advantages and limitations in sensitivity, throughput, and biological context. The following table compares the performance of key target-identification platforms in profiling CEL, based on experimental data from recent studies.
Table 1: Comparison of Target-Identification Platforms in Profiling Celastrol
| Platform/Strategy | Core Principle | Key CEL Targets Identified | Typical Experimental Readout | Advantages | Limitations |
|---|---|---|---|---|---|
| Chemical Proteomics (ABPP) [98] [101] | Uses a chemical probe (e.g., biotin-labeled CEL) to covalently capture and enrich interacting proteins from a complex lysate. | PRDX1/2/4/6, HMGB1, HSP90β, PKM2, CAND1, COMT [98] | Pull-down assay followed by LC-MS/MS; validation via CETSA, SPR [98]. | Direct identification of covalent binding partners; can probe native proteome. | Requires probe synthesis which may alter bioactivity; biased toward covalent binders. |
| Protein Microarray [98] [101] | High-throughput screening of compound binding against thousands of immobilized recombinant proteins. | PRDX2 (in gastric cancer) [101] | Fluorescence-based detection of binding events on the chip. | Ultra-high throughput; uses purified proteins. | Lacks cellular context; post-translational modifications may be absent. |
| Thermal Proteome Profiling (TPP) [101] | Measures protein thermal stability shifts in intact cells upon compound treatment using mass spectrometry. | Identifies targets based on ligand-induced stabilization or destabilization. | Quantitative MS of soluble proteins after heat denaturation. | Label-free; works in live cells; can detect both stabilising and destabilising binders. | Complex data analysis; lower throughput; may miss low-abundance targets. |
| Network Pharmacology & Bioinformatics [98] | Computational integration of omics data, literature mining, and pathway analysis to predict targets and mechanisms. | Predicts key nodes like STAT3, NF-κB, AMPK pathways [98] [99]. | Network interaction maps, gene ontology enrichment. | Hypothesis-generating; leverages existing big data; low cost. | Predictions require experimental validation; indirect evidence. |
| Functional Phenotypic Screening + Omics [99] [102] | Measures phenotypic response (e.g., cell death, cytokine secretion), then uses transcriptomics/proteomics to infer upstream targets. | Links CEL's anti-cancer effect to YAP/VEGF pathway downregulation [102]; connects anti-obesity effect to AMPK/NF-κB [99]. | Cell viability, ELISA, Western blot, RNA-Seq. | Preserves biological context and functional relevance. | Causality is indirect; observed changes may be downstream effects. |
| Cellular Thermal Shift Assay (CETSA) [98] | A subset of TPP; validates target engagement by measuring protein aggregation upon heating in cell or tissue lysates with/without compound. | Used to validate engagement of HMGB1, PKM2, HSP90, etc., identified by other methods [98]. | Immunoblot or MS analysis of remaining soluble target protein. | Simple validation tool; works with endogenous proteins in lysates or cells. | Not a discovery tool; requires a priori target hypothesis and specific antibody. |
Key Performance Insight: No single platform is sufficient. For example, Chemical Proteomics directly identified CEL's covalent modification of Cys106 on HMGB1, a key mechanism for its anti-sepsis and neuroprotective effects [98]. Conversely, phenotypic screening in cancer models revealed that CEL potently inhibits the YAP/VEGF immunosuppressive axis, an effect later leveraged in a nanocomplex for "painless" tumor immunotherapy [102]. This complementary use of platforms transforms CEL from a phenotypic hit into a mechanistically mapped development candidate.
Comparative Efficacy: CEL vs. Alternative Therapeutic Strategies
CEL's multi-target profile suggests potential advantages over single-target agents, particularly for complex diseases like cancer and chronic inflammation. The following table compares the efficacy and mechanistic rationale of CEL with other standard or emerging therapeutic approaches, based on head-to-head experimental data or well-established mechanisms.
