Navigating Chemical Space: A Comparative Analysis of Natural Products, Approved Drugs, and Combinatorial Libraries
This article provides a comprehensive analysis for researchers and drug development professionals on the distinct and overlapping regions of chemical space occupied by natural products (NPs), approved drugs, and combinatorial...
Navigating Chemical Space: A Comparative Analysis of Natural Products, Approved Drugs, and Combinatorial Libraries
Abstract
This article provides a comprehensive analysis for researchers and drug development professionals on the distinct and overlapping regions of chemical space occupied by natural products (NPs), approved drugs, and combinatorial compounds. It explores foundational definitions and historical evolution, delves into modern computational methodologies for exploration and analysis, addresses key challenges in data and methodology, and presents a comparative validation of their structural diversity and biological relevance. The synthesis offers actionable insights for library design and future hybrid strategies in drug discovery.
Defining the Terrain: Foundational Concepts and Historical Evolution of Chemical Spaces
Conceptualizing Chemical Space and the Biologically Relevant Chemical Space (BioReCS) Framework
The concept of chemical space (CS) provides a fundamental framework for understanding and navigating the universe of all possible chemical compounds [1]. This multidimensional space is defined by molecular properties—both structural and functional—that serve as coordinates, positioning compounds based on their characteristics and relationships [1]. Within this vast theoretical universe lies the Biologically Relevant Chemical Space (BioReCS), the subset of molecules that interact with living systems, encompassing both beneficial and detrimental biological activities [1].
BioReCS spans numerous application domains including drug discovery, agrochemistry, flavor and odor science, food chemistry, and natural product research [1]. It includes not only therapeutic agents but also promiscuous compounds, poly-active molecules, and substances with toxic or allergenic effects [1]. The systematic exploration of this space is central to modern chemoinformatics and drug discovery, requiring specialized databases, molecular descriptors, and visualization techniques to map its complex topography [1] [2].
This comparison guide examines key regions of BioReCS—specifically natural products, combinatorial libraries, and approved drugs—within the context of a broader thesis on chemical space exploration. We provide objective performance comparisons, supporting experimental data, detailed methodologies, and essential resources to equip researchers with tools for effective navigation of biologically relevant chemical territories.
Quantitative Comparison of Chemical Subspaces
The exploration of BioReCS proceeds through distinct chemical subspaces (ChemSpas), each characterized by shared structural or functional features [1]. The following tables provide a quantitative foundation for comparing the key regions relevant to drug discovery.
Table 1: Representative Public Databases for BioReCS Exploration [1] [3]
| Type of Data Set / Area Covered | Exemplary Data Sets | Size Range (Number of Compounds) | Primary Utility in BioReCS Mapping |
|---|---|---|---|
| Drugs & Clinical Candidates | DrugBank, ChEMBL, ClinicalTrials.gov | ~4,500 approved (DrugBank) to ~2.4 million (ChEMBL) | Source of annotated bioactive molecules; defines "drug-like" subspace [3]. |
| Natural Products | COCONUT, NPASS | ~695,000 (COCONUT) to ~13,500 (NPASS with activity) | Covers evolved bioactive scaffolds; high structural diversity [3]. |
| Peptides | Peptipedia v2.0 | ~3.9 million sequences | Represents beyond Rule of 5 (bRo5) space; important for PPI modulation [3]. |
| Macrocycles | MacrolactoneDB | ~14,000 | Specialized class for challenging targets (e.g., PPIs, membrane proteins) [3]. |
| Food & Flavor Chemicals | FooDB, Flavor Molecule Compilations | >14,000 unique flavor molecules | Maps sensory BioReCS; intersection with nutraceuticals [1] [3]. |
| Toxic Chemicals | TOXNET, DSSTox | >35,000 chemical weapons | Defines "dark" BioReCS; crucial for safety prediction [3]. |
| Virtual Libraries (Synthetically Accessible) | Enamine REAL, GDB | Billions to 10^26 (proprietary spaces) | Represents vast unexplored synthetic regions of chemical space [4]. |
Table 2: Comparison of Natural Products, Combinatorial Compounds, and Approved Drugs [1] [5] [6]
| Property / Metric | Natural Products (NPs) & NP-Derived Drugs | Combinatorial & Synthetic Libraries | Approved Drugs (All Sources) |
|---|---|---|---|
| Chemical Space Coverage | Explore evolved, biologically pre-validated regions; high scaffold diversity. | Can target specific regions theoretically; bias towards synthetic feasibility. | Occupies a well-defined "drug-like" subspace within BioReCS. |
| Typical Molecular Complexity | Higher: More sp3 carbons, stereocenters, oxygen atoms; often macrocyclic [6]. | Lower: Designed for synthesis; often comply with Rule of 5. | Variable, but trend towards increased complexity for novel targets [6]. |
| Bioactivity Hit Rate | Historically high due to evolutionary selection. | Lower, but improving with DNA-encoded libraries and better design. | N/A (Endpoint). |
| Role in New Approvals (2014-2024) | 45 NP-derived NCEs approved (11.3% of all NCEs) [5]. | Primary source for synthetic NCEs (majority of small-molecule approvals). | 579 total drugs approved (388 NCEs, 191 NBEs) [5]. |
| Major Challenge | Supply, synthesis, and characterization [6]. | Achieving sufficient complexity and 3D shape diversity. | Optimizing multiple properties simultaneously (efficacy, safety, PK). |
| Key Discovery Method | Bioassay-guided isolation, genome mining, phenotypic screening [6]. | High-throughput screening (HTS), virtual screening (VS), combinatorial chemistry [4]. | Lead optimization from various starting points [4]. |
Table 3: Clinical Pipeline Analysis of Natural Product-Derived Compounds (Data up to 2025) [5]
| Category | Number Identified | Key Trend |
|---|---|---|
| NP-derived NCEs Approved (2014-Jun 2025) | 45 | Average of ~5 approvals per year; includes antibiotics, anticancer agents. |
| NP-Antibody Drug Conjugates (ADCs) Approved | 13 | Growing modality; uses NP toxins as warheads. |
| NP Compounds in Clinical Trials / Registration (End of 2024) | 125 | Demonstrates continued pipeline activity. |
| New NP Pharmacophores in Development | 33 | Indicates ongoing innovation, though only one discovered in the past 15 years. |
Experimental Protocols for BioReCS Navigation
High-Throughput Screening (HTS) of Compound Libraries
Objective: To experimentally probe regions of BioReCS by testing large physical libraries for activity against a therapeutic target. Protocol Summary:
- Library Curation: Select a diverse collection of 100,000 to several million compounds from corporate or commercial sources [4].
- Assay Development: Implement a robust biochemical or cell-based assay with a high signal-to-noise ratio, suitable for automation.
- Automated Screening: Utilize robotic liquid handlers and plate readers to test compounds at a single concentration (typically 10 µM) in microtiter plates.
- Hit Identification: Apply statistical thresholds (e.g., >3 standard deviations from mean) to identify initial "hits" from the primary screen.
- Hit Validation: Confirm activity of primary hits through dose-response experiments to generate IC50/EC50 values. Performance Consideration: While HTS directly tests physical-chemical space, it is constrained by library size (typically <5 million compounds), a mere fraction of the vast virtual chemical space estimated to contain up to 10^63 drug-like molecules [4].
Virtual Screening (VS) of Ultra-Large Chemical Spaces
Objective: To computationally search massively enlarged regions of chemical space (billions to trillions of virtual molecules) for potential hits. Protocol Summary:
- Target Preparation: Generate a 3D structure of the target protein, often from crystallography or homology modeling.
- Virtual Library Preparation: Access an on-demand database like Enamine REAL (containing billions of makeable compounds) or a proprietary chemical space [4].
- Molecular Docking: Use high-performance computing (e.g., thousands of CPU cores) to predict how each virtual molecule binds to the target. Advanced platforms like VirtualFlow can dock billions of compounds within weeks [4].
- Hit Selection: Rank compounds based on docking scores and visual inspection of predicted binding poses.
- Synthesis & Testing: Procure or synthesize the top-ranking virtual hits for experimental validation. Performance Data: A landmark study docked 281 million compounds from the ZINC database over a week using 500 CPU cores [4]. This approach can explore a chemical space orders of magnitude larger than HTS, accessing novel scaffolds outside traditional libraries.
Genome Mining for Natural Product Discovery
Objective: To explore the biosynthetic gene cluster (BGC) encoded region of BioReCS by predicting and engineering novel natural products. Protocol Summary:
- Genome Sequencing: Sequence the genome of a microbial strain (bacteria, fungi) or an environmental metagenomic sample [6].
- BGC Prediction: Use bioinformatics tools (e.g., antiSMASH, DeepBGC) to identify genomic regions encoding NP biosynthetic machinery [6].
- Priority Assessment: Predict the chemical structure of the putative NP and prioritize BGCs based on novelty and bioactivity potential.
- Cluster Activation/Heterologous Expression: Employ synthetic biology to "awaken" silent BGCs in a host organism for production [6].
- Compound Isolation & Characterization: Isolve the produced novel NP and determine its structure and biological activity. Performance Consideration: This method accesses the underexplored "dark matter" of microbial BioReCS, potentially yielding completely novel scaffolds with evolved bioactivity, but requires significant effort in genetic engineering and characterization [6].
Visualizing Chemical Space Relationships and Workflows
Diagram 1: Hierarchical Organization of Chemical Space and BioReCS
Diagram 2: Integrated Workflow for Exploring BioReCS
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Reagents and Materials for Chemical Space Research
| Item / Solution | Function in BioReCS Research | Example / Application |
|---|---|---|
| Curated Bioactivity Databases | Provide ground-truth data to map known regions of BioReCS and train AI/ML models. | ChEMBL: Annotated bioactive molecules for target-based exploration [1]. InertDB: Curated inactive molecules to define boundaries of BioReCS [1]. |
| Molecular Descriptors & Fingerprints | Translate chemical structures into numerical vectors for computational analysis and similarity searching. | Molecular Quantum Numbers (MQNs): 42 integer descriptors for universal chemical space mapping [2]. MAP4 Fingerprint: Works across small molecules to peptides [1]. |
| On-Demand Virtual Libraries | Provide access to synthetically tractable, ultra-large regions of chemical space for virtual screening. | Enamine REAL Space: Billions of makeable compounds for structure-based VS [4]. GDB Databases: Enumerated small molecules from first principles [2]. |
| Specialized Compound Libraries | Probe specific chemical subspaces with focused diversity. | Natural Product Libraries: Isolated or semi-synthetic NPs for phenotypic screening [6]. Macrocycle Libraries: For targeting PPIs and membrane proteins [1]. |
| Gene Cluster Prediction Software | Identifies biosynthetic potential in genomes to access novel NP chemical space. | antiSMASH: Predicts BGCs in microbial genomes [6]. DeepBGC: Uses deep learning for improved BGC prediction [6]. |
| Metabolomics Platforms | De-replicates known compounds and validates the production of novel NPs from activated BGCs. | LC-MS/MS with GNPS: Annotates NP structures by mass spectrometry networking [6]. |
| Color Palette Tools (for Visualization) | Ensures clarity, accessibility, and effective communication in chemical space visualizations. | SAMSON HCL Palette: Perceptually uniform color mapping for molecular attributes [7]. Color Deficiency Emulators: Check visualizations for colorblind accessibility [7] [8]. |
Thesis Context: Chemical Space and Drug Discovery
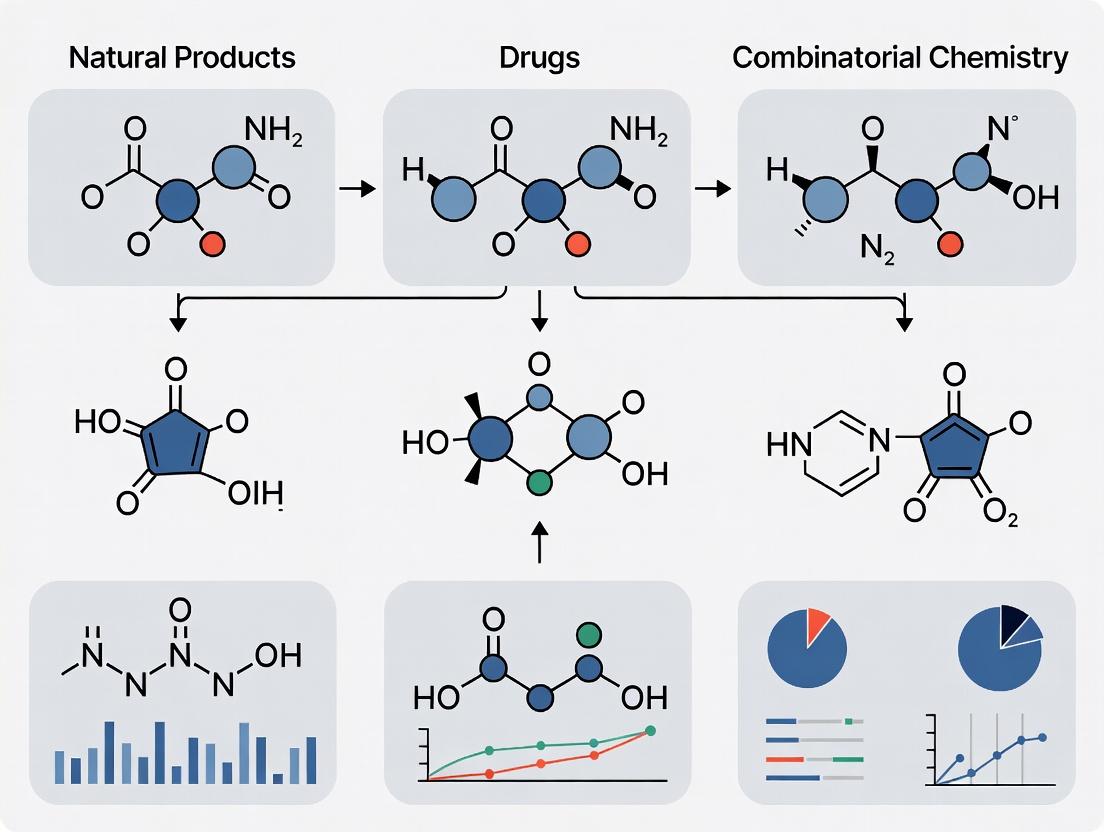
The exploration of chemical space—the theoretical universe of all possible organic molecules—remains a central challenge in drug discovery. This guide frames the comparison between natural products (NPs) and combinatorial/synthetic compound libraries within the broader thesis that these two sources occupy complementary and often non-overlapping regions of biologically relevant chemical space [9]. NPs are the result of evolutionary tuning over millions of years, yielding structures pre-validated for interactions with biological macromolecules [10]. In contrast, combinatorial chemistry offers rapid, exhaustive exploration of synthetic accessibility but may not consistently probe regions of chemical space with high biological relevance [10]. Modern strategies, including pseudo-natural product design and generative AI, seek to merge these paradigms, leveraging evolutionary wisdom to guide synthetic exploration toward novel, bioactive chemotypes [10] [11].
Comparative Performance Guide: Natural Products vs. Combinatorial Libraries
The following tables provide an objective, data-driven comparison of the performance, structural characteristics, and screening outcomes of NPs and combinatorial compounds.
Clinical Success and Molecular Characteristics
Table 1: Comparative Analysis of Clinical Output and Drug-Likeness
| Metric | Natural Products & NP-Derived Drugs | Combinatorial/Synthetic Libraries (Typical) | Data Source & Notes |
|---|---|---|---|
| New Chemical Entities (NCEs) Approved (2014-2024) | 44 (7.6% of all 579 approved drugs; 11.3% of NCEs) [5]. | Majority of small molecule NCEs. | Analysis of global drug approvals [5]. |
| Average Annual Approval Rate (2014-2025) | ~5 NP/NP-derived drugs per year [5]. | Variable; dominates annual NCE output. | Includes 45 NP/NP-D NCEs and 13 NP-antibody drug conjugates [5]. |
| Novel Pharmacophores in Pipeline (as of 2024) | 33 new pharmacophores in clinical development [5]. | Predominant source of novel scaffolds, but often less complex. | Only one new NP pharmacophore discovered in the past 15 years, highlighting a discovery gap [5]. |
| Typical Molecular Complexity | Higher fraction of sp³-hybridized carbons, more stereogenic centers, increased oxygenation [10] [6]. | Higher fraction of sp²-hybridized carbons, more aromatic rings, simpler stereochemistry. | Complexity is linked to evolutionary selection for specific bioactivity [10]. |
| Compliance with "Rule of Five" | Often non-compliant (higher MW, more H-bond donors/acceptors) [6]. | Designed for high compliance. | Despite non-compliance, many NPs show excellent oral bioavailability [6]. |
| Structural Uniqueness | Scaffolds often not represented in synthetic libraries; high density of functional groups [9]. | Scaffolds may be over-represented in corporate screening collections [9]. | Uniqueness underpins ability to hit "difficult" biological targets. |
Screening and Hit Identification Performance
Table 2: Comparison of Screening and Hit-Finding Efficiency
| Aspect | Natural Product Extracts/Libraries | Combinatorial/Diversity-Oriented Libraries | Supporting Experimental Data & Context |
|---|---|---|---|
| Hit Rate in Phenotypic Screens | Historically high; NPs account for a disproportionate number of first-in-class drugs [9]. | Often lower, but improved with better library design (e.g., fragment-based, NP-inspired) [10]. | High hit rate attributed to evolutionary pre-validation for bioactivity [10]. |
| Chemical Feasibility & Resupply | Major challenge: sourcing, total synthesis, or engineered production required [12]. | High: synthesis routes and building blocks are defined from the outset. | A key historical reason for pharma's shift away from NPs [12]. |
| Speed from Hit to Identified Compound | Slow: requires bioassay-guided fractionation and structure elucidation [12]. | Fast: compound structure is known immediately upon hit identification. | Technological advances (LC-MS/MS, metabolomics) are accelerating NP dereplication [6]. |
| Exploration of Chemical Space | Covers a deep but narrow region honed by evolution [10]. | Can explore broad, synthetically accessible regions, but may be biologically sparse [10]. | Pseudo-NP design aims to combine depth and breadth [10]. |
| Cost of Library Curation | High: collection, extraction, standardization [9] [12]. | Lower: based on automated, parallel synthesis. | Early combinatorial chemistry promised lower cost and unlimited size [9]. |
Chemical Space Analysis: Coverage and Overlap
Advanced cheminformatic methods enable the comparison of vast chemical spaces that cannot be fully enumerated [13].
Table 3: Comparison of Large, Defined Chemical Spaces
| Chemical Space / Library Type | Estimated Size | Design Principle & Coverage | Key Characteristic |
|---|---|---|---|
| Natural Product Space (defined by known NPs) | ~2,000 core fragment groups [10]. | Defined by biosynthetic pathways and evolutionary selection. | Biologically pre-validated but limited by evolutionary constraints [10]. |
| REAL Space (Enamine) | ~4 billion (10⁹) accessible compounds [13]. | Built from reliable reactions and in-stock building blocks; high synthesis success rate (>80%). | Focus on readily accessible and synthesizable molecules [13]. |
| KnowledgeSpace (Public) | Up to 10¹⁴ virtual compounds [13]. | Built from published reactions and commercial building blocks. | Large and diverse, but variable chemical feasibility [13]. |
| BICLAIM (Corporate) | >10²⁰ virtual products [13]. | Scaffold-centric, defined by deconstructing known products into cores and side chains. | Focus on scaffold exploration and novelty [13]. |
Key Finding from Comparative Analysis: A study comparing BICLAIM, REAL, and KnowledgeSpace using 100 drug-like query molecules found a remarkably low structural overlap. Only three compounds were found in the nearest-neighbor hit sets of all three spaces, demonstrating their complementarity [13]. This supports the thesis that NP space and high-quality synthetic spaces are likely non-redundant.
Diagram 1: Chemical Space Relationships (100 chars)
Experimental Protocols for Key Comparisons
Protocol: Phenotypic Screening of Pseudo-Natural Product Libraries
This protocol is used to evaluate novel pseudo-NP scaffolds designed to explore new regions of biologically relevant chemical space [10].
- Library Design & Synthesis: Deconstruct known NPs into fragment-sized pieces (MW 120-350 Da, AlogP < 3.5) [10]. Combine fragments from different biosynthetic origins in unprecedented connectivities (e.g., fused, spiro, bridged) via complexity-generating synthesis to create a pseudo-NP library [10].
- Cell-Based Phenotypic Assay: Treat target cells (e.g., reporter cell lines, primary immune cells) with compounds at relevant concentrations (e.g., 1-10 µM). Use unbiased, target-agnostic assays to probe broad biology [10].
- Examples: Monitor glucose uptake, autophagy flux, Wnt/Hedgehog signaling activity, T-cell differentiation markers, or induction of reactive oxygen species [10].
- Morphological Profiling (Cell Painting Assay): As a higher-content follow-up. Stain cells with fluorescent dyes for multiple organelles. Acquire high-content images and extract ~1,000 morphological features to generate a perturbation "fingerprint" for each active compound [10].
- Hit Validation & Target Identification: Confirm activity in dose-response. For promising hits, use chemoproteomics (e.g., affinity-based protein profiling), CRISPR-based genetic screens, or biophysical methods to identify the molecular target[s].
Protocol: Cheminformatic Comparison of Large Chemical Spaces
This protocol is used to assess the overlap and complementarity of virtual chemical spaces too large to enumerate [13].
- Query Panel Selection: Curate a panel of 100 reference molecules. For drug-relevant comparisons, filter approved small molecule drugs by standard drug-like properties (e.g., MW < 600, cLogP < 6) and select randomly [13].
- Nearest-Neighbor Search: For each query molecule, search each chemical space (e.g., BICLAIM, REAL) using a topological pharmacophore descriptor like Feature Trees (FTrees). Retrieve the 10,000 most similar molecules from each space without full enumeration [13].
- Overlap Analysis: Pool the unique hits from each space for all queries. Calculate pairwise structural overlaps using traditional fingerprints (e.g., MDL public keys, ECFP4) and Tanimoto similarity [13].
- Feasibility Scoring: Assess the synthetic feasibility of the retrieved hits using computational scores such as the Synthetic Accessibility score (SAscore) or retrosynthetic analysis tools [13].
Research Reagent Solutions: The Scientist's Toolkit
Table 4: Essential Reagents and Materials for NP/Combinatorial Comparative Research
| Reagent / Material | Function in Research | Key Application in Comparison Studies |
|---|---|---|
| Feature Trees (FTrees) Software [13] | A topological, pharmacophore-based molecular descriptor and search tool. | Enables similarity searching and comparison of non-enumerable fragment-based chemical spaces [13]. |
| Cell Painting Assay Kits [10] | A multiplexed fluorescent dye set for staining organelles (nucleus, ER, mitochondria, etc.). | Provides an unbiased phenotypic fingerprint to compare the bioactivity profiles of NP-derived vs. synthetic compounds [10]. |
| Validated Building Block Sets (e.g., for REAL Space) [13] | Curated collections of chemically diverse and synthetically reliable reagents. | Used to construct high-quality combinatorial libraries or pseudo-NP scaffolds with a high predicted synthesis success rate. |
| DNA-Encoded Library (DEL) Kits | Allows combinatorial synthesis where each compound is linked to a unique DNA barcode. | Facilitates the ultra-high-throughput screening (billions of compounds) of synthetic combinatorial spaces against purified protein targets. |
| LC-MS/MS and GNPS Platform [6] | Liquid chromatography-tandem mass spectrometry for compound separation, detection, and identification. | Critical for dereplicating natural product extracts (avoiding rediscovery) and characterizing novel pseudo-NPs [6]. |
Diagram 2: Integrated Drug Discovery Workflow (99 chars)
The comparative data underscore that natural products and combinatorial compounds are not mutually exclusive but are powerful complements. NPs provide evolutionarily refined starting points with high success rates in hitting novel biology, while combinatorial methods offer scalable exploration [9]. The future lies in integrative strategies—such as pseudo-NP design [10], biosynthetic engineering [6], and CSP-informed evolutionary algorithms [14]—that use computational tools to translate the lessons of evolutionary tuning into the efficient exploration of synthetically accessible chemical space. This synergy aims to generate novel, "beautiful" molecules that are both biologically relevant and pragmatically developable [11].