Table 2: Efficacy Comparison of Celastrol with Alternative Therapeutic Modalities
| Disease Context | Celastrol (Multi-Target) | Single-Target Agent (Example) | Combination Therapy (Example) | Key Comparative Data & Insight |
|---|---|---|---|---|
| Cancer (e.g., Breast Cancer) | Mechanism: Induces immunogenic cell death (ICD) via HMGB1/ATP release; inhibits YAP to reduce VEGF and immunosuppression [102].Efficacy (In Vivo): CEL-loaded nanocomplex (PM-CEL) significantly inhibited 4T1 tumor growth and reduced pain-related behavior [102]. | Mechanism: Anti-VEGF monoclonal antibody (e.g., Bevacizumab) inhibits angiogenesis.Efficacy: Reduces tumor growth but can induce resistance and does not directly alleviate pain. | Mechanism: Chemotherapy (e.g., Doxorubicin) + Immune Checkpoint Inhibitor (e.g., anti-PD-1).Efficacy: Potent but often with severe systemic toxicity and immune-related adverse events. | Insight: CEL's unique dual action (anti-tumor + analgesic via YAP/VEGF axis) offers a integrated therapeutic benefit not found in single-target angiogenesis inhibitors [102]. Its multi-target profile may mimic a "built-in" combination therapy, potentially lowering resistance risk. |
| Metabolic Disease (Obesity/Diabetes) | Mechanism: Simultaneously inhibits pro-inflammatory NF-κB pathway and activates metabolic sensor AMPK, improving insulin sensitivity and reducing adipogenesis [99]. | Mechanism: PPARγ agonist (e.g., Thiazolidinediones) improves insulin sensitivity.Efficacy: Effective but linked to weight gain, edema, and cardiovascular risk. | Mechanism: Metformin (AMPK activator) + anti-inflammatory drug (e.g., Salsalate).Efficacy: Addresses multiple facets but involves polypharmacy with increased side effect burden. | Insight: CEL inherently combines the beneficial actions of two drug classes (AMPK activator + anti-inflammatory), addressing both metabolic dysfunction and chronic low-grade inflammation, a core feature of obesity [99]. |
| Inflammatory Disease (e.g., Arthritis) | Mechanism: Covalently modifies multiple redox proteins (PRDXs) and inhibits HSP90, leading to broad suppression of inflammatory mediators [98]. | Mechanism: TNF-α inhibitor (e.g., Adalimumab).Efficacy: High efficacy but non-responsive in a significant subset of patients; risk of infections. | Mechanism: Methotrexate + TNF-α inhibitor.Efficacy: Standard of care for severe cases, but toxicity and monitoring requirements are high. | Insight: By targeting upstream, convergent nodes like PRDXs and HSP90, CEL may modulate multiple inflammatory pathways (TNF-α, IL-1β, IL-6) simultaneously, potentially benefiting patients unresponsive to single-cytokine blockade [98]. |
| Antifungal Therapy [103] | Mechanism: (Emerging) While not a primary antifungal, its anti-inflammatory and host-immunomodulatory properties could be adjunctive. | Mechanism: Azoles (e.g., Fluconazole) inhibit fungal ergosterol synthesis.Limitation: Narrow spectrum, drug-drug interactions, and rising resistance. | Mechanism: Echinocandin + Azole.Efficacy: Broad spectrum but remains vulnerable to multidrug resistance development. | Insight: In the context of difficult-to-treat fungal infections, CEL's value may lie not in direct antifungal activity but as an anti-virulence or host-directed therapy adjunct, similar to novel strategies targeting quorum sensing or biofilm formation [103] [104]. |
Detailed Experimental Protocols for Key Validation Platforms
Activity-Based Protein Profiling (ABPP) for CEL Target Fishing
This protocol is used to identify proteins that form covalent bonds with CEL [98] [101].
- Probe Synthesis: Synthesize a biotin- or alkyne-tagged CEL derivative. The tag is typically attached via a flexible linker to a chemically amenable site on CEL (e.g., at a hydroxyl group) to minimize interference with its bioactivity.
- Cell Lysate Preparation: Treat cultured cells (e.g., RAW264.7 macrophages for sepsis models) with CEL or vehicle. Lyse cells in a non-denaturing buffer to preserve native protein structures.
- Affinity Enrichment: Incubate the cell lysate with the CEL-probe. For alkyne probes, perform a "click chemistry" reaction (CuAAC) with azide-biotin. Pass the mixture through streptavidin-coated beads to capture probe-bound protein complexes.
- Stringent Washing: Wash beads extensively with high-salt and detergent buffers to remove non-specifically bound proteins.
- Protein Elution & Identification: Elute bound proteins by boiling in SDS-PAGE buffer or via competitive displacement with excess free CEL. Subject eluates to tryptic digestion and analysis by Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS). Identify proteins by searching fragment spectra against a protein database.
- Validation: Confirm identified targets through orthogonal methods:
- Cellular Thermal Shift Assay (CETSA): Treat cells with CEL, heat-denature cell lysates at a gradient of temperatures, and measure the stabilization of the candidate target protein via immunoblot [98].
- Surface Plasmon Resonance (SPR): Measure the binding kinetics (KD) of CEL to the purified recombinant target protein [98].
Thermal Proteome Profiling (TPP) for Label-Free Target Discovery
This protocol detects target engagement based on ligand-induced changes in protein thermal stability across the entire proteome [101].
- Cell Treatment & Heating: Divide a suspension of live cells (e.g., HCT116 colon cancer cells) into multiple aliquots. Treat one set with CEL and another with vehicle (DMSO) for a sufficient time to allow binding.
- Heat Denaturation: For each sample, create a series of tubes heated at different temperatures (e.g., from 37°C to 67°C) for a fixed time (e.g., 3 minutes).
- Soluble Protein Harvest: Lyse heated cells and separate the soluble (non-denatured) protein fraction from the aggregated protein by high-speed centrifugation.
- Proteomic Preparation & Quantification: Digest the soluble proteins from each temperature point with trypsin. Label the peptide samples from the CEL-treated and vehicle-treated groups with different isobaric tandem mass tags (TMT).
- Mass Spectrometry Analysis: Pool the labeled samples and analyze by LC-MS/MS. Quantify the abundance of each protein in each temperature channel.
- Data Analysis: For each protein, plot the melting curve (soluble protein fraction vs. temperature). A significant shift in the melting curve (ΔTm) between the CEL-treated and vehicle groups indicates direct target engagement or a proximal functional effect.
Functional Phenotypic Screening with Mechanistic Deconvolution
This protocol connects a phenotypic readout to molecular mechanisms, as used in CEL's cancer immunotherapy study [102].
- Nanocomplex Synthesis (PM-CEL): Prepare polydopamine/bovine serum albumin/manganese dioxide composite nanoparticles (PM) by oxidizing dopamine with KMnO4 in the presence of BSA. Load CEL into the hydrophobic cavities of the nanoparticles via stirring to form PM-CEL [102].
- In Vitro Phenotypic Assays:
- Cytotoxicity (MTT assay): Treat 4T1 breast cancer cells with PM-CEL, free CEL, or empty NPs. Measure cell viability after 24-48 hours.
- Immunogenic Cell Death (ICD) Assessment: Measure hallmarks of ICD:
- Surface Calreticulin (CRT): Detect via immunofluorescence staining and confocal microscopy.
- ATP Release: Quantify using a chemiluminescence assay.
- HMGB1 Release: Measure in cell supernatant via ELISA [102].
- Mechanistic Probe (Western Blot): Lyse treated cells and analyze by immunoblotting to quantify changes in key pathway proteins (e.g., YAP, VEGF, cleaved caspase-3).
- In Vivo Validation:
- Establish a syngeneic 4T1 tumor model in mice.
- Intratumorally inject PM-CEL or controls.
- Monitor tumor volume and measure mechanical allodynia (pain) using von Frey filaments.
- Harvest tumors for immunohistochemical analysis of CD8+ T cell infiltration, YAP, and VEGF expression [102].
Visualization of Signaling Pathways and Experimental Workflows
Diagram 1: CEL's Multi-Target Signaling Network (760px max)
Diagram 2: Phenotypic to Target-Based Validation Workflow (760px max)
The Scientist's Toolkit: Research Reagent Solutions
The following table details essential reagents and materials used in the experimental platforms discussed for CEL research [98] [102] [101].
Table 3: Essential Research Reagents for Celastrol Target Identification & Validation
| Reagent/Material | Supplier Examples | Function in CEL Research | Key Application Protocol |
|---|---|---|---|
| Biotin-PEG-NHS Ester | Thermo Fisher, Sigma-Aldrich | Used to synthesize biotin-labeled CEL probes for affinity enrichment in chemical proteomics (ABPP). | Covalently links biotin to a reactive group (e.g., -OH) on a CEL derivative via a polyethylene glycol (PEG) linker [101]. |
| Streptavidin Magnetic Beads | Pierce, Dynabeads | Solid-phase support for capturing biotinylated CEL-protein complexes from cell lysates during pull-down assays. | Essential for the enrichment step in ABPP to isolate potential targets before MS analysis [98] [101]. |
| Isobaric Tandem Mass Tags (TMT) | Thermo Fisher Scientific | Multiplexed protein quantification in Thermal Proteome Profiling (TPP). Allows comparison of protein solubility across multiple temperatures and conditions in a single MS run. | Used to label peptides from soluble fractions of heat-treated, CEL-exposed cells for quantitative proteomics [101]. |
| Recombinant Human Proteins/Protein Microarrays | CDI Laboratories, Thermo Fisher (ProtoArray) | Contains thousands of immobilized proteins for high-throughput screening of direct CEL binding. | Used to identify CEL's interaction with specific targets like PRDX2 in a non-cellular context [101]. |
| Anti-HMGB1, Anti-YAP, Anti-p-AMPK Antibodies | Cell Signaling Technology, Abcam | Validate target engagement (CETSA) and measure downstream pathway modulation (Western blot, immunofluorescence). | Critical for orthogonal validation of targets identified by MS and for elucidating functional consequences (e.g., YAP downregulation by CEL) [98] [102]. |
| Polydopamine Precursors & BSA | Sigma-Aldrich | Components for synthesizing the nanocomplex drug delivery platform (PM) to improve CEL's solubility and bioavailability. | Used in the formulation of PDA-BSA-MnO2-CEL (PM-CEL) for in vivo efficacy and mechanism studies [102]. |
| Cellular Thermal Shift Assay (CETSA) Kit | Cayman Chemical, proprietary protocols | Standardized reagents and protocols for measuring drug-induced thermal stabilization of target proteins. | Used to confirm CEL's direct binding to suspected targets like PKM2 or HMGB1 in cell lysates or intact cells [98]. |
The transition from an assay readout to a viable clinical candidate represents one of the most formidable challenges in drug discovery. This journey is fraught with attrition, where promising in vitro activity frequently fails to predict therapeutic efficacy and safety in humans. The selection of the primary screening strategy—target-based versus phenotypic—fundamentally shapes this translational pathway, a decision of particular consequence in natural products (NP) research. Target-based assays offer precision and mechanistic clarity by focusing on a predefined molecular target, while phenotypic assays, which measure holistic changes in cells or organisms, may better capture complex biological responses and serendipitously reveal novel mechanisms of action (MOA) [4]. This guide provides a comparative analysis of these strategies, evaluating their predictive value for clinical translation through the lens of experimental performance, validation frameworks, and application within the complex chemical space of natural products.