The pursuit of new therapeutic agents is a fundamental exploration of chemical space—the vast universe of all possible small organic molecules. Historically, this exploration has followed two parallel paths: the investigation of natural products (NPs) evolved by biology and the construction of combinatorial compound libraries synthesized by chemists. These two paradigms occupy complementary yet distinct regions of chemical space, a fact with profound implications for drug discovery success [15] [9].
The advent of combinatorial chemistry in the late 20th century promised a revolution: the ability to synthesize thousands to millions of compounds in parallel, creating an "explosion" of synthetic molecules for high-throughput screening (HTS) [16]. This shifted industry focus away from natural products, which were seen as difficult and costly to source and characterize [9]. However, the initial promise of combinatorial chemistry—that sheer volume would yield a plethora of new drugs—was not fully realized, leading to a critical reassessment of library design principles [17] [9].
Today, the field recognizes that quality and design trump sheer quantity. The modern thesis posits that the most effective drug discovery strategy lies not in choosing between natural or synthetic sources, but in intelligently integrating their strengths. This involves designing combinatorial libraries that capture the desirable, biologically relevant molecular features of natural products while leveraging synthetic efficiency and scalability [17] [18]. This comparison guide objectively examines the performance, design principles, and experimental approaches of combinatorial libraries relative to natural products, providing researchers with a framework for strategic chemical space exploration.
Comparative Analysis of Molecular Properties and Chemical Space
Combinatorial compounds and natural products differ systematically in their underlying structural and physicochemical properties. These differences directly influence their performance in biological screens, their "drug-likeness," and their success in progressing through development pipelines.
Key Property Distributions: A landmark comparative study analyzed the property distributions of drugs, natural products, and early-generation combinatorial compounds [18]. The findings reveal that combinatorial libraries often occupy a different, and sometimes narrower, region of chemical space than natural products and marketed drugs.
Table 1: Comparative Analysis of Molecular Properties Across Compound Classes [18]
| Molecular Property | Typical Combinatorial Compounds (Early Libraries) | Natural Products | Marketed Drugs |
|---|---|---|---|
| Average Molecular Weight | Lower (often <500 Da) | Higher | Intermediate |
| Number of Chiral Centers | Fewer (often 0 or 1) | More numerous | Intermediate |
| Aromatic Ring Count | Higher prevalence | Lower prevalence | Intermediate |
| Saturation (Fsp3) | Lower (more flat, aromatic) | Higher (more complex, 3D shapes) | Intermediate |
| Heteroatom Ratio (O, N, S) | Different patterns (e.g., more N) | Distinct, varied patterns | Balanced |
| Structural Complexity | Often simpler, more linear | High (complex ring systems, bridged cycles) | Variable, optimized for synthesis |
The data indicates that while drug molecules derive from both synthetic and natural sources, they often occupy a hybrid property space. Early combinatorial libraries, designed for synthetic ease, tended to be achiral, aromatic, and planar, lacking the stereochemical and scaffold complexity characteristic of many natural products [18]. This "complexity gap" may explain why some large combinatorial screens failed to produce high-quality leads, as the molecules did not sufficiently interrogate the biologically relevant regions of chemical space occupied by natural macromolecule-interacting ligands [9].
Chemical Space Coverage: Natural products are the result of billions of years of evolutionary selection for biological interaction. Consequently, they exhibit privileged scaffold architectures and pharmacophores that are pre-validated for binding to proteins and nucleic acids [15]. Combinatorial chemistry, in its modern, more sophisticated form, seeks to mimic this by designing libraries based on natural product-inspired scaffolds or by using computational methods to ensure library members populate desirable, "drug-like" regions of property space [17].
Library Design Principles: From Diversity-Oriented to Focused Synthesis
The philosophy of combinatorial library design has evolved significantly, moving from massive, diversity-driven collections to smaller, smarter, and more focused libraries.
The Evolution of Design Strategy: The initial paradigm of maximizing molecular diversity as the primary goal proved insufficient [17]. Contemporary design is a multi-objective optimization problem that balances synthetic feasibility, predicted Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) properties, and relevance to a biological target or target family [17].
Table 2: Evolution of Combinatorial Library Design Principles
| Design Paradigm | Primary Goal | Typical Library Size | Advantages | Limitations |
|---|---|---|---|---|
| Early Diversity-Oriented | Maximize structural diversity | Very Large (10⁵ - 10⁶) | Broad exploration of chemical space; many novel structures. | Often poor drug-likeness; high attrition; high cost of synthesis/screening. |
| Focused/Target-Family | Optimize binding to a specific target or protein family | Medium (10³ - 10⁴) | Higher hit rates; more relevant chemical space; incorporates known SAR. | Requires prior target/structure knowledge; limited serendipity. |
| Lead-Like/Drug-Like | Optimize physicochemical properties for developability | Medium (10³ - 10⁴) | Improved pharmacokinetic predictions; lower late-stage attrition. | May exclude valid chemotypes; relies on accuracy of predictive models. |
| Natural Product-Inspired | Mimic structural complexity & features of NPs | Variable | Biologically pre-validated scaffolds; novel yet relevant chemical space. | Synthetic challenge; complex chiral synthesis. |
| Dynamic Combinatorial (DCC) | Identify best binders via template-directed amplification | Small (10² - 10³) | Direct selection by biological target; thermodynamic optimization of binders [19]. | Requires compatible, reversible chemistry; analytical complexity. |
Modern Computational Design: Computational tools are now central to library design. They enable virtual screening of proposed libraries for ADMET properties, prediction of synthetic accessibility, and selection of building blocks to maximize desired diversity or similarity metrics [17]. This in-silico filtering helps ensure that synthesized libraries have a higher probability of containing viable lead compounds.
Dynamic Combinatorial Chemistry (DCC): DCC represents a powerful convergence of synthesis and screening. In DCC, libraries are formed under thermodynamic control using reversible chemical reactions (e.g., formation of acylhydrazones, imines, or disulfides) [19]. When a biological target (a protein or nucleic acid) is introduced, it acts as a template, selectively amplifying the library members that bind to it strongest, according to Le Chatelier's principle. This process directly identifies high-affinity ligands from a complex mixture, effectively performing synthesis and screening simultaneously [19].
Diagram: Workflow for Target-Directed Dynamic Combinatorial Chemistry (DCC). The process involves generating a library under thermodynamic control, introducing the biological target to amplify the best binders, and analyzing the shifted equilibrium to identify hits [19].
Experimental Protocols & Analytical Comparisons
Robust experimental and analytical methods are critical for both generating combinatorial libraries and comparing their outputs to natural product leads. Key protocols involve parallel synthesis, purification, and high-throughput characterization.
Representative Synthetic & Screening Protocol: Dynamic Combinatorial Library (DCL) Formation and Analysis This protocol, adapted from contemporary DCC practices, is used to generate and screen a library for binders to a protein target [19].
- Objective: To identify novel acylhydrazone-based inhibitors of a target enzyme (e.g., α-Glucosidase) from a dynamic combinatorial library.
- Materials:
- Building Blocks: A set of 5 acylhydrazides and 3 aldehydes, each solubilized in DMSO to create 100 mM stock solutions.
- Template: Purified target protein in a compatible aqueous buffer (e.g., PBS, pH ~6.5).
- Catalyst: Aniline (100 mM in buffer).
- Controls: Library without template; template alone.
- Procedure: a. DCL Assembly: In a 96-well plate, combine building blocks in buffer (final concentration 1-2 mM each) with 5 mM aniline catalyst. Final DMSO concentration ≤ 5%. b. Equilibration: Divide the master library mix. To the test sample, add the target protein (final concentration 1-5 µM). The control sample receives buffer only. Seal the plate and incubate at room temperature with gentle shaking for 48-72 hours to reach thermodynamic equilibrium. c. Quenching: Lower the pH of the solution to ~3.0 using a dilute acid (e.g., formic acid) to freeze the dynamic exchange by protonating the aniline catalyst. d. Analysis: Analyze both test and control samples via LC-MS (Liquid Chromatography-Mass Spectrometry). Use reverse-phase chromatography (C18 column) with a water/acetonitrile gradient.
- Data Analysis: Compare the LC-MS chromatograms and extracted ion counts for all possible acylhydrazone products between the test (+protein) and control (-protein) samples. Ligands amplified in the presence of the template will show a significant increase in peak area/height. Identify these hits by their mass.
- Validation: Independently synthesize the amplified hits and measure their binding affinity (e.g., IC50, Kd) using standard enzymatic or biophysical assays (e.g., Microscale Thermophoresis - MST) [19].
Analytical Method Comparison: HPLC vs. UPLC for Library Analysis The analysis of complex mixtures from combinatorial or natural product extracts demands high-resolution chromatography. Ultra-Performance Liquid Chromatography (UPLC) has largely superseded HPLC for this purpose.
Table 3: Performance Comparison of HPLC vs. UPLC for Compound Library Analysis [20]
| Parameter | High-Performance LC (HPLC) | Ultra-Performance LC (UPLC) | Implication for Library Analysis |
|---|---|---|---|
| Typical Particle Size | 3-5 μm | <2 μm | Smaller particles in UPLC reduce band broadening. |
| Operating Pressure | <6000 psi | Up to 15,000 psi | Higher pressure enables use of smaller particles. |
| Theoretical Plates | Lower | ≥2x Higher | Greatly improved resolution of complex mixtures. |
| Analysis Time | Longer (10-60 min) | ~3-5x Faster (2-10 min) | Higher throughput for screening fractions or purity checks. |
| Mobile Phase Consumption | Higher | ≥80% Reduction [20] | Lower cost and environmental impact (Green Chemistry). |
| Peak Capacity | Lower | Higher | Can separate more components in a single run, crucial for complex natural product extracts or DCLs. |
A specific comparative study demonstrated that for gradient separations of active pharmaceutical ingredients (APIs) and intermediates, UPLC methods provided equivalent or superior resolution while saving over 80% of mobile phase solvent compared to HPLC methods [20].
The Scientist's Toolkit: Essential Reagents & Materials
Successful execution of combinatorial and comparative natural product research requires specialized reagents, materials, and instrumentation.
Table 4: Key Research Reagent Solutions & Materials
| Category | Item | Typical Function & Application | Key Consideration |
|---|---|---|---|
| Library Synthesis | Diverse Building Blocks (e.g., amino acids, carboxylic acids, boronic acids, aldehydes, acylhydrazides). | Provide structural variation in combinatorial libraries. Sourced from commercial "large stock" collections. | Chemical diversity, purity, compatibility with chosen reaction chemistry. |
| Library Synthesis | Solid Supports (e.g., polystyrene resins, functionalized PEG). | Enable solid-phase parallel synthesis; excess reagents drive reactions; simplifies purification. | Swelling properties, loading capacity, linker chemistry for cleavage. |
| Dynamic Chemistry | Reversible Reaction Components (e.g., aniline, p-anisidine, nucleophilic catalysts). | Catalyze the reversible formation of imines, acylhydrazones, etc., in DCC for library equilibration [19]. | Biocompatibility (aqueous buffer, mild pH), catalytic efficiency. |
| Analytical | UPLC/HPLC Columns (e.g., C18 reverse-phase, sub-2 μm particles). | High-resolution separation of complex library mixtures or natural product extracts [20]. | Particle size, pressure rating, stationary phase chemistry for analyte retention. |
| Analytical | LC-MS & HRMS Systems | Primary tool for analyzing DCLs, purity checks, and identifying compounds in mixtures. Provides mass and fragmentation data. | Sensitivity, mass accuracy, compatibility with high-flow UPLC. |
| Screening | Validated Biological Targets (e.g., purified enzymes, protein domains, nucleic acid constructs). | Act as templates in DCC or targets in HTS for identifying bioactive library members [19]. | Stability under assay conditions, purity, relevance to disease pathway. |
| Natural Products | Metabolomics Tools (e.g., LC-MS with multivariate analysis software). | Profiling and comparing chemical feature diversity across natural product extracts to guide library building [21]. | Ability to detect a broad range of secondary metabolites. |
The rise of combinatorial chemistry has fundamentally transformed drug discovery from a linear, one-compound-at-a-time endeavor into a parallelized, systems-oriented science. However, its greatest lesson has been that synthetic explosion must be guided by intelligent design. The comparative analysis clearly shows that the most promising path forward is a hybrid one.
Future research will continue to blur the lines between natural and synthetic chemical space. This will be achieved through:
- Advanced Library Design: Increased use of AI and machine learning to design libraries that optimally populate the biologically relevant "middle earth" of chemical space between flat combinatorial compounds and highly complex natural products.
- Integration of Biosynthesis: Employing synthetic biology to create engineered natural product "libraries" via pathway refactoring and combinatorial biosynthesis.
- Broader DCC Applications: Expanding dynamic combinatorial and DNA-encoded library technologies to more challenging targets, including protein-protein interactions and RNA structures [19].
- Quantitative Natural Product Library Development: Implementing metabolomics-driven strategies, as demonstrated with fungal genera like Alternaria, to rationally build natural product libraries with maximized chemical diversity from a minimal set of isolates [21].
The ultimate goal is not to declare one approach the winner, but to develop a synergistic toolkit. By leveraging the synthetic power of combinatorial chemistry, the biologically validated inspiration of natural products, and the predictive power of computational design, researchers can more efficiently navigate the vastness of chemical space toward new and more effective therapeutics.
The concept of "chemical space" represents the total possible configuration of all organic molecules, estimated to exceed 10⁶⁰ compounds for small carbon-based molecules alone [22]. Within this vast universe, the subset of biologically relevant chemical space—where molecules interact with living systems—is the primary hunting ground for drug discovery. This guide provides a comparative analysis of three principal sources that populate this space: clinically approved drugs, natural products (NPs), and compounds from combinatorial chemistry.
Approved drugs represent a unique, pre-validated region of chemical space. Their passage through clinical trials confirms not only their efficacy against specific biological targets but also their adherence to critical pharmacokinetic and safety profiles in humans. Consequently, they serve as an indispensable benchmark for evaluating and mapping new chemical entities. Understanding how the chemical spaces of NPs and combinatorial libraries overlap with, or diverge from, this validated region is fundamental to designing more efficient discovery strategies. This comparison is framed within an ongoing paradigm shift: from serendipitous discovery and massive random screening toward rational, target-aware design informed by computational power and a deeper understanding of chemical biology [23] [24].
Comparative Analysis of Chemical Space Occupancy
The physicochemical and structural properties of molecules from different origins reveal distinct footprints within chemical space. Analysis using tools like ChemGPS-NP and Principal Component Analysis (PCA) allows for the visualization and comparison of these footprints [22].
Table 1: Comparative Physicochemical and Structural Profiles of Chemical Spaces
| Property / Characteristic | Approved Drugs (Benchmark) | Natural Products (NPs) | Combinatorial Compounds | Implication for Discovery |
|---|---|---|---|---|
| Primary Source | Synthetic, semi-synthetic, natural-derived | Biological organisms (plants, microbes, marine life) | Synthetic combinatorial libraries [25] | Defines starting diversity and novelty potential. |
| Molecular Complexity & Rigidity | Moderate complexity; balance of flexibility/rigidity | High complexity and structural rigidity; more stereocenters [22] | Often lower complexity; more flexible bonds [22] | NP rigidity favors selective target binding; combinatorial flexibility aids optimization. |
| Aromaticity | Moderate aromatic ring count | Lower aromaticity; more aliphatic and heterocyclic rings [22] | Higher aromaticity on average [22] | Impacts planarity, solubility, and protein interaction modes. |
| Compliance with "Rule of 5" (Ro5) | ~95% compliant for oral drugs [24] | ~60% compliant; many are bioavailable "beyond Ro5" [22] | Designed for high Ro5 compliance [24] | NPs access unique, "druggable" space beyond traditional rules. |
| Typical Molecular Weight | Optimized for oral bioavailability (often <500 Da) | Broader distribution; can be higher | Tightly controlled for library design | Influences membrane permeability and ADME properties. |
| Chemical Space Coverage | Defines the "clinically validated" region | Covers unique regions sparsely populated by synthetic libraries [22] | Often clusters in high-density regions around common scaffolds [26] | NPs can pioneer novel target interactions; combinatorial libraries may over-sample known areas. |
| Lead/Drug-Likeness | Inherently "drug-like" (post-validation) | High "lead-likeness"; pre-validated by evolution [22] | Varies; can be optimized for "drug-likeness" | NPs provide privileged starting points; combinatorial libraries require filtering. |
The data indicates that NPs occupy regions of chemical space distinct from typical synthetic medicinal chemistry compounds, including many combinatorial libraries. They exhibit greater structural rigidity, higher sp³ carbon count (greater three-dimensionality), and lower aromatic character [22]. Importantly, a significant portion of NPs violates Lipinski's Rule of Five while remaining pharmacologically active, demonstrating that the orally druggable chemical space extends beyond these classic guidelines [22]. This makes NPs invaluable for targeting challenging protein classes like protein-protein interactions.
Conversely, combinatorial chemistry, while capable of generating immense numbers of compounds, has faced criticism for producing libraries with limited structural diversity and a bias toward flat, aromatic structures that may not optimally interact with complex biological targets [23]. The modern trend has shifted from "larger is better" to designing smaller, focused, and smarter libraries based on known pharmacophores or target structural information [23] [27].
Benchmarking Performance: Computational and Experimental Metrics
Evaluating how well compounds from different sources perform in the drug discovery pipeline requires robust benchmarking. The Compound Activity benchmark for Real-world Applications (CARA) provides a framework for assessing computational activity prediction models by distinguishing between two key real-world tasks: Virtual Screening (VS) and Lead Optimization (LO) [26].
Table 2: Benchmarking Compound Libraries: A CARA Framework Perspective [26]
| Benchmarking Aspect | Virtual Screening (VS) Assay Context | Lead Optimization (LO) Assay Context | Implications for Library Strategy |
|---|---|---|---|
| Objective | Identify initial "hit" compounds from large, diverse libraries. | Optimize potency & properties of a congeneric series from a hit. | Guides library design for specific discovery phases. |
| Chemical Distribution | Diffused pattern: Compounds are structurally diverse with low pairwise similarity. | Aggregated pattern: Compounds are highly similar (congeneric). | VS requires broad, diverse libraries (e.g., diverse NP sets). LO requires focused, analog libraries. |
| Typical Library Source | Diverse NP extracts, large combinatorial libraries, commercial screening collections. | Focused combinatorial libraries, medicinal chemistry analog series. | Matches library diversity to the task. |
| Key Predictive Challenge | Identifying active scaffolds from vast chemical space ("needle in a haystack"). | Accurately ranking subtle potency changes from minor structural modifications. | VS models require good recall of actives; LO models require precise quantitative prediction. |
| Performance of Data-Driven Models | Meta-learning and multi-task learning strategies show effectiveness [26]. | Traditional single-assay QSAR models can perform decently [26]. | No single model excels at both tasks; strategy must be task-aware. |
This benchmarking reveals a critical insight: no single chemical library or computational model is optimal for all stages of discovery. Natural product libraries, with their broad, evolutionarily pre-validated diversity, are exceptionally well-suited for the Virtual Screening phase, where the goal is to identify novel chemical starting points [28]. In contrast, focused combinatorial libraries are indispensable for the Lead Optimization phase, where systematic, incremental structural changes are needed to refine potency and drug-like properties [23] [27].
Methodologies for Comparative Analysis and Validation
Core Experimental Protocols
To systematically compare and validate compounds from different chemical spaces against the approved drug benchmark, researchers employ several key methodologies.
Protocol 1: Adjusted Indirect Comparison for Efficacy Benchmarking This statistical method is used to compare the efficacy of two treatments (e.g., a new NP-derived candidate vs. an approved drug) when head-to-head trial data are unavailable but both have been tested against a common comparator (e.g., placebo or standard therapy) [29].
- Identify Studies: Locate two separate randomized controlled trials (RCTs). RCT A compares Drug X to Common Comparator C. RCT B compares Approved Drug Y to the same Common Comparator C.
- Extract Effect Estimates: For each trial, extract the relative treatment effect (e.g., mean difference, risk ratio, hazard ratio) of the experimental drug versus C, along with its variance (standard error²).
- Calculate Indirect Effect: The adjusted indirect comparison estimate for X vs. Y is the difference between the two direct effects: Effect(X vs. Y) = Effect(X vs. C) – Effect(Y vs. C).
- Calculate Variance: The variance of the indirect estimate is the sum of the variances of the two direct comparisons: Var(X vs. Y) = Var(X vs. C) + Var(Y vs. C). This results in a wider confidence interval, reflecting greater uncertainty [29].
- Interpretation: A result where the confidence interval for the indirect effect excludes the null value (e.g., 0 for mean difference, 1 for risk ratio) suggests a statistically significant difference between X and Y.
Protocol 2: Chemical Space Mapping with ChemGPS-NP This protocol maps and visualizes the position of compound collections within a global chemical space framework [22].
- Compound Set Preparation: Curate datasets (e.g., a list of approved drugs, an NP library, a combinatorial library) in SMILES or structure file format.
- Descriptor Calculation: For each compound, calculate a standard set of 35 molecular descriptors covering size, lipophilicity, polarity, polarizability, flexibility, and hydrogen-bonding capacity.
- PCA Score Prediction: Using the web-based ChemGPS-NP tool, project the descriptor values for the new compounds onto the existing principal component analysis (PCA) model. This model is built on a reference set that defines the chemical space map [22].
- Visualization & Analysis: Plot the compounds using the first few principal components (e.g., PC1 vs. PC2, PC3 vs. PC4). Analyze clusters, outliers, and density distributions. Regions densely populated by approved drugs define the "clinically validated" space. Sparsely populated regions by synthetic compounds but occupied by NPs indicate opportunity zones for novel discovery [22].
Protocol 3: In vitro Bioactivity and Selectivity Profiling This protocol benchmarks the biological performance of new hits against approved drugs.
- Panel Selection: Assemble a panel of related target proteins (e.g., a kinase family, GPCR subtypes) including the primary intended target.
- Dose-Response Assays: Test the new hit compound and a relevant approved drug in parallel across the panel at a range of concentrations (e.g., 0.1 nM – 100 µM) using standardized biochemical or cell-based assays.
- Data Analysis: Calculate IC₅₀ or EC₅₀ values for each compound/target pair. Generate a selectivity heatmap or radar chart.
- Benchmarking: Compare the potency (IC₅₀) and selectivity index (ratio of IC₅₀ for off-target vs. primary target) of the new hit to the approved drug. A new NP-derived hit may show comparable potency but a distinct selectivity profile, indicating a potentially differentiated therapeutic mechanism.
The Scientist's Toolkit: Essential Reagents & Platforms
Table 3: Key Research Reagents and Platforms for Chemical Space Exploration
| Tool / Reagent | Category | Primary Function in Benchmarking | Key Consideration |
|---|---|---|---|
| ChEMBL Database [26] | Bioactivity Database | Provides curated bioactivity data for approved drugs and millions of other compounds, enabling the extraction of assay data for indirect comparisons and model training. | Critical for defining benchmark activity values and understanding structure-activity relationships (SAR). |
| Cortellis Drug Discovery Intelligence [30] | Commercial Intelligence Platform | Integrates biological, chemical, and pharmacological data to benchmark experimental performance of drug candidates against historical and competitor data. | Used for assessing the competitive landscape and validating target-drug-disease linkages. |
| DNA-Encoded Library (DEL) Technology [25] | Combinatorial Library Platform | Enables the synthesis and affinity-based screening of ultra-large libraries (billions of compounds) to identify novel binders for a protein target. | Useful for rapidly exploring vast synthetic chemical space and generating hits for difficult targets. |
| High-Resolution Mass Spectrometry (HR-MS) & NMR [28] | Analytical Chemistry | Enables the dereplication (identification of known compounds) and structural elucidation of novel natural products, crucial for mapping NP space. | Essential for quality control and confirming the novelty of isolates from NP sources. |
| ChemGPS-NP Web Service [22] | Computational Chemistry Tool | Provides a publicly available platform for mapping and navigating the chemical space of large compound collections relative to a defined reference space. | The standard 35-descriptor set ensures consistent, comparable projections across studies. |
| Rule of 5 (Ro5) and PAINS Filters | Computational Filters | Initial filters to assess drug- or lead-likeness and flag compounds with substructures prone to assay interference. | While useful, they should not be used rigidly, especially for NPs which may be active beyond Ro5 [22]. |
Integrated Pathways and Strategic Workflows
The following diagrams illustrate the logical relationships between chemical sources, discovery strategies, and benchmarking outcomes.