Comparative Analysis of Assay Strategy Performance
The predictive validity of an assay strategy is best quantified by its sensitivity (ability to correctly identify true hits), specificity (ability to reject false positives), and the resultant positive predictive value (PPV) (probability that a positive result is a true positive) [105] [106]. The table below synthesizes data from recent studies to compare the performance profiles of different assay approaches relevant to NP research.
Table 1: Performance Characteristics of Key Assay Strategies
| Assay Strategy | Typical Sensitivity | Typical Specificity | Key Strengths | Major Limitations | Best Application in NP Pipeline |
|---|---|---|---|---|---|
| Target-Based Biochemical | High (for target engagement) | Moderate to High | High mechanistic clarity, quantitative, amenable to HTS. | Misses compounds requiring cellular metabolism or acting on unknown targets. Low biological context. | Secondary validation of target engagement following phenotypic hit. |
| Imaging-Based High-Content Phenotypic [4] | High (for multifaceted perturbations) | High (with multi-parameter analysis) | Captures complex phenotypes, predicts MOA/toxicity, enables cell-based SAR. | Technically complex, data-intensive, requires expert interpretation. | Primary screening and mechanistic deconvolution of NP libraries. |
| Genomic (e.g., GRO-cap for eRNA) [107] | Highest for unstable transcripts (e.g., eRNAs) | High with specific tools (e.g., PINTS) | Unbiased detection of active regulatory elements, high resolution. | Specialized, primarily for modulating transcription/epigenetics. | Identifying NPs that modulate gene expression via enhancer/promoter activity. |
| Integrated Multi-Omics (e.g., NP-VIP) [8] | Very High (via orthogonal confirmation) | Very High (via orthogonal confirmation) | High-confidence target identification, reduces false positives, systems-level view. | Resource-intensive, complex data integration. | Late-stage lead optimization and target validation for NP leads. |
A critical insight is that no single metric is sufficient. For clinical translation, PPV is paramount, as it directly reflects the likelihood that a candidate selected from an assay will succeed in later stages [106]. However, PPV is heavily influenced by the prevalence of true actives in the screened library and the assay's inherent specificity [105]. Phenotypic strategies, by incorporating broader biological context, can achieve higher specificity against complex disease phenotypes, thereby potentially increasing PPV despite often being more resource-intensive than simple target-based screens [4].
Detailed Experimental Protocols
The predictive value of any strategy is contingent on rigorous, standardized experimental design. Below are detailed protocols for two pivotal approaches highlighted in the comparison.
This protocol is designed for the untargeted, multi-parameter profiling of NP-induced cellular effects.
1. Cell Preparation & Compound Treatment:
- Seed cells (e.g., U2OS) in multi-well imaging plates at an optimized density for 24-hour growth.
- Treat cells with natural product compounds or reference controls (e.g., from the LOPAC1280 library) for a defined period (typically 6-24 hours). Include a DMSO vehicle control.
2. Multiplexed Fluorescent Staining:
- Fix cells with paraformaldehyde (4%).
- Permeabilize with Triton X-100 (0.1%).
- Perform multiplexed immunofluorescence staining using a panel of antibodies and dyes targeting 14+ cellular markers. A core panel includes:
- Nuclear DNA (Hoechst): For cell count, nuclear morphology.
- Phospho-histone H3 (Ser10): Mitosis marker.
- Phospho-H2AX (Ser139): DNA damage response.
- RelA (p65): NF-κB pathway activation.
- α-Tubulin: Cytoskeleton integrity.
- Lysosomal-associated membrane protein 1 (LAMP1): Lysosome number/position.
- Wheat Germ Agglutinin (WGA): Plasma membrane morphology.
3. Image Acquisition & Analysis:
- Acquire high-resolution images using an automated high-content microscope (e.g., ImageXpress Micro) with a 20x or 40x objective.
- Use image analysis software (e.g., CellProfiler) to segment cells and nuclei and extract ~150 quantitative features per cell (morphology, intensity, texture).
- Analyze 500-1000 valid cells per treatment condition.
4. Data Processing & Cytological Profiling:
- Normalize feature values to the vehicle control.
- Reduce dimensionality to a set of ~20 interpretable "core features".
- Generate a cytological profile (CP)—a multidimensional fingerprint—for each compound.
- Use hierarchical clustering to compare NP CPs against a reference library of compounds with known MOA to predict putative targets or mechanisms [4].
This protocol employs an orthogonal strategy to deconvolute NP mechanisms, combining in silico, interactome, and phenotypic data.
1. Virtual Screening & Compound Library Preparation:
- Isolate and characterize compounds from a natural source (e.g., Salvia miltiorrhiza).
- Perform molecular docking of NP constituents against a target library relevant to the disease of interest (e.g., ischemic stroke targets).
2. Chemical Proteomics (Interaction):
- Prepare NP-derived chemical probes (e.g., with a biotin tag for affinity purification).
- Incubate probes with cell lysates or live cells.
- Capture probe-protein complexes using streptavidin beads.
- Wash, elute, and digest bound proteins.
- Identify and quantify captured proteins using liquid chromatography-tandem mass spectrometry (LC-MS/MS).
3. Metabolomics (Phenotype):
- Treat a relevant cell model with the NP extract.
- Quench metabolism and extract intracellular metabolites.
- Analyze metabolite profiles using techniques like GC-MS or LC-MS.
- Identify significantly altered metabolic pathways.
4. Data Integration & Target Validation:
- Integrate the three datasets: virtual screening hits (potential), chemical proteomic binds (direct interaction), and metabolomic perturbations (functional outcome).
- Intersect lists to identify high-confidence targets (e.g., PARP1, STAT3 in the case of S. miltiorrhiza) [8].
- Validate targets using orthogonal methods: siRNA knockdown, cellular thermal shift assay (CETSA), or reporter gene assays to confirm functional relevance to the observed phenotype.
Visualizing Assay Strategies and Workflows
The logical relationships and workflows of the discussed strategies can be visualized as follows.
Diagram: Parallel and Convergent Pathways in NP-Based Drug Discovery. This diagram illustrates two primary entry points for natural product research: a phenotypic screening pathway (green) that begins with complex cellular profiling, and an integrated deconvolution pathway (red) that combines orthogonal techniques for target identification. These pathways can converge, as phenotypic hits are funneled into target validation workflows.
Diagram: High-Content Phenotypic Screening Workflow. This linear workflow details the key experimental and computational steps in generating cytological profiles for mechanism-of-action prediction, from cell treatment and multiplexed staining to automated image analysis and data clustering.
The Scientist's Toolkit: Essential Research Reagent Solutions
The execution of the protocols above relies on specialized reagents, platforms, and tools. The following table details key solutions that enable high-predictivity assay development.
Table 2: Key Research Reagent Solutions for Predictive Assay Development
| Item / Solution | Category | Primary Function in Assay Development | Key Advantage for Translation |
|---|---|---|---|
| Simple Western (e.g., Bio-Techne) [108] | Automated Immunoassay Platform | Replaces traditional Western blot for protein quantification from complex samples (cell lysates, biopsies). | Provides robust, reproducible, quantitative data with minimal sample consumption, essential for preclinical and clinical study support under GCP/ISO standards [108]. |
| Validated Antibody Panels for HCS [4] | Cell Staining Reagents | Enable multiplexed, high-content imaging of specific cellular targets (e.g., pH3, γH2AX, LAMP1). | Allow simultaneous measurement of multiple disease-relevant pathways and toxicity markers, increasing the informational depth and predictive specificity of phenotypic screens. |
| LOPAC1280 or Similar Pharmacologically Active Library [4] | Reference Compound Collection | Serves as a benchmark set with known mechanisms of action for phenotypic screening. | Enables pattern-matching and predictive MOA classification of unknown NPs based on cytological profile similarity, bridging phenotypic readouts to target hypotheses. |
| Chemical Proteomics Probe Kits [8] | Target Identification Tools | Facilitate the creation of affinity probes from NP ligands to pull down interacting proteins from cell lysates. | Directly identifies physical protein-compound interactions within the native cellular environment, deconvoluting phenotypic hits into tangible molecular targets. |
| PINTS Computational Tool [107] | Bioinformatics Software | Identifies active promoters and enhancers from nascent transcriptomics data (e.g., GRO-cap). | Offers high sensitivity and specificity in detecting unstable eRNAs, providing a robust computational method to interpret complex genomic assay data for epigenetic drug discovery. |
| Statistical Design of Experiments (DoE) Software [109] | Assay Optimization Tool | Systematically varies multiple assay parameters (e.g., concentration, time, reagent volume) to find optimal conditions. | Uses empirical modeling to maximize assay robustness, signal-to-noise, and reproducibility early in development, reducing variability that can compromise predictive value later. |
The path from assay readout to clinical candidate is not a choice between target-based and phenotypic strategies but a strategic integration of both. Phenotypic assays, particularly high-content imaging, offer a powerful, unbiased entry point for natural products, capturing their complex bioactivity in a physiologically relevant context and providing a high-specificity filter for translational potential [4]. However, their predictive value is fully realized only when combined with subsequent deconvolution strategies, such as the integrated NP-VIP approach or advanced genomic techniques, which validate mechanisms and identify molecular targets [107] [8]. Furthermore, the adoption of quantitative, automated platforms (e.g., Simple Western) that meet clinical regulatory standards is critical for ensuring that early-stage assay data is robust and reproducible enough to inform costly late-stage development decisions [108]. Ultimately, a hybrid workflow—using phenotypic discovery to identify promising biological activity and target-based methods for mechanistic validation and optimization—represents the most predictive framework for translating the unique complexity of natural products into successful clinical candidates.