Chemical Space Navigation to Clinical Validation
Decision Tree for Comparative Efficacy Analysis
Synthesis and Strategic Outlook
Mapping chemical space with approved drugs as the benchmark reveals a complementary relationship between NPs and combinatorial chemistry. Natural products serve as pioneering explorers, uncovering biologically relevant but synthetically underserved regions of chemical space. They provide privileged, evolutionarily refined scaffolds ideal for initial hit discovery, particularly for challenging targets. Combinatorial chemistry, guided by computational design, serves as the optimizing engineer, efficiently populating the regions around these hits to refine potency, selectivity, and drug-like properties toward the validated benchmark space [23] [27].
The future of effective chemical space navigation lies in integrating these paradigms. Strategies include:
- Biology-Inspired Combinatorial Synthesis: Using NP scaffolds as cores for generating combinatorial libraries to explore related chemical space more systematically [28].
- AI-Enabled De Novo Design: Using generative models trained on approved drugs and NP structures to propose novel compounds that inherently possess drug-like properties while exploring new regions [24] [27].
- Advanced Benchmarking Platforms: Utilizing comprehensive platforms like CARA [26] and Cortellis [30] to make data-driven decisions by continuously benchmarking new entities against the validated performance of approved drugs across multiple parameters.
The clinically validated chemical space defined by approved drugs is not a static endpoint but a dynamic, expanding frontier. By using it as a foundational benchmark, researchers can strategically direct the exploration of natural product diversity and the power of combinatorial synthesis to populate this frontier with the next generation of effective therapeutics.
Tools for Navigation: Computational and Analytical Methods for Chemical Space Exploration
The systematic representation of chemical structures is a cornerstone of modern computational drug discovery. Molecular descriptors and fingerprints translate the vast, multidimensional space of chemical structures into quantifiable data, enabling comparison, prediction, and navigation [31]. This capability is critical within the broader thesis of chemical space comparison, which seeks to understand the relationships and coverage differences between the rich, evolutionarily refined space of Natural Products (NPs), the vast, synthetically accessible realm of combinatorial compounds, and the focused libraries of drug-like molecules [5] [6].
Natural products are distinguished by high structural complexity, including more sp³-hybridized carbons and oxygen atoms, which often translate to potent and selective bioactivity [6]. Despite a historical decline in focus, NPs and NP-derived compounds accounted for 9.7% (56 of 579) of all new drug approvals between 2014 and 2024, underscoring their enduring relevance [5]. Conversely, combinatorial chemistry can generate libraries of unprecedented size, with proprietary collections like GSK's XXL space containing up to 10²⁶ virtual compounds [32]. Bridging these domains requires robust molecular representations that can capture essential structural and chiral features to enable meaningful comparison and identify complementary regions of chemical space for new therapeutic leads [31] [33].
Comparative Performance of Molecular Representations
Different molecular representations capture varying aspects of chemical structure, leading to significant differences in performance for predictive modeling tasks. The following tables summarize key experimental findings from benchmarking studies.
Table 1: Performance Benchmark of Fingerprints and Descriptors in Odor Prediction [34]
| Feature Set | Model | AUROC | AUPRC | Accuracy (%) | Precision (%) | Recall (%) |
|---|---|---|---|---|---|---|
| Morgan Fingerprints (ST) | XGBoost | 0.828 | 0.237 | 97.8 | 41.9 | 16.3 |
| Morgan Fingerprints (ST) | LightGBM | 0.810 | 0.228 | 97.7 | 39.5 | 17.4 |
| Morgan Fingerprints (ST) | Random Forest | 0.784 | 0.216 | 97.6 | 37.2 | 15.8 |
| Classical Descriptors (MD) | XGBoost | 0.802 | 0.200 | 97.6 | 36.1 | 15.1 |
| Functional Group (FG) | XGBoost | 0.753 | 0.088 | 97.0 | 22.3 | 9.8 |
Table Note: Benchmark on a dataset of 8,681 odorants. Results show Morgan (circular) fingerprints paired with a gradient-boosting algorithm (XGBoost) deliver superior performance for capturing complex structure-property relationships [34].
Table 2: Performance of Chirality-Sensitive Descriptors in Enantiomer Separation Prediction [33]
| Descriptor Type | Base Model | Chirality Enhancement | Prediction Accuracy (Elution Order) |
|---|---|---|---|
| Morgan Fingerprints | Random Forest | Integrated CIP labels | 0.82 |
| Latent Space Vector (Transformer) | Random Forest | Delta (ori-opp) | 0.75 |
| Latent Space Vector (CDDD) | Random Forest | Delta (ori-ns) | 0.71 |
| Latent Space Vector (Transformer) | Random Forest | Original (no enhancement) | 0.65 |
Table Note: Evaluation on a dataset of 1,929 enantiomer pairs for Chiralpak AD-H column. Classical fingerprints outperformed latent space vectors from SMILES encoders, but "delta" operations (arithmetic between molecule and enantiomer descriptors) significantly improved chiral encoding [33].
Table 3: Drug Approvals by Origin (2014-2024) and Representation Challenge [5]
| Compound Class | Number of Approvals | % of Total (579) | Key Representation Challenges |
|---|---|---|---|
| All NP-derived | 56 | 9.7% | High complexity, stereochemistry, polycyclic scaffolds |
| NP-derived New Chemical Entities | 44 | 7.6% | Capturing 3D conformation and pharmacophore geometry |
| NP Antibody-Drug Conjugates | 12 | 2.1% | Linker chemistry and payload-specific descriptors |
| Synthetic/Small Molecule | 523 | 90.3% | Focus on drug-likeness, lead-like property ranges |
Experimental Protocols for Benchmarking Representations
Protocol: Benchmarking Fingerprints and Descriptors for Property Prediction
This protocol is adapted from a large-scale comparative study of machine learning models for odor decoding [34].
Dataset Curation:
- Source: Assemble a multi-label dataset from curated public sources (e.g., Pyrfume-data archive).
- Standardization: Merge entries by PubChem CID. Retrieve canonical SMILES via PubChem's PUG-REST API.
- Label Consolidation: Standardize diverse property labels (e.g., odor descriptors) into a controlled vocabulary with expert guidance to minimize noise.
Feature Generation:
- Morgan Fingerprints: Generate using the RDKit library (radius=2, nBits=2048 is common). Consider using the Morgan algorithm from optimized MolBlock conformations [34].
- Classical 2D Descriptors: Calculate using RDKit or similar. Standard set includes Molecular Weight, LogP, Topological Polar Surface Area (TPSA), hydrogen bond donors/acceptors, and rotatable bond count [34].
- Functional Group Fingerprints: Generate by scanning SMILES against a predefined list of SMARTS patterns for key functional groups.
Model Training & Evaluation:
- Algorithm Selection: Benchmark tree-based algorithms (Random Forest, XGBoost, LightGBM) known for handling high-dimensional, sparse fingerprint data [34].
- Validation: Implement stratified k-fold cross-validation (e.g., 5-fold) on an 80/20 train/test split. For multi-label tasks, use a one-vs-rest strategy.
- Metrics: Report Area Under the ROC Curve (AUROC), Area Under the Precision-Recall Curve (AUPRC), accuracy, precision, and recall.
Protocol: Evaluating Chirality-Sensitive Descriptors
This protocol is based on a study evaluating descriptors for chiral chromatography prediction [33].
Chiral Data Preparation:
- Dataset: Obtain a set of enantiomer pairs with an associated chiral property (e.g., chromatographic elution order).
- Critical Splitting: Split data into training and test sets by enantiomer pair to prevent data leakage. Both enantiomers must reside in the same set.
Descriptor Calculation:
- Baseline Fingerprints: Generate Morgan fingerprints with chirality tags enabled (e.g., use
useChirality=Truein RDKit). - Latent Space Descriptors:
- Use a pre-trained SMILES encoder (e.g., Transformer, CDDD model) to generate a latent vector for each canonical SMILES.
- Create "delta" descriptors to enhance chiral information: calculate the vector difference between (a) original molecule and its enantiomer ("ori-opp"), or (b) original molecule and its stereochemistry-depleted SMILES ("ori-ns") [33].
- Baseline Fingerprints: Generate Morgan fingerprints with chirality tags enabled (e.g., use
Modeling & Analysis:
- Train a classifier (e.g., Random Forest) to predict the chiral property.
- Compare the performance of different descriptor sets. Analyze misclassifications to understand model and descriptor limitations.
Visualization of Workflows and Chemical Space
Molecular Representation to Chemical Space Analysis
Molecular Representation for Predictive Modeling
Table 4: Key Software Tools and Resources for Molecular Representation
| Tool/Resource Name | Type | Primary Function in Representation | Application Context |
|---|---|---|---|
| RDKit | Open-source Cheminformatics Library | Calculates molecular descriptors, generates Morgan fingerprints, handles SMILES I/O and stereochemistry. | Core toolkit for standard descriptor/fingerprint generation [34] [33]. |
| CDDD Model | Pre-trained Neural Network | Generates continuous latent space vector descriptors from SMILES strings. | Exploring novel, data-driven descriptors; transfer learning [33]. |
| GTM (Generative Topographic Mapping) | Dimensionality Reduction Algorithm | Creates interpretable 2D maps of chemical space from high-dimensional descriptors. | Visualizing and comparing libraries (e.g., NP vs. combinatorial) [31] [32]. |
| CoLiNN | Specialized Neural Network | Predicts chemical space projection for combinatorial products directly from building blocks, avoiding enumeration. | Ultra-large combinatorial library (e.g., DEL) analysis and design [32]. |
| PUG-REST API (PubChem) | Web API | Retrieves canonical SMILES and standardized compound data by identifier. | Essential for dataset curation and standardization [34]. |
| AntiSMASH/DeepBGC | Bioinformatics Platform | Identifies biosynthetic gene clusters (BGCs) in genomic data for NP discovery. | Genome mining for novel natural product scaffolds [6]. |
The systematic exploration of chemical space—a theoretical multi-dimensional space where each point represents a unique molecule defined by its properties—is foundational to modern drug discovery and cheminformatics [35]. With public repositories like ChEMBL and PubChem now containing millions of compounds and the emergence of ultra-large virtual libraries exceeding a billion molecules, the practical analysis of this space presents a monumental computational challenge [35] [36]. A core thesis in contemporary research interrogates whether the rapid growth in the number of available compounds translates to a corresponding increase in meaningful chemical diversity, particularly when comparing distinct regions such as natural products, approved drugs, and combinatorial synthetic compounds [35].
Traditional tools for assessing similarity and diversity, such as pairwise Tanimoto similarity calculations and classic clustering algorithms like Taylor-Butina, scale quadratically (O(N²)) with library size. This scaling makes them prohibitively expensive for analyzing today's massive datasets [37] [38]. This guide provides a comparative analysis of two innovative solutions to this bottleneck: the iSIM (instant similarity) framework and the BitBIRCH clustering algorithm. We objectively evaluate their performance against established alternatives, detailing experimental protocols and presenting data within the critical context of comparative chemical space research.
Methodological Foundations: iSIM and BitBIRCH Explained
The iSIM Framework: Linear-Scaling Similarity Assessment
The iSIM framework provides an exact or highly accurate approximation of the average pairwise similarity within a set of N molecules in linear time (O(N)), bypassing the need for N² comparisons [39].
Core Protocol: For a library represented by binary fingerprints (e.g., ECFP4, RDKit), molecules are arranged in an N×M matrix, where M is the fingerprint length. The key step is the column-wise sum, producing a vector K = [k₁, k₂, …, kₘ], where each kᵢ is the count of "on" bits in that column [39]. From this vector, the instant Tanimoto (iT) is calculated as:
iT = Σᵢ [kᵢ(kᵢ−1)] / Σᵢ [kᵢ(kᵢ−1) + kᵢ(N−kᵢ)] [35] [39].
This iT value represents the library's average internal similarity (lower values indicate greater diversity). The framework also introduces the concept of complementary similarity to identify molecules central to (medoids) or on the periphery of (outliers) the chemical space [35].
The BitBIRCH Algorithm: Efficient Hierarchical Clustering
BitBIRCH is a clustering algorithm designed for binary fingerprints that adapts the Balanced Iterative Reducing and Clustering using Hierarchies (BIRCH) approach for cheminformatics [37] [38].
Core Protocol: BitBIRCH constructs a CF-tree (Clustering Feature tree) using compact Bit Feature (BF) vectors to represent subclusters. A BF for a cluster j is defined as BFⱼ = [Nⱼ, lsⱼ, cⱼ, molsⱼ], where:
Nⱼ: Number of molecules in the cluster.lsⱼ: Linear sum vector of the fingerprints.cⱼ: Centroid of the cluster.molsⱼ: List of molecule indices [37] [38].
The lsⱼ vector, in conjunction with iSIM, allows for the efficient calculation of cluster radius and diameter using the Tanimoto metric as molecules are absorbed into leaf nodes of the tree. This structure enables single-pass clustering with O(N) time complexity [37].
Table 1: Core Technical Specifications of iSIM and BitBIRCH
| Feature | iSIM Framework | BitBIRCH Algorithm |
|---|---|---|
| Primary Function | Calculate average similarity/internal diversity of a set | Partition molecules into similarity-based clusters |
| Computational Scaling | O(N) with number of molecules (N) | O(N) with number of molecules (N) |
| Core Innovation | Column-wise fingerprint summation enabling n-ary comparison | Bit Feature (BF) vector & CF-tree for binary data |
| Key Metric Output | Instant Tanimoto (iT), Complementary Similarity | Cluster membership, centroids, and diameters |
| Representation Compatibility | Binary fingerprints, real-value descriptors (normalized) | Binary molecular fingerprints |
Diagram Title: iSIM Calculation Workflow for Library Diversity
Quantitative Performance Comparison with Alternative Tools
Computational Efficiency Benchmarks
The most significant advantage of iSIM and BitBIRCH is their transformative computational efficiency compared to traditional pairwise methods.
Experimental Protocol for Timing Benchmarks: Libraries of varying sizes (e.g., 50k to 1.5 million molecules) are prepared using standardized RDKit 2048-bit fingerprints [40]. For each library, the time to compute the average Tanimoto similarity is measured for iSIM versus the exhaustive pairwise method. Similarly, total clustering time is measured for BitBIRCH versus the standard RDKit implementation of Taylor-Butina clustering. Experiments are run on identical hardware (e.g., a single 10 GB compute node) [41] [40].
Table 2: Computational Performance Benchmark
| Library Size (Molecules) | Task | Traditional Method (Time) | iSIM / BitBIRCH (Time) | Speed-Up Factor | Source/Experimental Context |
|---|---|---|---|---|---|
| ~5,000 | Clustering | Taylor-Butina (RDKit): ~1.46 s | BitBIRCH: ~0.78 s | ~1.9x | OpenCADD dataset; user time measured [40]. |
| 1,500,000 | Clustering | Taylor-Butina (RDKit): Projected days-hours | BitBIRCH: Minutes | >1,000x | Theoretical projection based on O(N) vs. O(N²) scaling [37] [42]. |
| 1,000,000,000 | Clustering | Taylor-Butina: Impossible on standard hardware | BitBIRCH: ~5 hours | Not Applicable | Parallel/iterative BitBIRCH approximation on high-performance computing resources [42]. |
| Variable (N) | Avg. Similarity | Pairwise Tanimoto: O(N²) scaling | iSIM: O(N) scaling | Increases with N | Fundamental algorithmic scaling [39]. |
Clustering Quality and Outcome Analysis
Increased speed is meaningless if it compromises result quality. Studies compare clustering outcomes using internal validation metrics and structural analysis.
Experimental Protocol for Quality Assessment: A standardized library (e.g., ChEMBL33 natural products subset, n=64,086) is clustered using BitBIRCH and Taylor-Butina at a comparable Tanimoto threshold [41]. Quality is assessed using:
- Internal Validation Indices: Calinski-Harabasz (higher is better) and Davies-Bouldin (lower is better) indices [42].
- Structural Analysis: The number of unique Murcko scaffolds within generated clusters, measuring chemical diversity preservation [41].
- Visual Inspection: t-SNE visualization of chemical space colored by cluster assignment [41].
Table 3: Clustering Quality Comparison (ChEMBL33 Natural Products)
| Quality Metric | Taylor-Butina Clustering | Original BitBIRCH | BitBIRCH with Refinement (Prune+Diameter) | Interpretation |
|---|---|---|---|---|
| Number of Clusters | Baseline | Often fewer, with one very large cluster | More balanced cluster distribution | Refinement strategies correct over-absorption. |
| Avg. Molecules per Cluster | Varies widely | Skewed by dominant cluster | More uniform distribution | Improved "granularity" of chemical space dissection [41]. |
| Unique Scaffolds per Cluster | Baseline | High count in large cluster indicates mixing | Tighter scaffold focus per cluster | Refined BitBIRCH produces more structurally coherent clusters [41]. |
| Internal Validation Indices | Baseline | Comparable or superior [38] | Improved over original BitBIRCH | BitBIRCH efficiency does not come at the cost of quality. |
Diagram Title: BitBIRCH Tree Structure and Molecule Absorption
Application in Chemical Space Comparison Research
The primary thesis context involves comparing the chemical space of natural products (NPs), approved drugs, and combinatorial libraries. iSIM and BitBIRCH enable this research at scale.
Experimental Protocol for Time-Evolution Analysis: Using successive yearly releases of databases like ChEMBL and DrugBank [35]:
- Subset Extraction: Isolate NPs, approved drugs, and synthetic compounds using metadata.
- Diversity Trend Analysis: Apply iSIM to each subset for each release year to calculate iT. Plot iT over time to assess if diversity increases with size [35].
- Space Zone Tracking: Use complementary similarity to identify medoids (core) and outliers (periphery) for each subset. Compute the Jaccard similarity (J) of these zones between consecutive years to measure core/periphery stability [35].
- Granular Clustering: Apply BitBIRCH to the entire library for key release years. Analyze the distribution of NPs, drugs, and synthetic compounds across the resulting clusters to visualize overlap and uniqueness.
Table 4: Hypothetical iSIM Analysis of Chemical Space Subsets (Time-Evolution)
| Database Release | Natural Products (iT) | Approved Drugs (iT) | Combinatorial Compounds (iT) | Key Insight |
|---|---|---|---|---|
| ChEMBL25 (2017) | 0.152 | 0.189 | 0.121 | Initial baseline diversity measures. |
| ChEMBL29 (2021) | 0.149 | 0.185 | 0.119 | Minimal iT change suggests new compounds expand space without collapsing diversity. |
| ChEMBL33 (2023) | 0.148 | 0.184 | 0.118 | Stabilizing iT indicates managed diversity growth across all subsets [35]. |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 5: Key Research Reagents and Software for Large-Scale Chemical Space Analysis
| Item Name | Type | Function in Workflow | Relevance to iSIM/BitBIRCH |
|---|---|---|---|
| RDKit | Open-Source Cheminformatics Library | Molecule I/O, standardization, fingerprint generation (Morgan/ECFP), scaffold analysis. | Primary tool for preparing the binary fingerprint matrices required as input for both iSIM and BitBIRCH [43] [40]. |
| ChEMBL / DrugBank / PubChem | Public Chemical/Bioactivity Databases | Source of curated, annotated molecular structures for natural products, drugs, and synthetic compounds. | Provides the raw data for time-evolution studies and comparative chemical space analysis [35]. |
| BitBIRCH Python Package | Specialized Clustering Algorithm | Efficient O(N) clustering of binary fingerprints. | The implementation of the algorithm, available on GitHub (mqcomplab/bitbirch), includes refinement options like pruning [41]. |
| SciKit-Learn | Machine Learning Library | Provides t-SNE for visualization and utilities for calculating cluster validation indices (Calinski-Harabasz). | Used for post-clustering analysis and quality validation [41]. |
| High-Performance Computing (HPC) Node | Computational Resource | Provides the memory and parallel processing capabilities for billion-molecule clustering. | Essential for running the parallel/iterative version of BitBIRCH on ultra-large libraries [42]. |
Practical Implementation and Integration
BitBIRCH is designed for integration into modern cheminformatics pipelines. Its Python API follows a scikit-learn-like syntax for ease of adoption [40]. The package includes refinement strategies such as:
- Pruning: Removing and reinserting the largest cluster to prevent dominance.
- Diameter Merge Criterion: Enforcing that new molecules are similar to all cluster members, not just the centroid, creating tighter clusters [41].
- Tolerance Parameter (ε): Controlling how much a new molecule can decrease a cluster's internal similarity [41].
These refinements ensure the algorithm is not only fast but also robust and tunable for specific research needs, such as ensuring high purity in clusters derived from mixed-origin chemical spaces.
The iSIM framework and BitBIRCH algorithm represent a significant leap forward in handling the scale of modern chemical data. As evidenced by comparative benchmarks, they offer a multi-order-of-magnitude speed advantage over traditional pairwise methods without sacrificing analytical quality. Within the thesis of chemical space comparison, these tools enable rigorous, large-scale temporal and structural analyses that were previously impractical—allowing researchers to quantitatively test hypotheses about the growth and convergence of spaces occupied by natural products, drugs, and synthetic compounds.
Future development lies in tighter integration with active learning and generative AI pipelines in drug discovery, where rapid, iterative diversity assessment and cluster-based selection are crucial. By overcoming the computational bottleneck, iSIM and BitBIRCH shift the research question from "Can we analyze this?" to "What meaningful patterns can we find?"
The pursuit of novel therapeutics is a journey through immense and structurally diverse chemical spaces. Historically, these spaces have been navigated via two primary, often divergent, paths: the exploration of Natural Products (NPs) and the construction of Synthetic Compounds (SCs). NPs, the products of biological evolution, occupy a region of chemical space characterized by high scaffold complexity, rich stereochemistry, and biological pre-validation [10]. In contrast, SCs, particularly those from combinatorial chemistry, often explore areas defined by synthetic accessibility and adherence to drug-like rules, resulting in different structural and property profiles [44]. A time-dependent chemoinformatic analysis reveals that while NPs have evolved to become larger and more complex, SCs have undergone more constrained shifts in physicochemical properties, influenced by NPs but not fully converging with them [44].
This divergence presents both a challenge and an opportunity for modern drug discovery. Virtual Screening (VS) has long been the computational workhorse for sifting through large libraries, but its success is inherently limited to the chemical space defined by the screened collection [45]. AI-Driven De Novo Design promises a paradigm shift, generating novel, optimized molecules from scratch rather than selecting from a pre-defined list [46]. This article provides a comparative guide to these methodologies, framing their performance and experimental validation within the broader thesis of bridging the distinct but complementary chemical spaces of natural products and synthetic compounds. By integrating the biological relevance of NPs with the expansive explorative power of generative AI and large synthetic libraries, researchers can now design novel chemical entities—pseudo-natural products and optimized synthetic leads—that transcend traditional boundaries [10] [47].
Performance Comparison of Virtual Screening Methodologies
Virtual screening is a critical first step in computationally identifying potential drug candidates. Its efficacy depends on accurate scoring functions and robust benchmarking. Recent advances have focused on improving both the metrics for evaluation and the algorithms for screening ultra-large libraries.
Benchmarking Metrics and Model Performance
A fundamental challenge in VS is accurately assessing model performance in a way that predicts real-world success. The traditional Enrichment Factor (EF) is limited as its maximum value is constrained by the inactive-to-active ratio in the benchmark set, making it unsuitable for estimating performance on the vast libraries used in practice [48]. In response, the Bayes Enrichment Factor (EFB) has been proposed. This metric uses a set of random compounds instead of presumed inactives, allowing for the estimation of much higher enrichments relevant to real-world screening scenarios [48]. The maximum EFB (EFmaxB) is suggested as a best-guess for a model's prospective performance.
Performance data on the Directory of Useful Decoys - Enhanced (DUD-E) benchmark illustrates the variation between traditional and new metrics, as well as between different docking and machine learning models [48].