Conclusion
The strategic choice between target-based and phenotypic assays for natural product discovery is not binary but synergistic. The future lies in integrated workflows that leverage the unbiased, biologically rich discovery power of modern phenotypic screening—augmented by AI and multi-omics—coupled with rigorous, mechanistic target engagement and deconvolution validation. As exemplified by platforms combining generative chemistry with phenomics[citation:7], and by the multi-faceted target mapping of compounds like celastrol[citation:6], this convergence accelerates the translation of complex natural products into well-understood therapeutics. Future success will depend on embracing systems biology, developing more physiologically relevant complex disease models, and continuing to advance computational tools that bridge phenotypic observations with molecular mechanisms, ultimately de-risking the path from natural product to novel medicine[citation:3][citation:5].
References
- 1. Phenotypic vs. target-based drug discovery for first-in-class ... [https://pubmed.ncbi.nlm.nih.gov/23511784/]
- 2. Target Identification vs. Phenotypic Screening in Drug ... [https://pharmafeatures.com/strategic-dichotomy-target-identification-vs-phenotypic-screening-in-drug-discovery/]
- 3. Recent advances in target identification technology of ... [https://www.sciencedirect.com/science/article/abs/pii/S0163725825000452]
- 4. High-resolution phenotypic profiling of natural products ... [https://www.nature.com/articles/srep44472]
- 5. Target fishing: from “needle in haystack” to “precise guidance” [https://pmc.ncbi.nlm.nih.gov/articles/PMC12635624/]
- 6. Natural products in drug discovery: advances and ... [https://www.nature.com/articles/s41573-020-00114-z]
- 7. High Throughput Screening Assays for Drug Discovery [https://bellbrooklabs.com/high-throughput-screening-assays-drug-discovery/?srsltid=AfmBOoqzaLKNrz0ARSa5ofbbSSVKaS5nYiUHv4CB9v-4RwZzHcvRK2vF]
- 8. Natural product virtual-interact-phenotypic target ... [https://www.sciencedirect.com/science/article/pii/S2095177924001989]
- 9. Phenotypic Versus Target-Based Screening for Drug ... [https://www.technologynetworks.com/drug-discovery/articles/phenotypic-versus-target-based-screening-for-drug-discovery-300037]
- 10. Phenotypic Drug Discovery: Recent successes, lessons ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9708951/]
- 11. The Resurrection of Phenotypic Drug Discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7549108/]
- 12. Natural product virtual-interact-phenotypic target ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11830376/]
- 13. Target identification and validation of natural products with ... [https://www.sciencedirect.com/science/article/abs/pii/S0163725820302205]
- 14. Currently Available Strategies for Target Identification of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8515416/]
- 15. Predicting compound activity from phenotypic profiles and ... [https://www.nature.com/articles/s41467-023-37570-1]
- 16. Top 5 Drug Discovery Trends 2025 Driving Breakthroughs [https://www.pelagobio.com/cetsa-drug-discovery-resources/blog/drug-discovery-trends-2025/]
- 17. Phenotypic and targeted drug discovery in immune therapeutics [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra03914b]
- 18. Determining the mode of action of bioactive compounds [https://www.sciencedirect.com/science/article/abs/pii/S0968089611009151]
- 19. Polypharmacology: drug discovery for the future - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3809828/]
- 20. Expanding the Range of 'Druggable' Targets with Natural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2878877/]
- 21. Recent advances in screening active components from natural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7606101/]
- 22. Phenotype-Guided Natural Products Discovery Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7505087/]
- 23. Target identification of small molecules: an overview of the ... [https://bmcbiotechnol.biomedcentral.com/articles/10.1186/s12896-023-00815-4]
- 24. Review Hit Triage and Validation in Phenotypic Screening [https://www.sciencedirect.com/science/article/pii/S2451945620303317]
- 25. Phenotypic chemical biology for predicting safety and efficacy [https://www.sciencedirect.com/science/article/pii/S1740674917300033]
- 26. Image-based profiling for drug discovery: due for a machine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7754181/]
- 27. Cell Painting, a high-content image-based assay for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5223290/]
- 28. A Next Generation Connectivity Map: L1000 platform and the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5990023/]
- 29. AI-driven discovery of dual antiaging and anti-AD therapeutics ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12629202/]
- 30. Anomaly detection for high-content image-based ... [https://www.sciencedirect.com/science/article/pii/S2405471225002625]
- 31. A statistical framework for high-content phenotypic profiling ... [https://www.nature.com/articles/s42003-022-04343-3]
- 32. Morphology and gene expression profiling provide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10246468/]
- 33. Phenotypic and target-based HTS in drug discovery [https://lifechemicals.com/blog/computational-chemistry/476-phenotypic-and-target-based-hts-in-drug-discovery]
- 34. Target deconvolution techniques in modern phenotypic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3594516/]
- 35. Target deconvolution strategies in drug discovery [https://www.nature.com/articles/nrd2410]
- 36. From Phenotypic Hit to Chemical Probe - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC7730769/]
- 37. Lab Services for Target Deconvolution and Target Discovery [https://www.cdilabs.com/services/target-discovery]
- 38. Deep learning based deconvolution methods: A systematic ... [https://pubmed.ncbi.nlm.nih.gov/40586100/]
- 39. Applications of the Cellular Thermal Shift Assay to Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12072176/]
- 40. CETSA and thermal proteome profiling strategies for target ... [https://www.sciencedirect.com/science/article/abs/pii/S0944711323002234]
- 41. The future of phenotypic drug discovery [https://www.sciencedirect.com/science/article/pii/S2451945621000106]
- 42. Cellular Thermal Shift Assay-Enabled Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC10108388/]
- 43. A widely-applicable high-throughput cellular thermal shift ... [https://www.nature.com/articles/s41598-018-27834-y]
- 44. CETSA-MS-based target profiling of anti-aging natural ... [https://www.sciencedirect.com/science/article/abs/pii/S0223523424000837]
- 45. A robust CETSA data analysis automation workflow for ... [https://www.sciencedirect.com/science/article/pii/S2472555224000340]
- 46. Advancing Natural Product Drug Discovery with CETSA [https://www.pelagobio.com/cetsa-drug-discovery-resources/blog/natural-product/]
- 47. Combined Analysis of Phenotypic and Target-Based ... [https://www.sciencedirect.com/science/article/pii/S2472555222073737]
- 48. Artificial intelligence and multi-omics in drug discovery [https://www.sciencedirect.com/science/article/pii/S3051062725000112?dgcid=rss_sd_all]
- 49. AI-driven multi-omics integration in precision oncology [https://pmc.ncbi.nlm.nih.gov/articles/PMC12634751/]
- 50. Network-based multi-omics integrative analysis methods in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11954193/]
- 51. Advancing drug discovery through multiomics [https://www.ddw-online.com/advancing-drug-discovery-through-multiomics-37448-202510/]
- 52. Recent strategies and methodological advances for ... [https://pubmed.ncbi.nlm.nih.gov/41313478/]
- 53. Emerging paradigms for target discovery of traditional medicines [https://pmc.ncbi.nlm.nih.gov/articles/PMC11910885/]
- 54. A genome-wide pan-GPCR perspective [https://pubmed.ncbi.nlm.nih.gov/40098666/]
- 55. Genome-wide pan-GPCR cell libraries accelerate drug ... [https://www.sciencedirect.com/science/article/pii/S2211383524002545]
- 56. Comparative analysis of chemical similarity methods for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5559407/]
- 57. Workflows for Experimental Scientists [https://www.genedata.com/workflows/experimental-scientists]
- 58. Capturing single-cell heterogeneity via data fusion ... [https://www.nature.com/articles/s41467-019-10154-8]
- 59. Biologically Relevant Heterogeneity: Metrics and Practical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5464733/]
- 60. Inferring sparse structure in genotype–phenotype maps - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10471201/]
- 61. The computationally guided design of selective targeters ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12537719/]
- 62. Label-free target identification in drug discovery via ... [https://www.sciencedirect.com/science/article/abs/pii/S1367593118301716]
- 63. Creating and screening natural product libraries - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8494140/]
- 64. Development of a natural product optimization strategy for ... [https://www.nature.com/articles/s41467-024-49484-7]
- 65. A similarity-based target prediction tool for natural products [https://www.sciencedirect.com/science/article/abs/pii/S0010482524014367]
- 66. Optimizing drug design by merging generative AI with a ... [https://www.nature.com/articles/s42004-025-01635-7]
- 67. The Deconvolution Revolution: Strategies for Target ... [https://momentum.bio/content/the-deconvolution-revolution-strategies-for-target-identification/]
- 68. Accelerating target deconvolution for therapeutic antibody ... [https://www.nature.com/articles/s41467-021-21518-4]
- 69. 67 million natural product-like compound database ... [https://www.nature.com/articles/s41597-023-02207-x]
- 70. The Natural Products Atlas: An Open Access Knowledge ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6891855/]
- 71. Patient-Derived Organoid Biobanks for Translational ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12387782/]