Table 1: Performance Comparison of Virtual Screening Models on the DUD-E Benchmark (Median Values) [48]
| Model | EF₁% | EFB₁% | EF₀.₁% | EFB₀.₁% | EFmaxB |
|---|---|---|---|---|---|
| Vina | 7.0 | 7.7 | 11 | 12 | 32 |
| Vinardo | 11 | 12 | 20 | 20 | 48 |
| Dense (Pose) | 21 | 23 | 42 | 77 | 160 |
State-of-the-Art Screening Platforms
The drive to screen multi-billion compound libraries has led to the development of high-performance platforms. RosettaVS, an AI-accelerated platform, exemplifies this advancement. It operates in two modes: a fast Virtual Screening Express (VSX) for initial triaging and a high-precision Virtual Screening High-precision (VSH) mode that incorporates full receptor flexibility for final ranking [45]. Its scoring function, RosettaGenFF-VS, combines enthalpy and entropy estimates.
On the CASF2016 benchmark, RosettaGenFF-VS achieved a top 1% enrichment factor (EF₁%) of 16.72, significantly outperforming the second-best method (EF₁% = 11.9) [45]. In a prospective test against two targets (KLHDC2 and NaV1.7), the platform identified hit compounds with a 14% and 44% experimental hit rate, respectively, with screening completed in under a week [45].
Integrative and Machine Learning-Based Approaches
Beyond traditional docking, machine learning models that learn evolutionary chemical binding similarity (ECBS) show promise. The Target-Specific ensemble ECBS (TS-ensECBS) model encodes features conserved across ligands binding to evolutionarily related targets [49]. When tested on a set of 51 kinases, the TS-ensECBS model outperformed both traditional 2D/3D ligand similarity methods and structure-based methods like molecular docking and pharmacophore modeling in prioritizing active compounds [49]. In a blind prospective screen for MEK1 inhibitors, this method alone identified 6 out of 13 confirmed hits, demonstrating its power in scaffold hopping and discovering novel chemotypes [49].
Table 2: Prospective Virtual Screening Performance Across Different Platforms
| Platform/Method | Target | Library Size | Experimental Hit Rate | Key Metric | Source |
|---|---|---|---|---|---|
| RosettaVS (AI-Accelerated) | KLHDC2 | Multi-billion | 14% (7 hits) | EF₁% = 16.72 | [45] |
| RosettaVS (AI-Accelerated) | NaV1.7 | Multi-billion | 44% (4 hits) | Completion <7 days | [45] |
| TS-ensECBS Model | MEK1 (Kinase) | Not specified | 46.2% (6/13 hits) | PR AUC = 0.93 | [49] |
| Dense (Pose) Model | DUD-E Avg. | N/A (Benchmark) | N/A | EFmaxB = 160 | [48] |
Experimental Protocol: RosettaVS Workflow [45]
- Library Preparation: A multi-billion compound library is prepared and pre-filtered.
- Active Learning Phase: A target-specific neural network is trained on-the-fly to predict docking scores, triaging compounds for full docking.
- VSX Docking: Top candidates from triage undergo rapid docking with RosettaVS's express mode.
- VSH Docking: The highest-scoring compounds from VSX are re-docked using the high-precision mode with full receptor flexibility.
- Ranking & Selection: Compounds are ranked using the RosettaGenFF-VS scoring function.
- Experimental Validation: Top-ranked compounds are procured and tested via binding assays (e.g., SPR) and, if successful, structure determination (e.g., X-ray crystallography).
Performance Comparison of AI-Driven De Novo Design Models
De novo design represents a generative approach to drug discovery, creating novel molecular structures that satisfy specified constraints. Deep learning, particularly transformer-based architectures, has revolutionized this field.
Generative Model Architectures and Performance
Current research focuses on adapting and optimizing advanced neural network architectures for molecular generation. Key innovations include modifications to the Generative Pre-trained Transformer (GPT) framework and the exploration of novel architectures like Mamba [46].
Table 3: Comparison of Deep Learning Models for De Novo Molecular Generation
| Model | Base Architecture | Key Innovation | Reported Advantage |
|---|---|---|---|
| MolGPT [46] | GPT (Decoder) | Conditional generation via scaffold token concatenation. | Established strong baseline for unconditional generation. |
| GPT-RoPE [46] | GPT | Rotary Position Embedding (RoPE). | Better handling of long-distance dependencies in sequences. |
| GPT-Deep [46] | GPT | DeepNorm layer normalization. | Improved training stability for very deep networks. |
| GPT-GEGLU [46] | GPT | GEGLU activation function. | Enhanced model expressiveness and flexibility. |
| Mamba [46] | Selective State Space | State space models for sequence modeling. | Linear-time scaling with sequence length, efficient for long contexts. |
| T5MolGe [46] | T5 (Encoder-Decoder) | Full encoder-decoder for conditional generation. | Learns mapping between property vectors and SMILES, enabling precise control. |
The T5MolGe model addresses a limitation of decoder-only models by using a full encoder-decoder structure. The encoder learns a dense representation of the desired conditional properties (e.g., targeting a specific mutant protein), which then guides the decoder to generate appropriate SMILES strings, offering more reliable property control [46].
Prospective Application in Drug Discovery
The ultimate test for generative models is the design of bioactive compounds for challenging targets. In one study, a conditional generation strategy targeting the L858R/T790M/C797S triple-mutant EGFR—a cause of resistance in non-small cell lung cancer—was employed [46]. The best-performing generative model (often a fine-tuned T5 or GPT variant) is used in a transfer learning strategy: first pre-trained on a large corpus of drug-like molecules, then fine-tuned on a smaller dataset of known EGFR inhibitors to generate novel, specific candidates for experimental testing [46].
Experimental Protocol: Conditional De Novo Design for a Mutant Target [46]
- Problem Definition: Specify the target (e.g., triple-mutant EGFR) and desired properties (inhibition, selectivity, drug-likeness).
- Model Selection & Training: Select a generative architecture (e.g., T5MolGe). Pre-train the model on a large dataset (e.g., ChEMBL, ZINC). Fine-tune the model on a focused dataset of relevant actives.
- Conditional Generation: Input the desired property profile into the model's encoder to generate thousands of novel molecular structures (SMILES).
- In Silico Filtering: Filter generated molecules for synthetic accessibility, predicted affinity (via docking or a scoring function), and ADMET properties.
- Synthesis & Testing: Prioritize top candidates for synthesis and subsequent in vitro and cellular assays to validate activity against the intended target.
Comparative Analysis of Chemical Spaces
Understanding the distinct characteristics of natural product and synthetic compound spaces is essential for guiding both virtual screening library selection and de novo design objectives.
Structural and Property Landscapes
A comprehensive, time-dependent analysis of over 186,000 NPs and SCs highlights their evolving differences [44]:
- Molecular Size & Complexity: NPs are generally larger (higher molecular weight, more heavy atoms) and possess more rings and stereocenters. Over time, newly discovered NPs have become even larger and more complex, while SC properties have fluctuated within a narrower, "drug-like" range [44].
- Ring Systems: NPs favor non-aromatic and fused ring systems (e.g., bridged, spiro), leading to more three-dimensional scaffolds. SCs are dominated by aromatic rings (especially benzene derivatives) and simpler ring assemblies [44].
- Biological Relevance: NPs, as products of evolution, are enriched in bioactive scaffolds. The biological relevance of SCs, as assessed by predictions from tools like PASS, is generally lower and has declined over recent decades [44].
Coverage and Complementarity of Large Virtual Spaces
The comparison of ultra-large, make-on-demand virtual chemical spaces reveals striking complementarity. A study comparing three large fragment spaces (BICLAIM, REAL Space, KnowledgeSpace) using a panel of 100 drug queries found a remarkably low overlap [13]. Only three compounds were found in the top hits from all three spaces. This demonstrates that different synthesis-driven virtual spaces explore largely non-overlapping regions of chemical universe, making the choice of space a critical determinant of accessible chemistry [13].
Table 4: Key Characteristics of Natural Product vs. Synthetic Compound Chemical Spaces [44]
| Characteristic | Natural Products (NPs) | Synthetic Compounds (SCs) |
|---|---|---|
| Scaffold Complexity | High; more stereocenters, more sp³ carbons. | Lower; more planar, aromatic structures. |
| Ring Systems | More non-aromatic rings, complex fused systems (bridged, spiro). | More aromatic rings (e.g., benzene), simpler ring assemblies. |
| Evolution Over Time | Increasing size and complexity. | Properties constrained within drug-like ranges; influenced by NPs but not converging. |
| Biological Pre-validation | Inherently high due to evolutionary selection. | Generally lower; must be designed or screened for. |
| Coverage of Chemical Space | Occupies a unique, biologically relevant but narrower region. | Can cover an extremely broad region, especially via virtual spaces. |
Visualizing Workflows and Chemical Space Bridging
Diagram 1: Bridging Chemical Spaces in Modern Drug Discovery. This workflow illustrates how NP and SC spaces inform both virtual screening of massive libraries and AI-driven de novo design, converging on validated hits through experimental testing.
Diagram 2: Workflow of a Modern AI-Accelerated Virtual Screening Platform. This detailed protocol shows the integration of active learning for efficiency, multi-tiered docking for accuracy, and final experimental validation.
The Scientist's Toolkit: Essential Research Reagents & Platforms
Table 5: Key Research Tools and Resources for Virtual Screening and De Novo Design
| Tool/Resource | Type | Primary Function in Research | Source / Example |
|---|---|---|---|
| BayesBind Benchmark | Benchmark Dataset | Provides a structurally dissimilar test set for evaluating VS models without data leakage, used with the EFB metric. | [48] |
| DUD-E / LIT-PCBA | Benchmark Dataset | Standard benchmarks for VS, containing known actives and decoys/inactives for multiple protein targets. | [48] |
| RosettaVS / OpenVS Platform | Software Platform | An open-source, AI-accelerated platform for high-performance docking and screening of ultra-large libraries. | [45] |
| TS-ensECBS Model | Machine Learning Model | Predicts chemical binding similarity based on evolutionary conserved features, enabling scaffold-hopping virtual screening. | [49] |
| Pseudo-NP Fragment Library | Chemical Design Principle | A collection of ~2000 fragments derived from deconstructing natural products, used to build novel, biologically relevant hybrids. | [10] |
| GPT-based & T5MolGe Models | Generative AI Model | Deep learning architectures (e.g., MolGPT, T5MolGe) for conditional or unconditional de novo generation of drug-like molecules. | [46] |
| REAL Space / Enamine | Make-on-Demand Chemical Space | A ultra-large virtual library (>4B compounds) with a high synthesis success promise, used for virtual screening. | [13] |
| RFdiffusion (Fine-tuned) | Generative AI Model | A protein diffusion model specialized for de novo design of antibody CDR loops and binding interfaces with atomic-level precision. | [50] |
| Schrödinger, Exscientia Platforms | Commercial AI Platform | Integrated drug discovery platforms combining physics-based simulation, generative AI, and automation for end-to-end lead design. | [51] |
The exploration of chemical space—the universe of all possible organic molecules—is a foundational challenge in modern drug discovery. This space is astronomically vast, estimated to contain over 10⁶⁰ drug-like molecules, yet only a minuscule fraction has been synthesized or tested for biological activity [52]. Within this context, integrative computational methodologies provide an essential toolkit for efficiently navigating this expanse to predict bioactivity and prioritize candidates for synthesis and testing. This guide objectively compares three core computational approaches—molecular docking, Quantitative Structure-Activity Relationship (QSAR) modeling, and molecular dynamics (MD) simulations—within the broader thesis of contrasting the chemical landscapes of natural products (NPs), synthetic drugs, and combinatorial compounds.
Natural products, with their evolutionary-optimized complexity and high sp³-carbon content, occupy a distinct and privileged region of chemical space known for high success rates in drug development [6]. Between 2014 and 2025, 45 new chemical entities derived from natural products were approved, representing 11.3% of all new small-molecule drugs [5]. In contrast, synthetic combinatorial libraries, often built from readily available scaffolds, offer unparalleled size and accessibility, with over 400 million compounds commercially available [53]. The strategic integration of docking, QSAR, and MD simulations allows researchers to leverage the unique advantages of each chemical domain, accelerating the identification of novel bioactive agents. These computational tools are no longer merely supportive; they are central to a transformative, target-focused paradigm that enhances the efficiency and success rate of the drug discovery pipeline [54].
Comparative Analysis of Core Computational Methodologies
The selection of a computational strategy depends on the stage of discovery, the available data, and the specific biological questions. The following table provides a direct comparison of the three core methodologies.
Table 1: Core Computational Methodologies for Bioactivity Prediction: A Comparative Guide
| Feature | Molecular Docking | QSAR Modeling | Molecular Dynamics (MD) Simulations |
|---|---|---|---|
| Primary Objective | Predict the binding pose and affinity of a ligand within a target protein's binding site. | Establish a quantitative mathematical relationship between molecular descriptors and biological activity. | Simulate the time-dependent behavior and stability of a protein-ligand complex in a solvated, near-physiological environment. |
| Key Strength | Structure-based design; visual insight into interaction modes (H-bonds, hydrophobic contacts). | Can predict activity for compounds lacking a known protein structure; high-throughput virtual screening. | Provides dynamic insight into conformational changes, binding stability, and mechanisms not apparent from static structures. |
| Principal Limitation | Accuracy depends on scoring functions and rigid/flexible treatment of the protein; may yield false positives. | Requires a dataset of known actives/inactives; predictive power limited to the chemical space of the training set. | Computationally expensive, limiting simulation time (ns-µs) vs. biological reality (ms-s); setup and analysis are complex. |
| Typical Output Metrics | Docking score (kcal/mol), predicted binding pose, intermolecular interaction maps. | Statistical coefficients (q², R², R²pred), predictive model equation, contribution plots of key descriptors. | RMSD, RMSF, radius of gyration (Rg), hydrogen bond lifetimes, binding free energy (MM/PBSA/GBSA). |
| Best Suited For | Virtual screening of large libraries against a known 3D protein structure; lead optimization. | Prioritizing synthesis from a homologous series; understanding key physicochemical properties driving activity. | Validating docking poses; studying allosteric mechanisms; estimating relative binding affinities of shortlisted hits. |
Integration and Complementary Roles
The true power of these tools is realized in integrative workflows. A standard pipeline may begin with ligand-based QSAR to screen an ultra-large virtual library, identifying a focused subset of promising scaffolds [52]. These candidates are then subjected to structure-based molecular docking against the target protein to evaluate complementarity and propose binding modes. Finally, top-ranking complexes undergo MD simulations to assess the stability of the proposed interactions, compute binding free energies, and filter out false positives that may bind only in a rigid, idealized model [55] [56]. This sequential integration leverages the high-throughput capacity of QSAR, the structural insights of docking, and the rigorous validation of MD, creating a robust funnel for candidate selection.
Performance Benchmarking: Experimental Data from Current Studies
Recent studies across diverse therapeutic targets demonstrate the performance of these methods individually and in concert. The data below, compiled from current literature, provides a benchmark for expected outcomes.
Table 2: Experimental Performance Metrics from Recent Integrative Studies (2024-2025)
| Study & Target | QSAR Model Performance | Top Docking Score (kcal/mol) | MD Simulation Results (Key Metrics) | Key Outcome |
|---|---|---|---|---|
| Imidazo-pyridines vs. Aurora Kinase A [55] | CoMSIA: q²=0.877, R²=0.995, R²pred=0.758 | N/A (Focused on designed compounds) | 50 ns MD; MM/PBSA confirmed stability of designed compounds (N3, N4, N5, N7) with 1MQ4. | QSAR models used to design 10 novel compounds; MD confirmed complex stability. |
| Fluorine-diamines vs. HCV NS5B [57] | 2D-QSAR: R²(ext)=0.5193, R²(int)=0.6427 | -241.463 (for designed compound SCD6) | 100+ ns MD; SCD6-3FQK RMSD ~2.00 Å; MM/GBSA = -117.85 ± 12.48 kcal/mol. | Designed compound SCD6 showed superior predicted affinity and stability. |
| Triazine-ones vs. Tubulin [56] | MLR Model: R²=0.849 | -9.6 (for Pred28) | 100 ns MD; Pred28-Tubulin RMSD lowest at 0.29 nm. | Pred28 identified as most stable and promising candidate for breast cancer therapy. |
| Machine Learning-Guided Docking [52] | CatBoost Classifier guided screening of 3.5B compounds. | Protocol specific to target. | N/A in initial screen. | Workflow reduced docking cost by >1000-fold, enabling screens of billion-compound libraries. |
Detailed Experimental Protocols for Key Methodologies
Protocol 1: Development and Validation of a Robust QSAR Model
This protocol is adapted from studies on imidazo[4,5-b]pyridine derivatives and 1,2,4-triazine-3(2H)-one derivatives [55] [56].
- Dataset Curation: Compile a homogeneous series of compounds (typically 30-100) with consistent biological activity data (e.g., IC₅₀, Ki). Convert IC₅₀ values to pIC₅₀ (-logIC₅₀) for linear modeling.
- Molecular Modeling and Descriptor Calculation:
- Generate 3D molecular structures using software like SYBYL or Gaussian.
- Perform geometry optimization using methods such as Density Functional Theory (DFT) with the B3LYP functional and a 6-31G(d,p) basis set.
- Calculate molecular descriptors: (a) Electronic descriptors (HOMO/LUMO energies, dipole moment, electronegativity) from quantum chemistry outputs; (b) Topological descriptors (molecular weight, logP, polar surface area) from packages like RDKit or ChemOffice.
- Data Division and Model Building:
- Randomly split data into a training set (75-80%) for model development and a test set (20-25%) for external validation.
- Use variable selection methods (e.g., genetic algorithm, stepwise regression) to identify the most relevant, non-correlated descriptors.
- Construct the model using techniques like Partial Least Squares (PLS) or Multiple Linear Regression (MLR).
- Model Validation:
- Internal Validation: Calculate cross-validated correlation coefficient (q²) via Leave-One-Out (LOO) or Leave-Group-Out (LGO).
- External Validation: Predict activity of the test set and calculate the predictive R² (R²pred). A model with q² > 0.5 and R²pred > 0.6 is generally considered predictive [55].
Protocol 2: Integrated Docking and Molecular Dynamics Simulation
This protocol is standard for validating protein-ligand interactions, as applied in studies of HCV NS5B and Tubulin inhibitors [57] [56].
- System Preparation:
- Protein: Obtain the 3D structure from the PDB. Remove water and heteroatoms, add missing hydrogen atoms, and assign protonation states (e.g., using H++ or PROPKA).
- Ligand: Prepare ligand topology and parameter files using tools like the GAFF force field and antechamber.
- Solvation and Neutralization: Place the protein-ligand complex in a solvent box (e.g., TIP3P water) with a buffer of ≥10 Å. Add ions to neutralize the system's charge.
- Molecular Docking:
- Define the binding site (often from a co-crystallized ligand).
- Perform docking using software like AutoDock Vina or Glide. Generate multiple poses and select the top-scoring pose based on both score and plausible interaction geometry.
- Molecular Dynamics Simulation:
- Energy Minimization: Perform steepest descent and conjugate gradient minimization to remove steric clashes.
- Equilibration: (1) Heat the system from 0 to 300 K over 50-100 ps under an NVT ensemble. (2) Adjust density over 100-500 ps under an NPT ensemble to reach 1 atm pressure.
- Production Run: Run an unrestrained simulation for a minimum of 50-100 ns (longer for complex systems) under NPT conditions (300K, 1 atm). Save trajectory frames every 10-100 ps.
- Trajectory Analysis:
- Stability: Calculate the Root Mean Square Deviation (RMSD) of the protein backbone and ligand.
- Flexibility: Calculate Root Mean Square Fluctuation (RMSF) of protein residues.
- Interactions: Analyze hydrogen bond occupancy and other non-covalent interactions.
- Energetics: Use the MM/PBSA or MM/GBSA method on trajectory snapshots to compute relative binding free energies.
Visualizing Integrative Workflows and Chemical Space
Diagram 1: Integrative Computational Drug Discovery Workflow
Diagram Title: Integrative Computational Drug Discovery Workflow
Diagram 2: Chemical Space Domains and Computational Access
Diagram Title: Chemical Space Domains and Computational Access
The Scientist's Toolkit: Essential Research Reagents & Solutions
Table 3: Key Research Reagent Solutions for Integrative Computational Studies
| Item / Resource | Function & Application | Example / Source |
|---|---|---|
| Curated Bioactivity Databases | Provide experimental data for QSAR model training and validation. Essential for linking chemical structure to biological response. | ChEMBL [54], PubChem BioAssay. |
| 3D Protein Structure Repositories | Source of atomic coordinates for target proteins, required for molecular docking and MD simulations. | Protein Data Bank (PDB) [54], AlphaFold DB. |
| Commercial & Virtual Compound Libraries | Sources of molecules for virtual screening. Includes purchasable compounds (for hit-to-lead) and ultra-large virtual libraries (for initial discovery). | ZINC15 [53] [52], Enamine REAL [52]. |
| Force Field Parameters | Sets of mathematical functions and constants used in MD simulations to calculate the potential energy of a molecular system. | CHARMM, AMBER, OPLS-AA (for proteins); GAFF (for small molecules). |
| Machine Learning-ready Molecular Descriptors | Numerical representations of molecular structure used as input for QSAR and ML models. | Morgan Fingerprints (ECFP) [52], CDDD descriptors [52], topological indices. |
| Free Energy Calculation Suites | Software tools to compute binding free energies from MD trajectories, providing a more accurate affinity estimate than docking scores. | MMPBSA.py (AMBER), gmx_MMPBSA (GROMACS). |
Navigating the Challenges: Data Gaps, Methodological Biases, and Optimization Strategies
Addressing Data Scarcity and Accessibility for Natural Product Libraries
The exploration of natural products (NPs) as a source for new therapeutics is fundamentally constrained by significant data scarcity and accessibility challenges. While NPs have historically been a prolific source of drug leads—approximately 50% of FDA-approved small-molecule drugs from 1981–2006 were NPs or their derivatives [58]—their modern discovery and development are hindered by limited, non-uniform, and often inaccessible data [58] [59]. This scarcity stands in stark contrast to the vast, ever-expanding libraries of synthetic compounds (SCs), which now number in the hundreds of millions [44].
The core of the problem lies in the intrinsic nature of NP discovery. Isolating and characterizing novel bioactive compounds from biological sources is a labor-intensive, low-yield process [58]. The development of the anticancer drug Taxol, for instance, spanned 30 years [58]. This results in datasets that are orders of magnitude smaller than those for SCs. Furthermore, NP data is often fragmented across specialized, non-standardized databases and buried in heterogeneous scientific literature, creating significant accessibility barriers [58] [59].
This data paucity critically undermines the application of modern Artificial Intelligence (AI) and Machine Learning (ML) methods, which are data-hungry by design and have revolutionized the screening and design of synthetic libraries [58] [59]. Consequently, the drug discovery community faces a paradoxical situation: NPs occupy a unique and biologically relevant region of chemical space [44], yet this space remains profoundly underexplored due to infrastructural data limitations. This comparison guide analyzes current strategies to overcome these hurdles, objectively evaluating their performance against methods used for combinatorial compound libraries, and provides the experimental and informatics frameworks necessary for advancement.
Comparative Analysis of Data Scarcity Handling Methods
The following table compares contemporary computational strategies designed to maximize insights from limited NP data, contrasting them with their typical application in data-rich SC environments.
Table 1: Comparison of AI/ML Strategies for Data-Scarce vs. Data-Rich Regimes
| Method | Core Principle | Typical Application in SC Research (Data-Rich) | Application & Efficacy in NP Research (Data-Scarce) | Key Experimental/Validation Metrics |
|---|---|---|---|---|
| Transfer Learning (TL) [59] | Leverages knowledge from a source model trained on a large, related dataset to improve learning on a small target dataset. | Used to fine-tune models between large synthetic libraries (e.g., ChEMBL to a proprietary SC library). Highly effective for related tasks. | Critical for NPs. A model pre-trained on massive SC databases (e.g., ChEMBL's 2.4M+ compounds) can be fine-tuned on small NP datasets (<100k molecules) for property prediction [59]. Performance gains are substantial but depend on source-target relevance. | Mean Squared Error (MSE) reduction in property prediction (e.g., bioactivity, solubility); Accuracy/F1-score improvement in classification tasks (e.g., toxicity, target class). |
| Active Learning (AL) [59] | An iterative process where a model selectively queries an "oracle" (experiment) to label the most informative data points from an unlabeled pool. | Used to optimize high-throughput screening campaigns, reducing the number of assays needed to find hits. | High potential for guiding NP isolation. Can prioritize which NP extracts or fractions to analyze spectroscopically based on predicted novelty or bioactivity [59]. Drastically reduces experimental cost and time. | Learning curves showing model performance (AUC, hit rate) vs. number of queries; Yield of novel bioactive entities per unit of experimental effort. |
| Data Augmentation (DA) & Synthesis (DS) [59] | DA creates modified versions of existing data; DS uses generative models to create entirely new, realistic synthetic data. | DA is common in image-based screening. DS (e.g., using GANs) generates novel virtual SC libraries for de novo design. | DA is challenging due to complex NP stereochemistry. DS is promising for generating "pseudo-NPs" by combining NP-inspired scaffolds [59] [44]. These molecules can occupy novel but biologically relevant chemical space. | Frechet ChemNet Distance (FCD) measuring similarity between real and generated NP distributions; Synthetic accessibility score (SAS) of generated molecules; In vitro validation hit rate. |
| Multi-Task Learning (MTL) [59] | A single model is trained jointly on multiple related tasks, sharing representations to improve generalization. | Common in polypharmacology to predict activity against multiple protein targets simultaneously using large bioactivity matrices. | Useful for multiplexed NP profiling. A single model can predict multiple bioactivities (e.g., antibacterial, anticancer, anti-inflammatory) from limited NP data, leveraging shared underlying features [59]. | Average performance improvement across all tasks vs. single-task models; robustness to noise in individual assay datasets. |
| Federated Learning (FL) [59] | Enables model training across decentralized data sources (e.g., different labs) without sharing the raw data itself. | Emerging in pharma consortia to build models on pooled but proprietary SC data without violating IP. | Ideal for fragmented NP data. Allows institutions with unique, small NP collections (e.g., marine samples, traditional medicine extracts) to collaboratively train a global model without surrendering physical samples or full datasets [59]. | Global model performance vs. models trained on any single institution's data; time to convergence across participants. |
Experimental Protocols for Key Methodologies
Protocol: Implementing Transfer Learning for NP Property Prediction
This protocol details the steps to adapt a model trained on large synthetic compound databases to predict properties for natural products [58] [59].
Source Model Selection & Data Preparation:
- Source Data: Obtain a large, public bioactivity dataset (e.g., ChEMBL release 33, containing >20 million bioactivities) [35]. Standardize molecules (remove salts, neutralize charges) and curate a specific endpoint (e.g., pIC50 for a kinase target family).
- Pre-training: Train a deep neural network (DNN) or graph neural network (GNN) from scratch on this source data. Use molecular fingerprints or graph representations as input. This model learns generalizable features of chemical structure-activity relationships.
Target NP Dataset Curation:
- Assemble a small, high-quality NP dataset with the same target property (e.g., 500-5,000 NPs with measured pIC50 for a specific kinase). Ensure rigorous dereplication to remove duplicates [58].
Transfer Learning Execution:
- Architecture Adaptation: Remove the final output layer of the pre-trained source model.
- Feature Extraction & Fine-tuning: Two-stage process:
- Stage 1 (Feature Extraction): Freeze all weights of the pre-trained model. Use its outputs as fixed feature vectors to train a new classifier/regressor (e.g., a Random Forest or a shallow neural network) on the NP target data.
- Stage 2 (Fine-tuning): Unfreeze some or all layers of the pre-trained model. Continue training the entire network on the NP target data using a very low learning rate (e.g., 1e-5) to gently adapt the learned features to the NP domain without catastrophic forgetting.
Validation:
- Perform rigorous k-fold cross-validation on the NP dataset.
- Compare the performance (MSE, R²) of the TL model against: (a) a model trained only on the small NP dataset from random initialization, and (b) a model trained only on the large SC source data and applied directly to NPs. Superior performance of the TL model demonstrates successful knowledge transfer [59].
Protocol: Active Learning-Guided Bioassay Prioritization
This protocol outlines an iterative computational-experimental cycle to efficiently discover bioactive NPs from a library of untested extracts [59].
Initial Setup & Model Training:
- Initial Seed Set: Start with a small, randomly selected subset of the NP extract library (e.g., 5%) that has been fully characterized (structure elucidated) and assayed for the target activity (e.g., inhibition of a parasite growth).
- Model Training: Train a classification model (e.g., Support Vector Machine) to predict active/inactive labels using molecular descriptors of the characterized NPs in the seed set.
Iterative AL Cycle:
- Prediction & Uncertainty Scoring: Use the trained model to predict activity for all remaining unlabeled extracts in the library. For each prediction, calculate an uncertainty score (e.g., entropy of the class probability, or distance from the decision boundary in an SVM).
- Query Selection: Rank all unlabeled extracts by their uncertainty score. Select the top N (e.g., 20) most uncertain extracts for experimental testing. This targets samples the model is least confident about, maximizing information gain.
- Experimental Labeling: Perform the bioassay and structural elucidation (e.g., via LC-MS/MS, NMR) on the selected N extracts to obtain definitive "active/inactive" labels and structures.
- Model Update: Add the newly labeled data to the training set. Retrain or update the ML model.
- Repeat steps 2a-2d for a fixed number of cycles or until a performance target is met (e.g., discovery of 10 novel active scaffolds).
Performance Evaluation:
- Plot a learning curve: cumulative number of novel active NPs discovered vs. total number of extracts assayed. Compare this curve to one generated from a random selection baseline. The AL approach should discover actives at a significantly higher rate [59].
Chemical Space Comparison: NPs vs. Synthetic Libraries
Quantitative cheminformatic analyses reveal fundamental and evolving differences between the chemical spaces of NPs and SCs, which directly influence data generation strategies and library design [35] [44].
Table 2: Time-Dependent Structural & Property Comparison of NPs vs. Synthetic Compounds (SCs) [44]
| Property Category | Trend in Natural Products (over time) | Trend in Synthetic Compounds (over time) | Implication for Library Design & Data Scarcity |
|---|---|---|---|
| Molecular Size (Weight, Heavy Atoms) | Consistent increase. Modern NPs are larger. | Constrained variation. Governed by drug-like rules (e.g., Lipinski's RO5). | NP data reflects broader size ranges, challenging standard ADMET prediction models trained on SCs. Requires TL/MTL adaptation. |
| Ring Systems | Increasing number of non-aromatic, fused rings (e.g., bridged, spiro). Higher glycosylation. | Dominated by aromatic rings (e.g., benzene, pyridine). More ring assemblies. | NP scaffolds are more complex and three-dimensional [44]. This structural complexity contributes to data scarcity (harder to characterize, synthesize) but offers novel bioactivity. |
| Hydrophobicity (CLogP) | Trend towards higher hydrophobicity. | More tightly clustered within a moderate range (typically 0-5). | NPs explore a wider lipophilicity space, which can be advantageous for challenging targets (e.g., protein-protein interfaces) but poses solubility challenges. |
| Chemical Diversity | High and increasing structural uniqueness over time. | Diversity increases with library size, but can plateau (adding molecules doesn't always add new chemotypes) [35]. | Even small, well-curated NP libraries can add significant novelty to a screening collection, justifying the high cost-per-compound data generation. |
| Biological Relevance | Inherently high due to evolutionary selection for biomolecular interaction. | Can decline as libraries grow via purely synthetic feasibility-driven expansion. | A unit of NP data has a higher prior probability of containing bioactive compounds, making AL and focused data generation more efficient. |
Visualizing Chemical Space and Workflow Strategies
The following diagrams, created using Graphviz DOT language, illustrate key concepts and workflows for addressing NP data scarcity.
Diagram 1: AI strategies for NP data scarcity. This workflow shows how multiple computational strategies integrate to build robust models from limited NP data, creating a synergistic cycle with experimental validation.
Diagram 2: Comparative chemical space of NPs and SCs. This diagram contrasts the defining characteristics of synthetic and natural product chemical spaces, highlighting the unique value and challenges of the NP region.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table lists key computational tools, databases, and resources essential for implementing the strategies described in this guide.
Table 3: Essential Toolkit for Addressing NP Data Scarcity
| Tool/Resource Name | Type | Primary Function in NP Research | Key Consideration |
|---|---|---|---|
| ChEMBL [35] | Public Bioactivity Database | Primary source dataset for Transfer Learning. Contains millions of standardized bioactivity records for SCs, used to pre-train predictive models. | Manually curated, high-quality. Contains a subset of NPs, but primarily SCs. |
| iSIM & BitBIRCH Algorithms [35] | Cheminformatics Algorithms | Quantify intrinsic similarity (iSIM) and perform efficient clustering (BitBIRCH) of ultra-large libraries. Critical for analyzing NP library diversity vs. SC libraries. | Enables O(N) scaling analysis, making large-scale NP-SC chemical space comparison feasible. |
| Dictionary of Natural Products (DNP) [44] | Commercial NP Database | A comprehensive, curated source of NP structures and data. Serves as a standard reference for NP chemical space analysis and dereplication. | Subscription-based. Essential for building clean, non-redundant NP datasets for model training. |
| COCONUT | Public NP Database | An open-access collection of NP structures. Useful for assembling large NP datasets for exploratory analysis and model training, complementing commercial sources. | Requires rigorous curation for quality control. |
| RDKit | Open-Source Cheminformatics Toolkit | Provides the foundational functions for molecular standardization, descriptor calculation, fingerprint generation, and model input preparation for both NPs and SCs. | The workbench for most custom cheminformatics pipelines. |
| GNINA or DeepDock | Deep Learning Docking Software | Structure-based virtual screening tools that can be used with NP libraries. Performance can be boosted via TL from models trained on large synthetic compound docking data. | Requires a protein target structure. Computational cost is higher than ligand-based methods. |
| Federated Learning Framework (e.g., Flower, NVIDIA FLARE) | ML Orchestration Software | Enables the setup of privacy-preserving collaborative learning networks across institutions holding private NP data, implementing the FL strategy. | Requires coordination and technical setup across participating entities. |
A persistent methodological bias conflates the sheer quantity of compounds in a library with its useful chemical diversity. This pitfall is particularly evident in the historical comparison of natural products (NPs), valued for their biological relevance and structural uniqueness, and vast libraries of synthetic or combinatorial compounds (SCs), prized for their accessibility and scale [44] [9]. While combinatorial chemistry can generate millions of novel structures, evidence suggests this numerical growth does not automatically equate to an expansion in functionally meaningful chemical space or in the discovery of new biological probes [35] [9]. True chemical diversity is defined not by cardinality but by the breadth of distinct molecular scaffolds, stereochemistry, functional groups, and coverage of biologically relevant chemical space (BioReCS)—the region occupied by molecules with biological activity [1]. This guide objectively compares the performance of NP-inspired discovery and combinatorial synthesis, using contemporary chemoinformatic analyses to highlight the critical distinction between quantity and diversity in effective drug discovery.
Experimental Protocols for Chemical Space Comparison
Comparative analyses rely on standardized chemoinformatic workflows to ensure objective evaluation. The following methodologies are foundational to recent studies.
2.1 Time-Dependent Chemoinformatic Analysis [44]
- Objective: To track and compare the structural evolution of NPs and SCs over time.
- Data Curation: NPs were sourced from the Dictionary of Natural Products; SCs were aggregated from 12 synthetic compound databases. Molecules were sorted chronologically by CAS Registry Number and grouped into sequential sets (e.g., 5,000 molecules per group).
- Descriptor Calculation: For each molecule, 39 physicochemical properties were computed (e.g., molecular weight, logP, ring counts). Molecular fragmentation was performed to generate Bemis-Murcko scaffolds, ring assemblies, and RECAP fragments.
- Diversity & Space Analysis: Chemical space was visualized and compared using Principal Component Analysis (PCA), Tree MAP (TMAP), and SAR Map. Scaffold diversity was quantified using Shannon entropy metrics.
- Biological Relevance Assessment: Predicted biological activities were generated using a PASS-like algorithm to compare the potential bioactivity profiles of NP and SC sets.
2.2 Intrinsic Similarity (iSIM) and Clustering for Library Growth Analysis [35]
- Objective: To determine if library growth (quantity) leads to increased chemical diversity.
- Tool - iSIM Framework: This method calculates the average pairwise Tanimoto similarity for an entire library in O(N) time, avoiding the computationally prohibitive O(N²) scaling. The iT (iSIM Tanimoto) value represents the library's internal diversity (lower iT = greater diversity).
- Tool - Complementary Similarity: Identifies molecules central to a library's chemical space (medoids) and those on the periphery (outliers) by calculating the change in iT when a molecule is removed.
- Tool - BitBIRCH Clustering: An efficient clustering algorithm for binary fingerprints (e.g., Morgan fingerprints) that groups molecules into structurally similar clusters. The formation of new clusters over time indicates diversity expansion.
- Protocol: Applied to sequential releases of public libraries (e.g., ChEMBL, DrugBank). For each release, iT is calculated, and clusters are generated. Growth in diversity is assessed by tracking iT trends and the emergence of new, distinct clusters not present in prior releases.
2.3 Similarity Networking for Focused NP Analysis [60]
- Objective: To visualize and assess the chemical diversity within a specific NP class (e.g., cyanobacterial metabolites).
- Protocol: Molecular structures are encoded into fingerprints (e.g., MACCS keys). A similarity matrix is calculated, and a network graph is constructed where nodes represent compounds and edges represent significant structural similarity. Analysis of network topology (e.g., clusters, singleton nodes) reveals the density and uniqueness of the chemical space covered.
Performance Comparison: Natural Products vs. Combinatorial Libraries
The following tables summarize key comparative data derived from the application of the above protocols.
Table 1: Comparison of Structural and Physicochemical Properties [44]
| Property | Natural Products (Trend Over Time) | Synthetic/Combinatorial Compounds (Trend Over Time) | Interpretation & Implication |
|---|---|---|---|
| Molecular Size | Steady increase (MW, volume, heavy atoms). | Constrained within a limited range. | NPs are becoming larger and more complex; SCs are bounded by "drug-like" rules (e.g., Lipinski's Rule of Five). |
| Ring Systems | Increasing number of rings, especially non-aromatic and fused rings. Glycosylation increasing. | Increase in aromatic rings (especially 5- and 6-membered). Stable count of non-aromatic rings. | NPs exhibit greater scaffold complexity and stereochemistry. SCs favor synthetically accessible flat, aromatic systems. |
| Complexity & Saturation | Increasing molecular complexity, decreasing fraction of sp³ carbons (Fsp³). | Relatively stable, lower complexity. Higher Fsp³ in later years. | Modern NPs are complex but less saturated. SC libraries initially lacked complexity; recent designs aim to mimic NP complexity. |
| Scaffold Diversity | High and increasing scaffold uniqueness. | Lower scaffold diversity; high redundancy of common rings (e.g., benzene). | A large SC library may contain millions of compounds built on a relatively small set of simple, similar scaffolds. |
Table 2: Assessment of Library Growth vs. Diversity Expansion [35]
| Analysis Metric | Finding in Public Library Analysis (e.g., ChEMBL) | Interpretation & Implication |
|---|---|---|
| Intrinsic Similarity (iT) | The iT value often remains stable or decreases only slightly across major library releases, despite massive growth in the number of compounds. | Quantity ≠ Diversity. Adding many structurally similar compounds does not meaningfully expand the occupied chemical space. |
| Cluster Analysis (BitBIRCH) | New library releases primarily add compounds to existing structural clusters rather than creating new, distinct clusters. | Library growth is often about filling in known regions of chemical space rather than pioneering new ones. This limits the discovery of novel chemotypes. |
| Complementary Similarity | The "medoid" core of the library remains stable; new "outlier" compounds are added but are few relative to total additions. | Most synthetic efforts target regions near well-explored, successful scaffolds. Truly novel outliers are rare, highlighting a bias toward known chemical space. |
Table 3: Biological Relevance and Drug Discovery Performance [44] [9]
| Criterion | Natural Products | Combinatorial/Synthetic Libraries | Supporting Data |
|---|---|---|---|
| Coverage of BioReCS | Occupy unique and relevant regions, evolved to interact with biomolecules. | Broader coverage of possible chemical space, but with declining biological relevance over time [44]. | NPs show higher predicted hit rates against biological targets. SCs require careful design to target BioReCS [1]. |
| Drug Discovery Success | ~68% of new small-molecule drugs (1981-2019) are derived from NPs [44]. | High-throughput screening (HTS) of combinatorial libraries has not yielded the expected avalanche of new drugs [9]. | Highlights the "productivity paradox" of combinatorial chemistry: more compounds screened, but not more leads. |
| Lead Optimization | Often require complex total synthesis or derivatization for optimization. | Ideally suited for rapid analog synthesis via combinatorial methods to explore structure-activity relationships (SAR). | Suggests an optimal strategy: discover novel leads from NPs, then optimize using combinatorial or parallel synthesis techniques. |
Visualizing Concepts and Workflows
Diagram 1: Methodology for Time-Dependent Chemical Space Comparison
Diagram 2: The Pitfall: Library Growth vs. Diversity Expansion
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Reagents and Solutions for Chemical Diversity Analysis
| Item / Solution | Function / Role in Analysis | Key Consideration for Bias Mitigation |
|---|---|---|
| Curated Natural Product Databases (e.g., Dictionary of Natural Products, COCONUT) | Provide standardized, annotated structural data for NPs as a benchmark for complexity and BioReCS [44] [1]. | Ensure temporal metadata is available for time-series analysis to avoid treating NPs as a static set. |
| Large Synthetic Libraries (e.g., Enamine REAL, ZINC, proprietary corporate libraries) | Represent the output of combinatorial chemistry for comparison. Serve as a source for virtual screening [27] [35]. | Must be analyzed in subsets (e.g., by date, vendor) to detect temporal trends and intrinsic redundancy. |
| Cheminformatics Toolkits (e.g., RDKit, OpenBabel) | Open-source libraries for calculating molecular descriptors, generating fingerprints, and performing fragmentations essential for standardized analysis [44] [35]. | Critical for implementing reproducible workflows and avoiding black-box commercial software biases. |
| Specialized Analysis Software (e.g., iSIM framework, BitBIRCH algorithm) | Enable efficient diversity analysis (iSIM) and clustering of ultra-large libraries (BitBIRCH), which traditional O(N²) methods cannot handle [35]. | These modern tools are essential for accurately assessing diversity in million+ compound libraries. |
| Visualization Platforms (e.g., TMAP, ChemSuite) | Generate intuitive 2D/3D maps of chemical space from high-dimensional descriptor data, allowing visual assessment of overlap and uniqueness [44] [1]. | Helps researchers move beyond single-number diversity metrics (e.g., molecule count) to a spatial understanding. |
| Bioactivity Databases (e.g., ChEMBL, PubChem BioAssay) | Provide experimental biological data to link chemical structures to regions of BioReCS and validate the biological relevance of explored chemical space [1] [35]. | Integrating bioactivity data is crucial to shift focus from "chemical diversity" to "relevant chemical diversity." |
The evidence clearly demonstrates that methodological bias favoring quantity over true chemical diversity has led to suboptimal library design and screening outcomes. While combinatorial chemistry excels at generating vast numbers of compounds and optimizing leads, NP research remains an unparalleled source of novel, biologically relevant scaffolds [44] [9].
Strategic Recommendations for Researchers:
- Adopt Advanced Metrics: Move beyond compound count. Implement iSIM for internal diversity and use BitBIRCH clustering to track the generation of genuinely new chemotypes in library expansion projects [35].
- Embrace Hybrid Design: Integrate NP-inspired complexity (e.g., sp³-richness, stereocenters, privileged NP scaffolds) into combinatorial library design to create "pseudo-natural product" libraries that marry diversity with synthetic accessibility [44] [9].
- Prioritize Biological Relevance: Design and select screening libraries based on predicted or measured coverage of BioReCS, using tools that integrate bioactivity data with chemical descriptors [1].
- Conduct Temporal Analysis: Regularly analyze the temporal evolution of both internal and external compound collections to identify trends towards homogeneity and correct course toward greater structural uniqueness [44].
Overcoming the quantity-diversity pitfall requires a conscious shift in methodology from a focus on combinatorial explosion to a principled exploration of chemical space, where the quality, uniqueness, and biological relevance of compounds are the primary metrics of success.
The pursuit of new therapeutic agents is a voyage through an almost incomprehensibly vast chemical universe. Estimates suggest the number of synthetically feasible, drug-like molecules exceeds 10^60, a figure that dwarfs the number of stars in the observable universe [61]. Navigating this space to discover novel, effective, and developable drugs is the central challenge of modern drug discovery. This endeavor necessitates a strategic comparison of distinct regions of chemical space: the biologically validated complexity of natural products, the optimized properties of marketed drugs, and the accessible expanses explored by combinatorial and synthetic compounds [61] [17].
Historically, these regions were explored in isolation. Early drug discovery relied heavily on natural products and their derivatives, which account for approximately 50% of marketed small-molecule drugs [61]. The advent of combinatorial chemistry in the late 1980s and 1990s promised a more systematic exploration, enabling the parallel synthesis of vast libraries containing millions of compounds [62]. However, the initial focus on maximizing sheer library diversity did not translate to a proportional increase in new drug candidates [17]. This led to a pivotal evolution in library design philosophy—from a singular focus on size and diversity to a multi-objective optimization that critically balances three core pillars: broad structural diversity to explore novel biology, optimal drug-likeness to ensure developmental viability, and practical synthetic feasibility to bridge the gap between virtual design and tangible molecules [17] [63]. This guide provides a comparative analysis of contemporary strategies and technologies designed to achieve this essential balance.
The Evolution of Library Design Strategy
The design of compound libraries for screening has undergone a fundamental shift, moving from a quantity-focused paradigm to one prioritizing quality, focus, and synthetic realism.
- From "Drug-Like" to "Lead-Like" and "Hit-Like": Initial library designs aimed for "drug-like" properties, guided by rules like Lipinski's Rule of Five. Analysis revealed that the molecular properties of optimized drugs differ from their initial hits; drugs tend to be larger and more lipophilic [17]. This insight spurred the "lead-like" concept, focusing on smaller, less complex molecules with room for optimization. Further refinement led to "hit-like" filters, which prioritize compounds with properties suitable for generating robust signals in high-throughput screening (HTS) assays [61].
- Incorporating ADMET Filters: A major driver of this evolution is the need to reduce attrition in late-stage development. It is now standard practice to integrate computational predictions of Absorption, Distribution, Metabolism, Excretion, and Toxicity (ADMET) properties during the library design stage to filter out compounds with probable pharmacokinetic or toxicity issues [62] [17].
- The Rise of Focused and Targeted Libraries: While diverse libraries remain crucial for novel target discovery, "focused libraries" designed around specific biological targets or protein families have become commonplace. These libraries leverage prior knowledge from virtual docking, pharmacophore models, or known active compounds to increase the probability of finding hits [17].
- Synthetic Feasibility as a First-Class Parameter: The ultimate test of a virtual design is its translation to a synthesized compound. Modern library design emphasizes synthetic accessibility, using retrosynthetic analysis and rules based on reliable, high-yielding reactions (e.g., amide couplings, Suzuki-Miyaura cross-couplings) to ensure designed molecules can be made quickly and reliably [63]. Tools like the RAscore (Retrosynthetic Accessibility Score) provide rapid, machine-learning-based assessments of synthetic feasibility for millions of compounds [63].
The following diagram illustrates this integrated, multi-objective workflow that defines modern, optimized library design.
Diagram 1: Multi-Objective Library Design Workflow. The process integrates three core objectives (diversity, drug-likeness, synthetic feasibility) through specific computational methods, producing a library for experimental validation, whose results feed back into iterative design refinement [17] [63].
Comparative Analysis of Chemical Space Generation Approaches
The strategies for generating and exploring chemical space can be broadly categorized. The table below compares key features of major approaches, highlighting their respective advantages in the context of the diversity-druglikeness-feasibility balance.