- 72. Organoid models in oncology: advancing precision cancer ... [https://www.cancerbiomed.org/content/early/2025/07/24/j.issn.2095-3941.2025.0127]
- 73. Breakthroughs and challenges of organoid models for ... [https://www.nature.com/articles/s41420-025-02505-w]
- 74. A data-driven journey using results from target-based drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12062751/]
- 75. Review Artificial intelligence in natural products research [https://www.sciencedirect.com/science/article/pii/S1875536425609022?dgcid=rss_sd_all]
- 76. Artificial Intelligence in Natural Product Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC11874025/]
- 77. Comparative study of the mechanism of natural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10555731/]
- 78. Identify and mitigate bias in electronic phenotyping [https://www.sciencedirect.com/science/article/pii/S1532046424000893]
- 79. Empowering natural product science with AI - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2025/np/d4np00008k]
- 80. Improving target assessment in biomedical research [https://www.nature.com/articles/s41573-020-0087-3]
- 81. Comparative analysis of chemical similarity methods for ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-017-0234-y]
- 82. Natural bioactive products as promising therapeutics [https://www.sciencedirect.com/science/article/pii/S0254629922002551]
- 83. Knowledge-based mechanistic modeling accurately ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10390488/]
- 84. Contrast analysis for competing hypotheses: A tutorial using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12572084/]
- 85. Contrast Analysis - Sage Research Methods [https://methods.sagepub.com/ency/edvol/the-sage-encyclopedia-of-communication-research-methods/chpt/contrast-analysis]
- 86. Translational learning from clinical studies predicts drug ... [https://www.nature.com/articles/s41540-017-0012-5]
- 87. Target identification and mechanism of action in chemical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5543995/]
- 88. Bridging the gap between target-based and phenotypic- ... [https://www.semanticscholar.org/paper/a3c6783aa67820e2f9578b44bd091ff9312c5e5b]
- 89. The Role of Phenotypic Screening in Drug Discovery [https://www.technologynetworks.com/drug-discovery/articles/the-role-of-phenotypic-screening-in-drug-discovery-293422]
- 90. Human Cell-Based in vitro Phenotypic Profiling for Drug ... [https://www.frontiersin.org/journals/big-data/articles/10.3389/fdata.2019.00047/full]
- 91. Phenotypic Screening - an overview [https://www.sciencedirect.com/topics/medicine-and-dentistry/phenotypic-screening]
- 92. Proteome-Wide CETSA® Profiling Accelerates Drug ... [https://www.pelagobio.com/cetsa-drug-discovery-resources/blog/proteome-wide-cetsa-profiling/]
- 93. Current Advances in CETSA - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9218591/]
- 94. DARTS vs CETSA: Choosing the Right Assay for Target ... [https://www.creative-proteomics.com/resource/darts-vs-cetsa-choosing-the-right-assay-for-target-validation.htm]
- 95. Perspective on CETSA Literature: Toward More ... [https://www.sciencedirect.com/science/article/pii/S2472555222065340]
- 96. MS CETSA deep functional proteomics uncovers DNA ... [https://www.nature.com/articles/s41467-025-59505-8]
- 97. Large-scale plasma proteomics comparisons through ... [https://www.nature.com/articles/s41586-023-06563-x]
- 98. Emerging Target Discovery Strategies Drive the Decoding of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12380071/]
- 99. Targeting metabolic diseases with celastrol [https://pubmed.ncbi.nlm.nih.gov/40015541/]
- 100. Emerging Target Discovery Strategies Drive the Decoding ... [https://pubmed.ncbi.nlm.nih.gov/40873643/]
- 101. Target discovery-directed pharmacological mechanism ... [https://www.degruyterbrill.com/document/doi/10.1515/mr-2024-0076/html?lang=en&srsltid=AfmBOootLh0PVojs-7opmlvhUx8xVC4VUOmlK5UbkooRtRerz5tN7UKv]
- 102. Celastrol loaded nanocomplex for painless tumor therapy ... [https://www.nature.com/articles/s41598-025-97055-7]
- 103. New antifungal strategies and drug development against ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12507853/]
- 104. New Antimicrobial Strategies to Treat Multi-Drug Resistant ... [https://www.mdpi.com/2079-6382/13/1/71]
- 105. Understanding and using sensitivity, specificity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2636062/]
- 106. Predictive Value - an overview | ScienceDirect Topics [https://www.sciencedirect.com/topics/nursing-and-health-professions/predictive-value]
- 107. A comparison of experimental assays and analytical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9288987/]
- 108. Better assays: the key step in moving drugs from lab to clinic [https://www.drugtargetreview.com/article/186778/better-assays-the-key-step-in-moving-drugs-from-lab-to-clinic/]
- 109. Assay Optimization: A Statistical Design of Experiments ... [https://www.sciencedirect.com/science/article/pii/S1535553505004077]