Table 1: Comparison of Chemical Space Generation Approaches
| Approach | Typical Scale | Key Strengths | Primary Considerations | Example / Application |
|---|---|---|---|---|
| Combinatorial Solid-Phase (OBOC) [62] | Thousands to millions | High diversity, one-bead-one-compound, suitable for on-bead screening. | Requires decoding of active beads, chemistry must be solid-phase compatible. | Peptide and peptidomimetic library screening. |
| DNA-Encoded Libraries (DELs) [62] | Billions to trillions | Unprecedented scale, efficient selection-based screening, amplifiable information. | Chemistry must be compatible with DNA tags; hit validation requires off-DNA synthesis. | Affinity selection against purified protein targets. |
| Parallel Synthesis / Focused Arrays [62] | Hundreds to thousands | High purity, known structures, flexible chemistry, excellent for lead optimization. | Lower diversity, higher cost per compound. | Analog series synthesis for SAR exploration. |
| Virtual "On-Demand" Libraries [13] [63] | Millions to billions (enumerated); >10^20 (un-enumerated spaces) | Vast, drug-like space, designed for synthetic feasibility (e.g., 2-3 step synthesis). | Hits are virtual until synthesized; success depends on reliability of synthetic rules. | REAL Space, AXXVirtual, Ultra-large virtual screening [13] [63]. |
| Natural Product-Inspired [61] [17] | Varies | Biologically relevant, complex scaffolds, high success rate in drug discovery. | Synthetic complexity can hinder lead optimization; sourcing and purification challenges. | Libraries based on privileged natural product scaffolds (e.g., macrocycles). |
A critical insight from recent comparative studies is that even massive, ostensibly comprehensive chemical spaces exhibit strikingly low overlap. A study comparing three large fragment spaces (BICLAIM, REAL Space, KnowledgeSpace) by searching the vicinity of 100 marketed drug queries found that, of nearly 1 million unique hits retrieved from each space, only three compounds were common to all three [13]. This profound complementarity underscores that no single source or approach can adequately cover relevant chemical space, necessitating a combined strategy.
Performance of Commercial Screening Collections
For many academic and industrial labs, sourcing compounds from commercial vendors is a primary strategy. The table below summarizes the scale and property profiles of major commercial screening collections, providing a basis for selection.
Table 2: Overview of Major Commercial Small-Molecule Screening Collections (Representative Data) [61]
| Compound Source | Collection Name | Number of Compounds | % Passing Lipinski's Rule of 5* | % Passing REOS Filters* |
|---|---|---|---|---|
| Enamine | HTS Collection | ~1.1 million | 90.7% | 79.6% |
| ChemDiv | Discovery Chemistry | ~790,000 | 73.8% | 72.1% |
| ChemBridge | Express Pick Library | ~442,000 | 84.0% | 66.6% |
| Life Chemicals | Stock | ~327,000 | 84.9% | 76.6% |
| Vitas-M Lab | HTS Stock | ~476,000 | 75.1% | 65.8% |
| Asinex | Gold & Platinum | ~364,000 | 79.6% | 73.0% |
| Reference: Marketed Drugs (DrugBank) | - | ~4,900 | 71.4% | 51.7% |
*Lipinski's Rule of 5 and REOS (Rapid Elimination of Swill) are standard filters for drug-likeness and the removal of problematic substructures, respectively [61]. Data illustrates vendor focus on providing "drug-like" compounds.
Experimental Protocols for Library Evaluation and Validation
Protocol 1: Assessing Chemical Space Overlap and Complementarity
This protocol, adapted from a published comparison study, evaluates the structural overlap between large chemical spaces without full enumeration [13].
- Query Selection: Assemble a panel of 100 reference molecules. The study used marketed drugs filtered for drug-like properties (MW < 600, clogP < 6, etc.) to focus on pharmaceutically relevant space [13].
- Similarity Search: For each query, perform a nearest-neighbor search in each chemical space (e.g., BICLAIM, REAL Space, KnowledgeSpace) to retrieve the top 10,000 most similar compounds. The study used the Feature Trees (FTrees) descriptor, which is adept at identifying scaffold hops [13].
- Hit Set Analysis: Pool the results for each space (yielding ~1 million compounds per space). Determine the number of unique structures.
- Overlap Calculation: Perform pairwise and multi-space structural comparisons on the unique hit sets using a standard fingerprint method (e.g., MDL public keys). Calculate the count of structures appearing in two or all three spaces [13].
- Interpretation: A low overlap count (as found in the study) indicates high complementarity, suggesting that different spaces explore distinct regions of chemical geography and that combining sources is beneficial.
Protocol 2: Validating Synthetic Feasibility of a Virtual Library
This protocol describes steps to ensure a computationally designed library can be translated into practice, as implemented in the development of the AXXVirtual library [63].
- Reaction Rule Definition: Limit library construction to 2-3 synthetic steps using 6-8 robust, high-yielding reaction types (e.g., amide coupling, Suzuki reaction, reductive amination).
- Building Block Sourcing: Select all building blocks (>3,000) from a real, in-stock inventory of a trusted chemical supplier to guarantee immediate availability [63].
- Virtual Library Enumeration: Generate all possible products from the defined reactions and building blocks.
- Computational Filtering: Apply sequential filters:
- Drug-likeness: Apply rules (e.g., Lipinski, Veber) and remove pan-assay interference compounds (PAINS) and toxicophores [63].
- Diversity Clustering: Use a scalable algorithm (e.g., leader clustering, BIRCH) to cluster molecules based on structural fingerprints and select a diverse subset [63].
- Synthetic Accessibility Scoring: Score all compounds with a tool like RAscore, a machine learning classifier trained on retrosynthetic analysis outcomes. Retain compounds with a high score (e.g., >0.8 on a 0-1 scale) [63].
- Empirical Validation: Synthesize a representative sample of compounds (e.g., 100-500) across different clusters and reaction pathways to confirm predicted yields, purity, and synthesis timelines (e.g., 2-3 weeks for 100 compounds) [63].
The Scientist's Toolkit: Key Reagent Solutions for Library Synthesis
Successful translation from virtual design to physical library hinges on reliable chemical building blocks and reactions.
Table 3: Essential Research Reagents for Focused and Combinatorial Library Synthesis
| Reagent Category | Function & Importance | Examples & Notes |
|---|---|---|
| Diverse Building Blocks | Provide structural variety; the "atoms" of combinatorial chemistry. Quality and availability are critical. | Commercially available sets of carboxylic acids, amines, boronic acids, heterocyclic cores. Sourced from in-stock inventories for speed [63]. |
| Robust Coupling Reagents | Enable high-yielding, reliable bond formations with minimal side products. | Amide Coupling: HATU, DIC, T3P. Cross-Coupling: Pd catalysts for Suzuki-Miyaura, Buchwald-Hartwig reactions [63]. |
| Solid Supports & Linkers | Essential for solid-phase combinatorial synthesis (e.g., OBOC, parallel synthesis). Allows for reaction driving and simplified purification. | Resins (Wang, Rink amide), cleavable linkers sensitive to TFA, light, or other specific conditions [62]. |
| DNA-Compatible Reagents | Specialized for DNA-Encoded Library (DEL) synthesis. Reactions must proceed in aqueous buffer without damaging the oligonucleotide tag. | Water-soluble catalysts, mild reducing agents, and bio-orthogonal reaction pairs (e.g., click chemistry) [62]. |
| Specialty & Sustainable Solvents | Medium for reaction execution. Shift towards green chemistry principles and safer solvents is growing. | Green Solvents: Cyrene, 2-MeTHF. Traditional: DMF, DMSO, acetonitrile. Considerations for waste reduction and operator safety are increasing [64] [65]. |
Innovation Frontiers: AI and Sustainable Design
The field of library design is being transformed by two converging trends: the integration of advanced artificial intelligence and a growing imperative for sustainable and safe-by-design chemistry.
- AI-Driven De Novo Design and Screening: Machine learning models are now used to predict molecular properties, synthetic pathways, and target activity with increasing accuracy [66] [65]. Generative AI models can design novel molecules with optimized multi-property profiles, while ultra-large virtual screening platforms can dock billions of compounds against a protein target in silico, identifying promising virtual hits for synthesis [66]. These technologies significantly compress the early discovery timeline.
- Safe and Sustainable-by-Design (SSbD): Emerging from the European Green Deal, the SSbD framework encourages a lifecycle approach to chemical design, prioritizing human and environmental safety from the outset [65]. This influences library design by promoting the use of bio-based feedstocks (e.g., from algae), the elimination of hazardous substances, and the adoption of circular chemistry principles to minimize waste [64] [65]. This trend is moving from a regulatory consideration to a source of competitive advantage and innovation.
The following diagram conceptualizes how these advanced tools guide navigation through the multi-dimensional challenges of modern library design.
Diagram 2: Modern Navigation Tools for Chemical Space. Advanced computational tools (AI/ML, ultra-large screening) and new design frameworks (SSbD) address the multi-faceted challenge of finding high-quality leads, accelerating and de-risking the discovery process [66] [65].
Optimizing library design is no longer a one-dimensional problem of maximizing size. It is a sophisticated balancing act that integrates diversity (to access novel biology), drug-likeness (to ensure developmental potential), and synthetic feasibility (to guarantee practical realization). As comparative studies show, the chemical universe is too vast and regions too complementary for any single approach to dominate [13]. The future lies in strategically combining diverse sources—from natural product-inspired scaffolds to billions of make-on-demand virtual compounds—and leveraging computational advances like AI and ultra-large screening to intelligently navigate this space [66] [63]. Success in drug discovery will belong to those who best master this integrated, multi-objective optimization, efficiently translating expansive virtual chemical space into tangible, high-quality therapeutic candidates.
Integrating Negative Data and Dark Chemical Matter to Define BioReCS Boundaries
The concept of the Biologically Relevant Chemical Space (BioReCS) encompasses all molecules with a measurable biological effect, whether therapeutic, toxic, or promiscuous [1]. Accurately defining its boundaries is a fundamental challenge in modern drug discovery. A purely positive definition—focusing only on known active compounds—paints an incomplete picture and leads to inefficiency in screening and design.
Integrating negative data (experimentally confirmed inactive compounds) and dark chemical matter (compounds repeatedly showing no activity across many high-throughput screens) is critical for establishing these boundaries [1]. These data define the "non-biologically relevant" space, which is just as informative as the active regions. This guide compares strategies for mapping BioReCS, providing experimental protocols and performance data for methods that incorporate these essential negative constraints. The analysis is framed within the broader thesis of chemical space comparison, contrasting the landscapes of natural products, approved drugs, and combinatorial compounds.
Foundational Approaches to Mapping BioReCS Boundaries
Defining BioReCS requires specialized methodologies that can handle its scale, diversity, and the critical integration of negative data.
Key Databases and Data Types for Boundary Definition
Systematic study relies on curated data. The table below summarizes essential public databases that contribute positive, negative, and "dark" data to delineate BioReCS.
Table 1: Public Compound Databases for BioReCS Boundary Analysis
| Database | Primary Content/Region of BioReCS | Role in Boundary Definition | Key Feature |
|---|---|---|---|
| ChEMBL [1] | Annotated bioactive molecules (drug-like). | Defines core "active" space; source of poly-active/promiscuous compounds. | Extensive bioactivity data from literature. |
| PubChem [1] | Massive repository of chemical structures and bioassays. | Provides both active and inactive bioassay results; source for negative data. | Contains hundreds of millions of activity data points. |
| InertDB [1] | Curated experimentally inactive & AI-generated putative inactive molecules. | Directly defines "non-bioactive" chemical space boundaries. | Contains 3,205 curated and 64,368 AI-generated inactives. |
| Dark Chemical Matter (Corporate Collections) [1] | Compounds with no activity across numerous HTS campaigns. | Defines regions of high-probability inactivity; crucial for negative boundaries. | Large-scale, empirically derived negative data. |
| COCONUT (COlleCtion of Open Natural prodUcTs) [1] | Diverse natural products. | Represents the biologically pre-validated, complex region of BioReCS. | Excludes synthetics and derivatives. |
Experimental & Computational Protocols for Boundary Analysis
Protocol 1: Comparative Analysis of Ultra-Large Chemical Spaces This protocol, adapted from studies comparing billion-member combinatorial libraries, is essential for understanding coverage and overlap in synthetic regions of BioReCS [13].
- Space Selection: Choose two or more large, non-enumerable fragment-based chemical spaces (e.g., a corporate space like BICLAIM (>10²⁰ compounds) and a commercially accessible space like Enamine's REAL Space (~4 billion compounds)) [13].
- Query Panel Definition: Select a panel of 100 reference molecules. To focus on drug-relevant boundaries, filter approved drugs using standard "drug-like" property filters (e.g., MW < 600 Da, clogP < 6) [13].
- Similarity Searching: For each query, perform a similarity search (e.g., using the Feature Trees method) in each chemical space to retrieve the 10,000 nearest neighbors without full enumeration [13].
- Overlap & Complementarity Analysis: Compare the unique hit sets from each space using structural fingerprints (e.g., MDL public keys). Calculate the Tanimoto similarity within and between hit sets. The extremely low overlap (<0.01%) between spaces like BICLAIM, REAL, and public KnowledgeSpace highlights their complementarity [13].
- Feasibility Assessment: Apply synthetic feasibility scores (e.g., SAscore, rsynth) to the hit sets to compare the "actionability" of the defined boundary regions [13].
Protocol 2: Integrating Negative Data via Machine Learning This protocol uses machine learning to explicitly model the boundary between active and inactive regions.
- Dataset Curation: Compile a balanced dataset from sources like ChEMBL (actives) and InertDB or PubChem bioassays (confirmed inactives) [1]. Include dark chemical matter if available.
- Descriptor Calculation: Compute molecular descriptors or fingerprints (e.g., ECFP4, MAP4) suitable for the compound classes. The MAP4 fingerprint is noted for its generality across different ChemSpas [1].
- Model Training & Boundary Definition: Train a classification model (e.g., Support Vector Machine, Random Forest) to distinguish actives from inactives. The decision hyperplane of the model provides a computational definition of the BioReCS boundary in the chosen descriptor space.
- Visualization & Analysis: Project the data and the model's decision boundary into 2D using tools like ChemPlot (with PCA, t-SNE, or UMAP) to visually inspect the separation and identify outliers or boundary-cliff regions [67].
Protocol 3: Portal Learning for Exploring Dark Genomic Space Portal Learning is a specialized deep learning framework designed to predict bioactivity in uncharted "dark" regions (e.g., proteins with no known ligands), which is a key challenge in expanding BioReCS boundaries [68].
- Problem Framing: Define the "dark" prediction task, such as predicting ligands for an entire gene family excluded from the training data.
- Model Architecture & Training: Implement the PortalCG framework. Its three novel components address the Out-of-Distribution (OOD) problem: a) Step-wise Transfer Learning (simulating the biological sequence-structure-function paradigm), b) Out-of-Cluster Meta-Learning (improving generalization to novel clusters), and c) Stress Model Selection (selecting models robust to distribution shifts) [68].
- Benchmarking: Rigorously evaluate performance against state-of-the-art methods using metrics like PR-AUC and ROC-AUC in an out-of-gene-family validation setting.
- Application: Use the trained model to predict novel chemical-protein interactions for undrugged targets, thereby proposing an expansion of the known BioReCS [68].
Diagram 1: Workflow for Defining BioReCS Boundaries (77 characters)
Diagram 2: Portal Learning Framework for Dark Space (68 characters)
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Tools and Reagents for BioReCS Boundary Experiments
| Tool/Reagent Category | Specific Example/Product | Function in Experiment |
|---|---|---|
| Public Bioactivity Databases | ChEMBL, PubChem BioAssay [1] | Sources of positive and negative bioactivity data for model training and validation. |
| Negative Data Repositories | InertDB, Corporate Dark Chemical Matter (DCM) Collections [1] | Provide high-confidence inactive compounds to define negative boundaries of BioReCS. |
| Computational Chemistry Software | FTrees software, RDKit, MOE [13] | Perform similarity searches in fragment spaces, compute molecular descriptors, and assess synthetic feasibility (e.g., rsynth score). |
| Chemical Space Visualization | ChemPlot (Python library) [67] | Generates 2D projections of chemical space using PCA, t-SNE, or UMAP; incorporates tailored similarity for property-aware visualization. |
| Large Make-on-Demand Libraries | Enamine REAL Space, WuXi GalaXi [13] [47] | Representative, synthetically accessible ultra-large libraries for comparative analysis and validation of boundary definitions. |
| Specialized Machine Learning Frameworks | PortalCG implementation of Portal Learning [68] | Deep learning framework specifically designed to generalize predictions to dark chemical and biological space (OOD problem). |
Performance Comparison: Natural Products, Drugs, and Combinatorial Compounds
The utility of BioReCS boundaries is most evident when comparing distinct chemical subspaces. The following table and analysis contrast the key regions relevant to drug discovery.
Table 3: Comparison of Chemical Subspaces within BioReCS
| Property / Metric | Natural Products (NPs) | Approved Drugs (Small Molecule) | Combinatorial Compounds (e.g., REAL Space) |
|---|---|---|---|
| Structural Complexity | High (e.g., more chiral centers, fused rings) [28]. | Moderate, optimized for synthesis and bioavailability. | Deliberately varied, often lower complexity by design. |
| Chemical Space Coverage | Occupy a specific, privileged region of BioReCS; high scaffold diversity [28]. | Cover a well-defined "drug-like" subspace (e.g., Lipinski's space). | Designed for maximal coverage of accessible, synthesizable space; extremely broad [13]. |
| Biological Pre-validation | Inherently high (evolutionarily selected for biological interaction) [28]. | Very high (clinically validated). | Low to none (requires screening to discover activity). |
| Role in Boundary Definition | Define the "active" boundary of complex, biologically relevant shapes. | Define the central, optimized "core" of therapeutic BioReCS. | Help map the outer, synthetically feasible perimeter of BioReCS; major source of negative data/DCM. |
| Synthetic Feasibility | Often low; can be challenging and costly to synthesize or modify [28]. | High (by necessity for manufacturing). | Very high (designed for rapid, reliable synthesis on-demand) [13]. |
| Overlap with Other Spaces | Limited direct scaffold overlap with typical combinatorial libraries, inspiring new chemotypes [28]. | Significant overlap with corporate screening libraries from which they were discovered. | Minimal overlap (<0.01%) between different ultra-large combinatorial spaces, indicating high complementarity [13]. |
Performance Insights from Comparative Analysis: A landmark study comparing ultra-large combinatorial spaces (BICLAIM, REAL Space, KnowledgeSpace) using a probe-based method found a strikingly low overlap of hit sets—only three compounds were common to all three spaces from searches based on 100 drug queries [13]. This demonstrates that even within the synthetic region of BioReCS, different strategies populate vastly different territories. This complementarity is a key performance metric: a well-defined boundary strategy should guide researchers to the most productive, unexplored region for their target.
For exploring dark biological space (e.g., proteins with no ligands), the Portal Learning (PortalCG) framework demonstrated superior performance. In rigorous benchmarks predicting ligand binding to out-of-cluster gene families, it outperformed AlphaFold2-based docking by 79% in PR-AUC and 27% in ROC-AUC, and significantly beat other state-of-the-art ligand prediction methods [68]. This shows that advanced ML methods integrating biological paradigms are high-performing tools for expanding the known boundaries of BioReCS into truly novel territory.
Defining the boundaries of the Biologically Relevant Chemical Space is not an academic exercise but a practical necessity for efficient drug discovery. As evidenced, integrating negative data and dark chemical matter is paramount to constructing meaningful boundaries. Performance comparisons reveal that chemical subspaces (natural products, drugs, combinatorial libraries) are highly complementary, and strategies like Portal Learning show exceptional promise in probing the "dark" regions beyond current knowledge.
The future of BioReCS mapping lies in the continued curation of high-quality negative data, the development of universal molecular descriptors capable of handling the full spectrum of chemical matter (including metallodrugs and macrocycles) [1], and the integration of generative AI models trained on both positive and negative boundaries to propose novel compounds with a higher prior probability of bioactivity. Researchers should adopt a hybrid strategy, using comparative analyses to select optimally complementary screening libraries and employing advanced ML frameworks to rationally explore the vast uncharted spaces that remain.
Comparative Landscapes: Validating Structural Diversity and Therapeutic Potential
The systematic exploration of chemical space is a foundational challenge in modern drug discovery. This space, encompassing all possible organic molecules, is vast, estimated to exceed 10^60 compounds, making exhaustive exploration impossible [35]. Consequently, researchers must navigate and sample this space strategically. Three primary sources dominate this endeavor: Natural Products (NPs), refined by evolution for biological interaction; approved Drugs, which represent chemical success stories; and synthetic Combinatorial Libraries, designed for breadth and efficiency [69]. A critical thesis in contemporary research posits that these three classes occupy distinct yet complementary regions of chemical space, and that a comparative, quantitative understanding of their diversity is essential for guiding future molecular discovery [44] [70].
Historically, NPs have been an unparalleled source of novel drug leads, with a significant percentage of approved small-molecule drugs originating directly or indirectly from natural scaffolds [28]. However, the rise of high-throughput screening (HTS) in the late 20th century shifted focus towards large combinatorial libraries of synthetic compounds (SCs), under the assumption that sheer numbers would yield success [44]. This shift often failed to deliver expected productivity, partly due to the limited structural diversity and biological relevance of many synthetic libraries compared to the evolved complexity of NPs [44] [69]. This historical context frames the central question: How can we objectively measure and compare the diversity of these compound classes to inform better library design and screening strategies?
This guide provides a comparative analysis grounded in recent chemoinformatic research. It moves beyond qualitative assessment to present quantitative metrics, standardized experimental protocols, and visualization tools for directly comparing NPs, drugs, and combinatorial libraries. The goal is to equip researchers with a practical toolkit for quantifying diversity, thereby enabling more informed decisions in library design, purchase, and screening campaigns to efficiently probe biologically relevant chemical space.
Quantitative Comparison of Structural and Property Space
A direct comparison of fundamental molecular properties reveals consistent, statistically significant differences between NPs, drugs, and typical combinatorial compounds. The following tables synthesize data from comprehensive time-dependent analyses and comparative studies [44] [28] [71].
Table 1: Comparative Analysis of Key Physicochemical Properties
| Property | Natural Products (NPs) | Approved Drugs | Combinatorial Library Compounds (Typical) | Implication for Discovery |
|---|---|---|---|---|
| Molecular Weight | Higher (~400-500 Da), increasing over time [44]. | Moderate, often compliant with Rule of 5 [28]. | Generally lower, tightly constrained by design rules [44] [71]. | NPs explore "beyond Rule of 5" space, relevant for complex targets like protein-protein interactions [28]. |
| Number of Rings | Higher, with more non-aromatic and fused ring systems [44]. | Moderate. | Lower, with a higher proportion of aromatic rings [44]. | NP ring systems are more complex and three-dimensional, contributing to structural novelty. |
| Oxygen Atoms | Significantly higher count [44]. | Moderate. | Lower. | Higher oxygen content relates to hydrogen-bonding capacity and polarity, influencing target engagement. |
| Nitrogen Atoms | Lower count [44]. | Moderate. | Higher count [44]. | Reflects synthetic accessibility of amine-containing building blocks in combinatorial chemistry. |
| Chiral Centers | High density of stereogenic centers [69]. | Variable, often contain at least one. | Deliberately minimized in many libraries. | Defines precise 3D shape; critical for specificity but complicates synthesis. |
| Lipophilicity (LogP) | More hydrophobic on average, trend increasing over time [44]. | Optimized for oral bioavailability. | Often designed within a specific, narrow logP range [71]. | Impacts membrane permeability and solubility; NPs may require derivatization for drug-likeness. |
Table 2: Diversity Metrics and Chemical Space Occupancy
| Metric | Natural Products (NPs) | Combinatorial Libraries | Analysis Method & Significance |
|---|---|---|---|
| Scaffold Diversity | High. Contain a vast array of unique, complex core structures [44] [69]. | Lower. Often explore many derivatives around a limited set of simple scaffolds [69]. | Measured via Bemis-Murcko scaffold analysis. High scaffold diversity increases chance of novel hit discovery. |
| Functional Group Diversity | Rich in complex ethers, alcohols, glycosides; fewer halogens [44]. | Rich in amides, sulfonamides, aryl halides, ureas [44]. | Fragment-based analysis (e.g., RECAP). Reflects different biochemical vs. synthetic origins. |
| Biological Relevance | High. Evolved to interact with biological macromolecules [69]. | Variable. Can be biased towards synthetic accessibility over bio-relevance [44]. | Assessed via hit rates in phenotypic or target-based assays. NPs show historically higher success rates as drug leads [28]. |
| Synthetic Accessibility | Generally low due to complexity. | Designed for high accessibility. | A key practical constraint. NP-inspired libraries aim to balance complexity with synthetic tractability [69]. |
| Temporal Diversity Trend | Expanding into new, complex regions of space over time [44]. | Cardinality grows, but intrinsic diversity may plateau without deliberate design [35]. | Quantified via time-series analysis of databases. Mere growth in library size does not guarantee diversity increase [35]. |
Experimental Protocols for Diversity Quantification
To reproducibly compare compound libraries, standardized computational protocols are essential. The following methodologies are widely adopted in cheminformatics.
Protocol for Time-Dependent Chemoinformatic Analysis
This protocol, based on the work of Liu et al. (2024), is designed to trace the evolution of chemical space for different compound classes over time [44].
- Data Curation & Chronological Sorting: Compile datasets from structured databases (e.g., Dictionary of Natural Products for NPs, ChEMBL or ZINC for synthetic compounds). Sort molecules strictly by their date of first report or CAS registry number.
- Molecular Standardization: Process all structures using a toolkit like RDKit to remove salts, standardize tautomers, and neutralize charges. Generate canonical SMILES strings.
- Descriptor Calculation: For each molecule, calculate a suite of 30-40 relevant physicochemical descriptors (e.g., molecular weight, heavy atom count, number of rotatable bonds, rings, H-bond donors/acceptors, topological polar surface area, LogP).
- Time-Slice Grouping: Divide the sorted list into sequential groups of equal size (e.g., 5,000 molecules per group). Each group represents a specific time period.
- Statistical Analysis: For each descriptor and within each time slice, calculate the population mean, median, and distribution. Plot these trends over time to visualize property evolution.
- Chemical Space Visualization: Apply dimensionality reduction techniques (e.g., Principal Component Analysis - PCA) to the descriptor matrix for each time slice. Visualize the occupancy and drift of chemical space using 2D or 3D scatter plots, with different colors for NPs and SCs.
Protocol for Assessing Library Diversity Using the iSIM Framework
This protocol leverages the efficient iSIM method to quantify the intrinsic diversity of a library or compare diversities between libraries, as detailed in recent methodological advances [35].
- Molecular Representation: Encode all molecules in the library(s) of interest into binary molecular fingerprints (e.g., Morgan fingerprints, RDKit fingerprints). The fingerprint length and type should be consistent for all comparisons.
- Calculate Intrinsic Similarity (iT): Use the iSIM algorithm to compute the global average pairwise Tanimoto similarity within a single library.
- The iT value is calculated directly from the fingerprint matrix K, where kᵢ is the count of "on" bits in the i-th fingerprint position across all N molecules:
iT = Σ[kᵢ(kᵢ-1)/2] / Σ[kᵢ(kᵢ-1)/2 + kᵢ(N-kᵢ)][35]. - A lower iT value indicates higher internal diversity.
- The iT value is calculated directly from the fingerprint matrix K, where kᵢ is the count of "on" bits in the i-th fingerprint position across all N molecules:
- Identify Structural Medoids and Outliers: For each molecule, compute its complementary similarity (the iT of the library after removing that molecule).
- Molecules with the lowest complementary similarity are central "medoids."
- Molecules with the highest complementary similarity are peripheral "outliers."
- Compare Library Releases (Temporal Analysis): Apply Steps 1-3 to sequential releases of a database (e.g., ChEMBL versions 1-33). Plot iT over releases to assess if diversity grows with size. Analyze the Jaccard similarity between the medoid/outlier sets of different releases to see how the core and periphery of the chemical space evolve [35].
- Cross-Library Comparison: Calculate the iT for different types of libraries (e.g., an NP library vs. a combinatorial library). The library with the lower iT is more diverse in the context of the chosen fingerprint representation.
Protocol for Diversity-Oriented Library Design Informed by NPs
This protocol outlines the design of combinatorial libraries that capture the diversity and biological relevance of NPs while maintaining synthetic feasibility [69].
- NP Scaffold Deconstruction & Privileged Fragment Identification: Analyze a large NP database to identify recurring, biologically relevant core scaffolds (e.g., macrocycles, complex polycycles) and side-chain fragments using scaffold network analysis and retrosynthetic fragmentation rules (RECAP).
- Simplify and Synthesize: Chemically simplify the identified privileged NP scaffolds to retain key 3D structural and functional group elements while removing excessive complexity that hinders synthesis.
- Diversity-Oriented Synthesis (DOS) Planning: Plan synthetic routes that introduce significant skeletal and stereochemical diversity from common intermediates. Aim to generate multiple distinct core scaffolds from a single starting material, rather than many analogs of a single core.
- Property-Based Filtering: Filter the virtually enumerated library using calculated physicochemical properties to ensure a degree of "drug-likeness" or "lead-likeness," while allowing for properties characteristic of NPs (e.g., higher MW, logP).
- Diversity Validation: Before full-scale synthesis, validate the projected diversity of the designed library using the iSIM protocol (3.2) or by comparing its projected chemical space occupancy via PCA with that of standard combinatorial libraries and known NPs.
Diagram 1: Workflow for Time-Dependent Chemical Space Analysis [44]
Diagram 2: The iSIM Framework for Diversity Quantification [35]
The Scientist's Toolkit: Research Reagent Solutions for Diversity Analysis
Table 3: Essential Databases, Software, and Tools
| Resource Name | Type | Primary Function in Diversity Analysis | Key Feature / Relevance |
|---|---|---|---|
| Dictionary of Natural Products (DNP) | Database | The definitive reference for NPs. Serves as the primary data source for time-dependent and property-based comparisons of NPs [44]. | Contains over 300,000 entries with extensive structural and source information. |
| ChEMBL | Database | A large, curated database of bioactive drug-like molecules. Used as a source for synthetic compounds and drugs, and for temporal release analysis [35]. | Manually extracted bioactivity data from literature; multiple versioned releases enable time-series study. |
| RDKit | Software (Cheminformatics) | Open-source toolkit for descriptor calculation, fingerprint generation, molecular standardization, and basic visualization [44] [35]. | The workbench for executing most protocols in Python. |
| iSIM & BitBIRCH Algorithms | Algorithm | Core methods for efficiently calculating the intrinsic diversity (iSIM) and performing clustering (BitBIRCH) on ultra-large libraries [35]. | Enable O(N) scaling for diversity analysis, making billion-molecule library analysis feasible. |
| COCONUT | Database | An open and comprehensive collection of NPs. Useful for building NP-focused screening libraries and fragment sets [44]. | Freely available, facilitates the creation of diverse NP-subset libraries. |
| ChemGPS-NP | Tool | A tool for navigating chemical space. Positions new molecules relative to maps defined by drugs and NPs [70]. | Helps identify if a compound library occupies regions close to drugs, NPs, or unexplored territory. |
| Enamine REAL / ZINC | Database | Commercial/Academic databases of readily available or make-on-demand synthetic compounds. Represent the "combinatorial library" space for virtual screening [44]. | Provide a real-world benchmark for the chemical space of modern synthetic libraries. |
Integrating Findings into a Broader Thesis on Chemical Space
The quantitative data and methods presented here directly support a broader, evolving thesis in chemical biology and drug discovery: that deliberate, metric-driven integration of NP-like complexity into synthetic library design is crucial for expanding into biologically relevant but underexplored regions of chemical space.
The evidence shows a divergence: NPs continue to evolve into larger, more complex, and more hydrophobic territories, driven by advances in isolation technology and representing a continuous source of novel scaffolds [44]. In contrast, the chemical space of many synthetic combinatorial libraries, while enormous in cardinality, risks stagnation in diversity—growing in size without substantially expanding its boundaries, a phenomenon detectable with tools like iSIM [35]. Approved drugs often occupy a strategic overlap, embodying a compromise between NP-inspired bioactivity and synthetic tractability [28].
This thesis reframes the role of combinatorial chemistry. Rather than being an alternative to NPs, its most powerful application may be in the systematic exploration of "pseudo-natural product" space—generating novel architectures that are inspired by NP motifs but inaccessible through biosynthesis [44] [69]. The success of NP-inspired libraries in yielding chemical probes and leads for challenging targets validates this approach [69]. Future research, powered by AI-driven generative chemistry and the quantitative metrics described here, will focus on consciously designing libraries that maximize not just size, but measured diversity and predicted biological relevance, thereby bridging the gap between the efficiency of synthesis and the evolved wisdom of nature [72] [51].
This comparison guide provides a quantitative and methodological framework for analyzing the convergent and divergent regions of chemical space occupied by natural products (NPs), approved drugs, and combinatorial compounds (CCs). Framed within a broader thesis on chemical evolution and library design, the guide details experimental cheminformatics protocols for mapping these territories, supported by current data on structural diversity, physicochemical properties, and biological relevance. The analysis reveals that while combinatorial libraries offer unparalleled size and synthetic accessibility, natural products occupy distinct regions characterized by greater structural complexity and validated biological relevance, creating unique opportunities for library design and drug discovery.
The concept of "chemical space," defined as the multi-dimensional descriptor space encompassing all possible molecules, serves as the foundational framework for comparing compound origins in drug discovery [73]. A prevailing thesis in modern research posits that the historical evolutionary pressures on natural products (NPs) have shaped a chemical space uniquely enriched for biological function, while combinatorial chemistry explores vast, synthetically accessible regions [44]. The intersection—where these spaces converge—often yields promising drug-like candidates, while their divergent territories highlight unexplored opportunities for innovation [74]. This guide objectively compares these domains using public database analytics, clustering algorithms, and dimensionality reduction techniques, providing researchers with a roadmap for navigating this complex landscape.
Methodological Framework for Chemical Space Comparison
The comparative analysis relies on cheminformatics tools designed to handle large-scale data. Key methodologies include:
- iSIM (Intrinsic Similarity) Framework: This tool calculates the average pairwise Tanimoto similarity within a library in O(N) time, bypassing the traditional O(N²) scaling. It provides a global internal diversity metric (iT), where lower iT values indicate greater diversity [35].
- BitBIRCH Clustering: An adaptation of the BIRCH algorithm for binary fingerprints, this method enables efficient clustering of ultra-large libraries (e.g., billions of compounds) based on Tanimoto similarity, allowing for granular analysis of chemical space formation [35].
- Generative Topographic Mapping (GTM) & CoLiNN: GTM is a dimensionality reduction technique that creates a fuzzy, interpretable 2D projection of chemical space. The Combinatorial Library Neural Network (CoLiNN) predicts a compound's position on a GTM map using only building block and reaction information, eliminating the need for full library enumeration—a critical advancement for analyzing virtual combinatorial spaces [32].
- Descriptor Analysis: Comparative studies calculate a suite of physicochemical properties (e.g., molecular weight, logP, ring counts, fraction of sp³ carbons) and generate molecular scaffolds/fragments to assess structural complexity and evolution over time [44].
Diagram: Chemical Space Analysis & Comparison Workflow
Comparative Analysis of Chemical Territories
Structural and Physicochemical Landscapes
A time-dependent analysis of NPs from the Dictionary of Natural Products and synthetic compounds (SCs) from major databases reveals distinct evolutionary paths and property distributions [44].
Table 1: Time-Evolving Physicochemical Property Comparison (NPs vs. Synthetic Compounds) [44]
| Property | Natural Products (NPs) Trend | Synthetic Compounds (SCs) Trend | Implication for Convergence |
|---|---|---|---|
| Molecular Size (Weight, Volume) | Consistent increase over time; larger than SCs. | Constrained variation within a "drug-like" range. | NPs explore larger, more complex regions; SCs fill a central, lead-like space. |
| Ring Systems | Increase in non-aromatic, fused rings (e.g., bridged, spiral) and sugar moieties. | Higher prevalence of aromatic rings (e.g., benzene derivatives). | NPs contribute complex, saturated scaffolds; SCs dominate flat, aromatic architectures. |
| Structural Complexity (fsp³, Chiral Centers) | Higher fraction of sp³ carbons and more chiral centers. | Generally lower fsp³ and fewer chiral centers. | NPs occupy more 3D-shaped territory; SCs are often flatter and less complex. |
| Hydrophobicity (LogP) | Tendency to increase over time. | More tightly regulated around optimal values. | NP space includes more hydrophobic extremes. |
Table 2: Chemical Space Diversity Metrics Across Public Libraries [35] [44]
| Library / Compound Class | Key Diversity Finding (via iSIM/Clustering) | Biological Relevance Proxy |
|---|---|---|
| ChEMBL (Bioactive Compounds) | High internal diversity; growth in size does not always equate to increased diversity [35]. | Directly derived from bioactivity data. |
| Natural Products (Dictionary of NP) | High and increasing scaffold diversity; less concentrated chemical space than SCs [44]. | Inherently evolved to interact with biomolecules. |
| Synthetic/Combinatorial Libraries | Can achieve enormous size (>10²⁶ virtually), but diversity is dependent on building block choice [32]. | Often lower hit rates in phenotypic screening, indicating potential relevance gap. |
| Approved Drugs | Occupy a constrained subspace at the intersection of NP-like complexity and SC-like synthesizability. | Validated therapeutic efficacy and safety. |
The Convergence: Drug Space
Approved drugs are not uniformly distributed but cluster at the intersection of accessible synthetic space and biologically relevant NP-like space. They often exhibit a hybrid profile: moderate molecular weight and logP (following drug-like rules) but incorporate structural motifs and complexity features (like chiral centers and fused ring systems) reminiscent of NPs [44]. This convergent region is a primary target for pseudo-natural product design, which combines NP fragments through novel synthetic linkages to explore biologically relevant yet unprecedented chemical territory [44].
The Divergence: Unique Territories
- Unique NP Territory: Characterized by high structural complexity (e.g., macrocycles, polycyclic systems with many stereocenters), oxygen-rich functional groups, and extreme physicochemical property values (very high or low logP) [44]. These regions are often under-explored by commercial screening libraries.
- Unique Combinatorial Territory: Defined by high synthetic accessibility and architectures built from common aromatic building blocks. This space contains vast numbers of novel, often flat, nitrogen- and halogen-rich compounds that do not resemble natural products [62] [32]. A risk here is the proliferation of structures that are easy to make but have low probability of biological activity.
Diagram: Time-Evolution of NP and Synthetic Chemical Spaces
Experimental Protocols for Key Analyses
Protocol 1: Assessing Library Diversity and Evolution Using iSIM & BitBIRCH
Objective: Quantify the internal diversity and time-evolution of a chemical library (e.g., sequential ChEMBL releases) [35].
- Data Curation: Obtain successive releases of a standardized database (e.g., ChEMBL 1-33). Isolate specific compound sets (e.g., all entries, NPs only).
- Fingerprint Generation: Calculate fixed-length molecular fingerprints (e.g., ECFP4) for all compounds in each release.
- iSIM Calculation: For each library release, compute the iT (iSIM Tanimoto) value using the column-sum method on the fingerprint matrix. A decreasing iT over releases indicates increasing diversity.
- Complementary Similarity: Calculate the complementary similarity for each molecule (iT of the set with the molecule removed). Identify medoids (lowest 5% complementary similarity) and outliers (highest 5%) for each release.
- Temporal Analysis: Use the Set Jaccard Index to measure the overlap between medoid/outlier sets from different releases, tracking the stability or shift of the library's core and periphery.
- BitBIRCH Clustering: Apply the BitBIRCH algorithm to the fingerprint data of the combined releases to cluster compounds. Analyze the birth of new clusters in later releases to identify newly explored chemical regions.
Protocol 2: Mapping Combinatorial Library Space Without Enumeration Using CoLiNN
Objective: Visualize and compare the chemical space of ultra-large virtual combinatorial libraries without computationally expensive full enumeration [32].
- Library Definition: Define the combinatorial library by its set of building blocks (BBs) and the reaction scheme connecting them.
- Building Block Processing: Standardize all BBs (e.g., using ChemAxon Standardizer) and compute descriptors or fingerprints for each.
- Model Application: Input the BB descriptors and encoded reaction scheme into a pre-trained CoLiNN model. CoLiNN uses neural networks to create embeddings for BBs and reactions, combining them to predict the "responsibility vector" for any potential product.
- Space Visualization: The predicted responsibility vectors for a representative sample of library products are aggregated to generate a Generative Topographic Map (GTM). The density on the map shows the library's coverage.
- Library Comparison: Overlay the GTMs of different combinatorial libraries (or a library vs. a reference like ChEMBL) to visually assess overlap and unique coverage, guiding library design towards under-explored or biologically relevant regions.
Diagram: Combinatorial Library Design & Analysis via CoLiNN
Table 3: Key Resources for Chemical Space Comparison Research
| Resource / Reagent | Type | Primary Function in Research | Source / Example |
|---|---|---|---|
| ChEMBL Database | Curated Bioactivity Database | Provides a benchmark set of drug-like and bioactive molecules for diversity comparison and relevance assessment [35]. | https://www.ebi.ac.uk/chembl/ |
| Dictionary of Natural Products (DNP) | Natural Product Database | Serves as the definitive source for NP structures for time-series and property analysis against synthetic compounds [44]. | CRC Press / Taylor & Francis |
| Enamine REAL / GSK XXL Space | Virtual Combinatorial Library | Represents ultra-large (billions to 10²⁶) synthetically accessible chemical spaces for exploration and comparison [32]. | Enamine Ltd.; GSK |
| RDKit or ChemAxon Toolkits | Cheminformatics Software | Open-source or commercial libraries for standardizing molecules, calculating descriptors, generating fingerprints, and applying algorithms. | rdkit.org; chemaxon.com |
| iSIM & BitBIRCH Algorithms | Computational Methodology | Core tools for efficient diversity calculation and clustering of massive libraries, as implemented in research code [35]. | Published algorithms (e.g., in [35]) |
| Commercially Available Building Blocks | Chemical Reagents | The foundational units for designing and synthesizing combinatorial libraries. Diversity and properties dictate the resulting library's chemical space [62] [32]. | eMolecules, Enamine, Sigma-Aldrich |
| DNA-Encoded Library (DEL) Kits | Synthetic & Screening Technology | Enables the experimental synthesis and affinity-based screening of vast combinatorial libraries (up to 10¹² compounds) for hit identification [62]. | Various Pharma/CRO Providers |
The journey from initial screening hits to clinically approved drug entities is a central challenge in modern pharmacology. This process is fundamentally governed by the exploration and exploitation of chemical space—the multidimensional universe of all possible organic compounds. Within this space, two primary continents exist: the evolutionarily refined realm of Natural Products (NPs) and the human-engineered domain of Synthetic and Combinatorial Compounds (SCs). NPs are characterized by greater structural complexity, three-dimensionality, and biological pre-validation, having evolved to interact with biological systems [44]. In contrast, SCs, guided by design principles like Lipinski’s Rule of Five, often occupy a more confined region of chemical space optimized for synthetic accessibility and predicted oral bioavailability [24].
This guide objectively compares contemporary strategies for navigating these chemical spaces to identify and optimize drug leads. It evaluates three parallel approaches: the rationalization of NP libraries using advanced metabolomics, the application of computational Bayesian models to focus synthetic libraries, and the multiparametric optimization of combinatorial hit series. The thesis posits that the integration of NP-inspired structural diversity with the precision and scalability of combinatorial and computational chemistry yields the most efficient path to viable clinical candidates.
Comparative Experimental Protocols and Workflows
The following section details the core methodologies from seminal case studies, providing a direct comparison of experimental workflows.
Protocol 1: Rationalized Natural Product Library Screening [75] This protocol uses mass spectrometry to reduce library redundancy and increase hit rates.
- Library Preparation & MS Data Acquisition: A large library of crude or pre-fractionated natural product extracts (e.g., 1,439 fungal extracts) is prepared. Untargeted Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) data is acquired for all library members.
- Molecular Networking & Scaffold Identification: MS/MS spectral data is processed through the GNPS platform to create a molecular network. Spectra are clustered into molecular families (scaffolds) based on fragmentation pattern similarity, correlating to structural similarity.
- Rational Library Design: Custom algorithms (e.g., in R) analyze the scaffold distribution. The algorithm iteratively selects the extract that adds the greatest number of new, unique scaffolds not yet represented in the growing rational sub-library, until a target scaffold diversity coverage (e.g., 80%, 95%, 100%) is achieved.
- Bioactivity Screening: The rationally designed sub-library and the full library are screened in parallel against relevant phenotypic (e.g., Plasmodium falciparum, Trichomonas vaginalis) and target-based (e.g., neuraminidase enzyme) assays.
- Hit Analysis & Dereplication: Bioactive extracts are analyzed, and features (unique m/z-retention time pairs) correlated with activity are identified. MS/MS data from hits is used to dereplicate known compounds and flag novel bioactive scaffolds.
Protocol 2: Computational Enrichment for Synthetic Library Screening [76] This protocol uses machine learning to prioritize compounds from commercial sources for anti-tuberculosis activity.
- Model Training Data Aggregation: Publicly available high-throughput screening (HTS) data against Myobacterium tuberculosis (Mtb) are aggregated (e.g., from the MLSMR, TAACF datasets). This includes both active and inactive compound structures with associated IC90/IC50 and mammalian cell cytotoxicity (e.g., Vero cells) data.
- Dual-Event Bayesian Model Development: A Bayesian machine learning model is trained to predict two linked properties: a) Potency against Mtb (IC90 < 10 μM), and b) Selectivity (Selectivity Index, SI = CC50/IC90 > 10). The model learns structural fingerprints associated with this desirable activity profile.
- Hit Series Clustering & Scaffold Selection: Active compounds from historical screens are clustered using cheminformatics tools (e.g., LeadScope). Common cluster scaffolds with high enrichment ratios (prevalence in actives vs. full library) are selected for follow-up.
- Commercial Analog Selection & Prioritization: Commercially available analogs of the selected cluster scaffolds are identified. These virtual compounds are scored using the pre-built Bayesian model.
- Focused Experimental Testing: The top-ranked compounds by Bayesian score are acquired and tested experimentally for Mtb inhibition and cytotoxicity. The hit rate from this computationally prioritized set is compared to that of random selection.
Protocol 3: Multiparametric Hit-to-Lead Optimization [77] This protocol involves the synthetic expansion and profiling of a combinatorial hit series for Chagas disease.
- Hit Identification & SAR Expansion: A confirmed hit compound (e.g., a 2-aminobenzimidazole) from phenotypic screening is used as a starting point. A library of 277 derivatives is designed and synthesized, systematically varying substituents to explore Structure-Activity Relationships (SAR).
- Primary Potency & Cytotoxicity Screening: All compounds are tested in a high-content phenotypic assay against intracellular Trypanosoma cruzi amastigotes to determine IC50. Parallel screening in mammalian host cells (e.g., Vero cells) determines CC50 for selectivity index (SI) calculation.
- In Vitro ADME Profiling: Key compounds showing potent anti-parasitic activity and acceptable selectivity are advanced to early ADME assays. This includes measuring microsomal stability (rat/human liver microsomes), kinetic solubility, and lipophilicity (ChromLogD).
- Multiparametric Optimization Analysis: Data for potency, selectivity, and ADME properties are analyzed concurrently. Compounds are evaluated in multi-dimensional space to identify leads that balance all key parameters, rather than just optimizing for peak potency.
- Lead Candidate Selection & In Vivo Rationale: The best-balanced compounds are selected based on a combined profile of high potency (IC50 < 0.3 μM), high SI, acceptable metabolic stability, and solubility. The final decision to progress to in vivo efficacy models is based on this integrated profile.
Table 1: Comparison of Key Experimental Protocols
| Protocol Feature | Rationalized NP Screening [75] | Computational Bayesian Enrichment [76] | Multiparametric Hit-to-Lead [77] |
|---|---|---|---|
| Starting Point | Large, redundant extract library | Historical HTS data & commercial catalogs | A single confirmed hit compound |
| Core Technology | LC-MS/MS & Molecular Networking | Bayesian Machine Learning & Clustering | Med. Chem. Synthesis & In Vitro Pharmacology |
| Primary Goal | Maximize scaffold diversity & hit rate | Enrich for active, non-toxic chemotypes | Optimize potency, selectivity & ADME simultaneously |
| Key Output | A minimized, diversity-maximized library | A prioritized list of compounds for testing | A refined lead candidate with balanced properties |
| Resource Intensity | High upfront analytical; lower screening | Low cost computational; focused testing | High synthetic & biological testing effort |
Figure 1: Comparative workflows for three major hit-finding and optimization strategies.
Performance Data: Hit Rates, Chemical Properties, and Success Metrics
The efficacy of each strategy is quantified through key performance indicators such as hit rate enrichment, compound property optimization, and progression to clinical trials.
Table 2: Hit Rate Comparison: Rationalized NP vs. Bayesian-Enriched Screening
| Screening Approach | Library Size | Hit Rate vs. P. falciparum | Hit Rate vs. T. vaginalis | Hit Rate vs. Neuraminidase |
|---|---|---|---|---|
| Full NP Library (Baseline) [75] | 1,439 extracts | 11.26% | 7.64% | 2.57% |
| 80% Scaffold Diversity Library [75] | 50 extracts | 22.00% | 18.00% | 8.00% |
| Random Selection (50 extracts) [75] | 50 extracts | 8-14% (range) | 4-10% (range) | 0-2% (range) |
| Bayesian-Prioritized Testing [76] | 550 compounds tested | 22.5% (vs. Mtb) | – | – |
Table 3: Multiparametric Optimization of a 2-Aminobenzimidazole Series [77]
| Optimization Parameter | Initial Hit (Compound 1) | Optimized Lead Candidate | Improvement Factor / Goal |
|---|---|---|---|
| Potency (IC50 vs. T. cruzi) | ~1.0 µM | < 0.3 µM | > 3-fold increase |
| Selectivity Index (SI) | Low (<10) | Significantly improved | Target: SI > 10 |
| Microsomal Stability (Human) | Low clearance | Moderate to high stability | Increased half-life |
| ChromLogD (Lipophilicity) | High | Optimized to lower range | Target for solubility |
| Kinetic Solubility | Problematic (low) | Improved but remained a key liability | Critical barrier for in vivo progression |
Table 4: Structural Evolution of Natural vs. Synthetic Chemical Space [44]*
| Structural Property | Natural Products (NPs) Trend | Synthetic Compounds (SCs) Trend | Implication for Drug Discovery |
|---|---|---|---|
| Molecular Weight & Complexity | Increases over time | Constrained within "drug-like" range | NPs explore larger, more complex scaffolds. |
| Ring Systems | More non-aromatic, fused rings | More aromatic, simple rings | NP scaffolds offer greater 3D shape diversity. |
| Stereogenic Centers | Higher proportion | Lower proportion | NPs are more chiral, impacting binding specificity. |
| Chemical Space Coverage | More diverse, less dense | More densely packed in specific regions | NP libraries reduce redundancy in screening. |
Figure 2: The evolution of chemical space exploration in drug discovery, showing the convergence of natural product (NP) and synthetic compound (SC) strategies [44].
The Scientist's Toolkit: Essential Reagents and Platforms
Table 5: Key Research Reagent Solutions and Platforms
| Tool / Reagent | Function in Workflow | Exemplar Use Case / Purpose |
|---|---|---|
| LC-MS/MS System & GNPS Platform | Untargeted metabolomics; molecular networking based on MS2 spectral similarity. | Dereplication and scaffold-based diversity analysis of NP extract libraries [75]. |
| Liver Microsomes (Human/Rat) | In vitro assessment of metabolic Phase I stability. | Determining intrinsic clearance during early ADME profiling of lead compounds [77]. |
| Reporter Cell Lines (e.g., THP-1, HepG2) | Phenotypic screening and cytotoxicity assessment. | Evaluating anti-mycobacterial activity and host cell toxicity in parallel [76]. |
| Bayesian Machine Learning Software (e.g., CDD Models) | Building predictive dual-event (potency & toxicity) models from HTS data. | Enriching commercial compound selections for targeted screening campaigns [76]. |
| Zebrafish (Danio rerio) Models | In vivo phenotypic screening for efficacy, toxicity, and ADME in a whole organism. | Bridging in vitro and mammalian studies; high-throughput in vivo validation [78]. |
| X-ray Free Electron Laser (XFEL) | Serial femtosecond crystallography for structure determination with minimal radiation damage. | Enabling high-throughput drug screening and binding studies at physiological temperature [79]. |
| Fragment Library (Ro3-compliant) | Low molecular weight, low complexity compounds for Fragment-Based Drug Discovery (FBDD). | Identifying weak-binding motifs that can be optimized into high-affinity leads [24]. |
The relentless pursuit of novel therapeutic agents demands continuous innovation in how researchers explore chemical space. Historically, drug discovery has navigated a path from natural products (NPs), valued for their biological relevance and complexity, to vast combinatorial libraries designed for high-throughput screening [44]. Today, the paradigm is shifting again toward ultra-large, make-on-demand virtual libraries and generative artificial intelligence (AI) models [47]. This guide provides an objective comparison of these dominant modern approaches—ultra-large combinatorial libraries, generative AI-designed libraries, and traditional natural product collections—framed within the broader thesis of chemical space exploration. We evaluate their performance in terms of size, structural diversity, synthetic feasibility, and potential to yield novel bioactive hits, supported by recent experimental data and detailed methodologies.
Core Methodologies for Chemical Space Comparison
Comparing chemical libraries that can contain billions to trillions of virtual compounds requires specialized methodologies, as traditional pairwise similarity calculations are computationally impossible [13].
1.1 Query-Based Comparison of Ultra-Large Libraries A seminal 2019 study developed a novel protocol to compare three gigantic fragment-based chemical spaces: the corporate BICLAIM space (>10²⁰ products), the public KnowledgeSpace (~10¹⁴ products), and the commercial Enamine REAL Space (~4×10⁹ products) [13].
- Query Set: 100 marketed drugs, filtered for drug-like properties (e.g., MW < 600 Da, clogP < 6), served as reference points in chemical space [13].
- Search Technology: The Feature Trees (FTrees) method was used. This pharmacophore-based descriptor reduces molecules to nodes representing rings and functional groups, enabling efficient similarity searching in fragment spaces and identification of scaffold hops [13].
- Procedure: For each query drug, the 10,000 most similar molecules were retrieved from each of the three spaces using FTrees-FS search technology [13].
- Analysis: The resulting hit sets (~1 million compounds per space) were analyzed for overlap using structural keys. Chemical feasibility of hits was assessed using the SAscore (based on fragment frequency in PubChem) and the rsynth score (based on retrosynthetic analysis) [13].
1.2 Time-Dependent Analysis of Natural vs. Synthetic Compounds A 2024 study conducted a time-dependent chemoinformatic analysis to understand the structural evolution of NPs versus synthetic compounds (SCs) [44].
- Datasets: 186,210 NPs from the Dictionary of Natural Products and 186,210 SCs from 12 databases were sorted chronologically by their CAS Registry Numbers [44].
- Grouping: Molecules were divided into 37 sequential groups of 5,000 each for time-series analysis [44].
- Property Calculation: 39 physicochemical properties (e.g., molecular weight, ring counts) were computed for all molecules [44].
- Structural Analysis: Bemis-Murcko scaffolds, ring assemblies, side chains, and RECAP fragments were generated and compared across time groups to assess diversity and complexity [44].
- Chemical Space Mapping: Principal Component Analysis (PCA) and other visualization tools were used to characterize and compare the occupied chemical space of NPs and SCs over time [44].
Ultra-Large Chemical Space Comparison Workflow [13]
Performance Comparison: Ultra-Large, Generative, and Natural Product Libraries
The following tables synthesize quantitative data from comparative studies to evaluate the three main library paradigms.
Table 1: Library Scale & Scope
| Library Type | Exemplar / Source | Estimated Size | Key Characteristics | Source |
|---|---|---|---|---|
| Ultra-Large Combinatorial | BICLAIM (Corporate) | >10²⁰ virtual products | Built from scaffolds & side chains; focus on drug-like chemical space. | [13] |
| Enamine REAL Space | ~4×10⁹ products | Built from reliable reactions & in-stock building blocks; >80% synthesis success. | [13] | |
| Generative AI Library | Generative AI Models (VAEs, GANs, Diffusion) | Virtually Unlimited | De novo generation optimized for specific properties (binding, ADMET). | [80] |
| Natural Product Collection | Dictionary of Natural Products | ~1.1×10⁶ known compounds | Evolved through biological selection; high structural complexity. | [44] |
Table 2: Structural Diversity & Evolution (Time-Dependent Analysis)
| Structural Property | Trend in Natural Products (Over Time) | Trend in Synthetic Compounds (Over Time) | Interpretation & Impact |
|---|---|---|---|
| Molecular Size | Steady, significant increase (MW, volume) [44]. | Variation within a limited, drug-like range [44]. | NPs explore larger, more complex regions of chemical space, while SCs are constrained by design rules. |
| Ring Systems | Increase in rings, especially non-aromatic and fused rings [44]. | Increase in aromatic rings (e.g., benzene derivatives) [44]. | NPs exhibit greater stereochemical and scaffold complexity; SCs favor synthetically accessible flat aromatics. |
| Chemical Space | Becomes less concentrated, more diverse [44]. | Remains more concentrated [44]. | NP chemical space is expanding into unique regions, whereas SC space, while broad, is more densely packed. |
| Biological Relevance | Inherently high due to evolutionary selection. | Has declined over time [44]. | NP-inspired design can inject bio-relevant complexity into synthetic libraries. |
Table 3: Hit Discovery Potential & Feasibility
| Metric | Ultra-Large Combinatorial (REAL Space) | Generative AI Library | Natural Product Collection |
|---|---|---|---|
| Synthetic Feasibility | Very High. Designed for reliable, rapid synthesis (~3-4 weeks) [13]. | Variable. Requires post-generation synthetic planning & validation [80]. | Often Low. Complex structures can pose significant synthesis/optimization challenges [28]. |
| Hit Novelty (vs. known drugs) | Moderate. High diversity but based on known reactions & building blocks. | Potentially Very High. Can explore entirely novel scaffolds optimized for a target [80]. | High. Provides unique, evolutionarily refined scaffolds often dissimilar to synthetic libraries [44]. |
| Experimental Validation | Direct synthesis and testing of selected virtual hits is routine. | Requires physical synthesis of AI-designed molecules; growing number of preclinical validations [80]. | Requires isolation, characterization, and often subsequent simplification or derivatization [28]. |
The Generative AI Paradigm: Protocols and Performance
Generative AI represents a fundamental shift from searching existing libraries to creating optimized ones de novo.
3.1 Core Generative Model Architectures Key models include Variational Autoencoders (VAEs), Generative Adversarial Networks (GANs), autoregressive models, and denoising diffusion probabilistic models (DDPMs) [80]. These models learn the underlying distribution of chemical structures from data (e.g., known molecules, protein structures) and sample from this distribution to generate novel candidates [80].
3.2 Specialized LLMs for Chemistry Models like GAMES (Generative Approaches for Molecular Encodings) are fine-tuned Large Language Models (LLMs) that generate valid SMILES strings, treating molecular design as a language task [81]. This allows for the rapid creation of targeted libraries. For downstream analysis, specialized LLMs like DrugGPT integrate biomedical knowledge bases to provide evidence-based analysis of drug properties, interactions, and recommendations, reducing "hallucinations" [82].
3.3 Experimental Validation Workflow
- Training: Models are trained on large corpora of chemical structures (e.g., ZINC, ChEMBL) and/or protein sequences and structures [80].
- Conditioned Generation: Models are guided by predictive algorithms to optimize generated molecules for specific properties (e.g., target affinity via docking scores, favorable ADMET profiles) [80].
- Filtering & Synthesis: Generated molecules are filtered for synthetic accessibility. Top candidates are synthesized [80].
- In vitro/in vivo Testing: Compounds are tested experimentally, with results potentially fed back to refine the model [80].
Generative AI-Driven Molecular Design Workflow [80]
The Scientist's Toolkit: Key Reagents & Solutions
Table 4: Essential Research Tools for Modern Chemical Space Exploration
| Tool / Reagent | Category | Primary Function in Research | Example / Source |
|---|---|---|---|
| Feature Trees (FTrees) Software | Cheminformatics | Enables similarity searching and comparison of ultra-large, non-enumerated fragment spaces by using a pharmacophore-based descriptor. | [13] |
| SAscore | Computational Filter | Predicts synthetic accessibility of a molecule based on fragment frequency in PubChem and molecular complexity. | [13] |
| SMILES String | Data Format | Standard text-based representation of a molecular structure; the foundational language for AI/ML models in chemistry (e.g., GAMES LLM). | [81] |
| rsynth Score (MOE) | Computational Filter | Assesses synthetic feasibility via retrosynthetic analysis and reagent database lookup. | [13] |
| Enamine REAL Space | Ultra-Large Library | A commercially accessible, make-on-demand virtual library built from robust chemistry with high synthesis success rates. | [13] |
| Generative AI Models (e.g., DDPMs) | AI Platform | De novo design of molecules optimized for multi-parameter objectives (potency, selectivity, ADMET). | [80] |
| Knowledge-Grounded LLM (e.g., DrugGPT) | AI Analysis Tool | Provides evidence-based analysis of drug properties, interactions, and recommendations by grounding responses in medical knowledge bases. | [82] |
The comparative analysis reveals a complementary landscape. Ultra-large combinatorial libraries (e.g., Enamine REAL) offer an unparalleled resource of readily synthesizable compounds, providing a tangible bridge between virtual screening and experimental testing [13]. Generative AI libraries excel at targeted exploration, capable of venturing into novel, optimized regions of chemical space that may be underserved by existing libraries [80]. Natural product collections remain an irreplaceable source of biologically pre-validated complexity and unique scaffolds, whose structural insights can inspire both combinatorial and generative design [44].
Future-proofing discovery lies not in choosing a single approach but in developing integrative strategies. This includes using generative AI to design molecules that mimic the desirable complexity of NPs, employing ultra-large libraries to efficiently synthesize and test AI-generated ideas, and using advanced comparison methodologies to map the coverage and identify gaps in these expansive chemical spaces. The convergence of these technologies, powered by ever-improving AI and automation, is poised to create a more efficient and productive ecosystem for the next generation of drug discovery [80] [47].
Conclusion
The comparative analysis reveals that natural products, approved drugs, and combinatorial libraries occupy distinct yet complementary regions of the biologically relevant chemical space (BioReCS). Natural products offer evolutionarily validated complexity often suited for challenging targets, while combinatorial libraries provide vast synthetic accessibility. Approved drugs serve as a crucial validation benchmark. The future of drug discovery lies not in favoring one space over another, but in developing integrated, AI-powered strategies that intelligently navigate and hybridize these spaces. This includes leveraging sustainable sourcing for NPs, applying advanced diversity metrics to combinatorial design, and utilizing ultra-large virtual screening to uncover novel chemotypes that bridge these domains, ultimately accelerating the development of effective therapeutics[citation:2][citation:5][citation:10].
References
- 1. Frontiers | On the biologically relevant chemical space: BioReCS [https://www.frontiersin.org/journals/drug-discovery/articles/10.3389/fddsv.2025.1674289/full]
- 2. Exploring Chemical Space for Drug Discovery Using the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3447393/]
- 3. Frontiersin [https://www.frontiersin.org/journals/drug-discovery/articles/10.3389/fddsv.2025.1674289/xml]
- 4. Chemical Space – the Final Frontier for Drug Compounds? [https://www.criver.com/eureka/chemical-space-final-frontier-drug-compounds]
- 5. Natural product-derived compounds in clinical trials and ... [https://pubs.rsc.org/en/content/articlelanding/2025/np/d5np00031a]
- 6. Revitalizing natural products for sustainable drug Discovery [https://www.sciencedirect.com/science/article/abs/pii/S2352554125001809]
- 7. Choosing the Right Color Palette in Molecular Visualizations [https://blog.samson-connect.net/choosing-the-right-color-palette-in-molecular-visualizations-why-it-matters]
- 8. Simple tools for mastering color in scientific figures [https://www.molecularecologist.com/2020/04/23/simple-tools-for-mastering-color-in-scientific-figures/]
- 9. Natural products and combinatorial chemistry: back to the ... [https://www.sciencedirect.com/science/article/abs/pii/S1367593104000572]
- 10. Chemical Evolution of Natural Product Structure - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8895405/]
- 11. In Search of Beautiful Molecules: A Perspective on Generative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12458704/]
- 12. Natural Products as a Foundation for Drug Discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2813068/]
- 13. Comparison of Large Chemical Spaces - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC6792285/]
- 14. Exploring organic chemical space for materials discovery ... [https://www.nature.com/articles/s41467-025-65564-8]
- 15. Natural products and combinatorial chemistry: back to the ... [https://pubmed.ncbi.nlm.nih.gov/15183325/]
- 16. A review of high throughput and combinatorial ... [https://www.sciencedirect.com/science/article/abs/pii/S0013468611013648]
- 17. Computational design strategies for combinatorial libraries [https://www.sciencedirect.com/science/article/abs/pii/S1367593103000577]
- 18. Property distributions: differences between drugs, natural ... [https://pubmed.ncbi.nlm.nih.gov/12546556/]
- 19. Dynamic combinatorial chemistry directed by proteins and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12236296/]
- 20. Theoretical and experimental comparison of mobile phase ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967309010140]
- 21. Building Natural Product Libraries Using Quantitative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8547436/]
- 22. Novel Chemical Space Exploration via Natural Products - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC2696019/]
- 23. Will Combinatorial Chemistry Keep Its Promise? - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3564354/]
- 24. Exploring chemical space for “druglike” small molecules in ... [https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2025.1553667/full]
- 25. Combinatorial libraries - Latest research and news [https://www.nature.com/subjects/combinatorial-libraries]
- 26. Benchmarking compound activity prediction for real-world ... [https://www.nature.com/articles/s42004-024-01204-4]
- 27. The Revolution of Combinatorial Chemistry [http://www.fortunejournals.com/articles/from-one-to-millions-the-revolution-of-combinatorial-chemistry.html]
- 28. Natural products in drug discovery: advances and ... [https://www.nature.com/articles/s41573-020-00114-z]
- 29. Overview of methods for comparing the efficacies of drugs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3895352/]
- 30. Cortellis Drug Discovery Intelligence Platform [https://clarivate.com/life-sciences-healthcare/research-development/discovery-development/cortellis-pre-clinical-intelligence/]
- 31. Chemical space visual navigation in the era of deep ... [https://www.sciencedirect.com/science/article/pii/S1359644625001059]
- 32. CoLiNN: A Tool for Fast Chemical Space Visualization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11916640/]
- 33. Evaluation of chirality descriptors derived from SMILES heteroencoders [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-01080-7]
- 34. A comparative study of machine learning models on molecular ... [https://www.nature.com/articles/s42004-025-01651-7?error=cookies_not_supported&code=2f580d13-3935-4da5-994f-767fa9029ce1]
- 35. Growth vs. Diversity: A Time-Evolution Analysis of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11870478/]
- 36. the emerging impact of chemoinformatics in chemistry - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC12333164/]
- 37. BitBIRCH: efficient clustering of large molecular libraries [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d5dd00030k]
- 38. BitBIRCH: efficient clustering of large molecular libraries - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11912344/]
- 39. iSIM: instant similarity - RSC Publishing [https://pubs.rsc.org/en/content/articlehtml/2024/dd/d4dd00041b]
- 40. New and fast clustering algorithm of chemical libraries ... [https://iwatobipen.wordpress.com/2024/09/01/new-and-fast-clustering-algorithm-of-chemical-libraries-cheminformatics-rdkit-clustering/]
- 41. BitBIRCH Clustering Refinement Strategies - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC11974763/]
- 42. BitBIRCH: Efficiently Clustering 1 Billion Molecules [https://www.schrodinger.com/life-science/resources/webinar/bitbirch-efficiently-clustering-1-billion-molecules/]
- 43. A Comparative Analysis of Cheminformatics Platforms [https://intuitionlabs.ai/articles/cheminformatics-platforms-comparison]
- 44. Time-Dependent Comparison of the Structural Variations ... [https://www.mdpi.com/1422-0067/25/21/11475]
- 45. An artificial intelligence accelerated virtual screening ... [https://www.nature.com/articles/s41467-024-52061-7]
- 46. Screening of multi deep learning-based de novo molecular ... [https://www.nature.com/articles/s41598-025-86840-z]
- 47. Navigating large chemical spaces in early-phase drug ... [https://www.sciencedirect.com/science/article/pii/S0959440X23000520]
- 48. An Improved Metric and Benchmark for Assessing the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10980085/]
- 49. Evolutionary chemical binding similarity approach ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7362480/]
- 50. Atomically accurate de novo design of antibodies with ... [https://www.nature.com/articles/s41586-025-09721-5]
- 51. Leading AI-Driven Drug Discovery Platforms [https://www.sciencedirect.com/science/article/abs/pii/S0031699725075118]
- 52. Rapid traversal of vast chemical space using machine ... [https://www.nature.com/articles/s43588-025-00777-x]
- 53. Predicted Biological Activity of Purchasable Chemical Space [https://pmc.ncbi.nlm.nih.gov/articles/PMC5780839/]
- 54. Integrative computational approaches for discovery and ... [https://www.frontiersin.org/journals/drug-discovery/articles/10.3389/fddsv.2024.1362456/full]
- 55. QSAR Study, Molecular Docking and Molecular Dynamic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11052498/]
- 56. one derivatives as tubulin inhibitors for breast cancer therapy [https://www.nature.com/articles/s41598-024-66877-2]
- 57. Molecular Dynamics Simulation, Molecular Docking ... [https://www.sciencedirect.com/science/article/pii/S3050787125001039]
- 58. Artificial Intelligence in Natural Product Drug Discovery [https://pmc.ncbi.nlm.nih.gov/articles/PMC11874025/]
- 59. Current strategies to address data scarcity in artificial ... [https://www.sciencedirect.com/science/article/abs/pii/S0010482524008199]
- 60. Chemical diversity of cyanobacterial natural products [https://pubs.rsc.org/en/content/articlelanding/2025/np/d4np00040d]
- 61. Rational Methods for the Selection of Diverse Screening ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4765079/]
- 62. Combinatorial Chemistry in Drug Discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC5645069/]
- 63. AXXVirtual: a chemistry-driven virtual library for drug ... [https://axxam.com/axxvirtual-a-chemistry-driven-virtual-library-for-drug-discovery/]
- 64. HPLC 2025 Preview: The Road To Sustainable Analytical ... [https://www.chromatographyonline.com/view/hplc-2025-preview-the-road-to-sustainable-analytical-chemistry]
- 65. 4 trends shaping the chemicals industry landscape in 2025 [https://www.elsevier.com/industry/4-key-chemicals-industry-trends]
- 66. Computational approaches streamlining drug discovery [https://www.nature.com/articles/s41586-023-05905-z]
- 67. ChemPlot, a Python library for chemical space visualization [https://chemrxiv.org/engage/chemrxiv/article-details/61fab8780aec1ae0ab0fafbc]
- 68. Exploration of Dark Chemical Genomics Space via Portal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8647653/]
- 69. Natural product diversity and its role in chemical biology ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3110584/]
- 70. Novel chemical space exploration via natural products - PubMed [https://pubmed.ncbi.nlm.nih.gov/19265440/]
- 71. Measuring Diversity: Experimental Design of Combinatorial ... [https://pubmed.ncbi.nlm.nih.gov/7739001/]
- 72. Natural product-informed exploration of chemical space to ... [https://pubs.rsc.org/en/content/articlelanding/2021/md/d0md00376j]
- 73. Chemical Space - an overview [https://www.sciencedirect.com/topics/computer-science/chemical-space]
- 74. Chemical Multiverse: An Expanded View of Chemical Space [https://pmc.ncbi.nlm.nih.gov/articles/PMC9787733/]
- 75. Rationally minimizing natural product libraries using mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11915828/]
- 76. Combining Computational Methods for Hit to Lead ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3946937/]
- 77. Hit-to-lead optimization of a 2-aminobenzimidazole series ... [https://www.sciencedirect.com/science/article/pii/S0223523422008273]
- 78. Hit-to-Lead Optimization with Zebrafish: Early Drug ... [https://www.zeclinics.com/blog/hit-to-lead-zebrafish-early-drug-development]
- 79. Case Study of High-Throughput Drug Screening and ... [https://www.mdpi.com/2073-4352/11/12/1579]
- 80. Generative AI for drug discovery and protein design [https://www.sciencedirect.com/science/article/pii/S2590098625000107]
- 81. SwRI develops chemistry LLM called GAMES for faster ... [https://www.swri.org/newsroom/press-releases/swri-develops-chemistry-llm-called-games-faster-drug-discovery]
- 82. A collaborative large language model for drug analysis [https://www.nature.com/articles/s41551-025-01471-z]