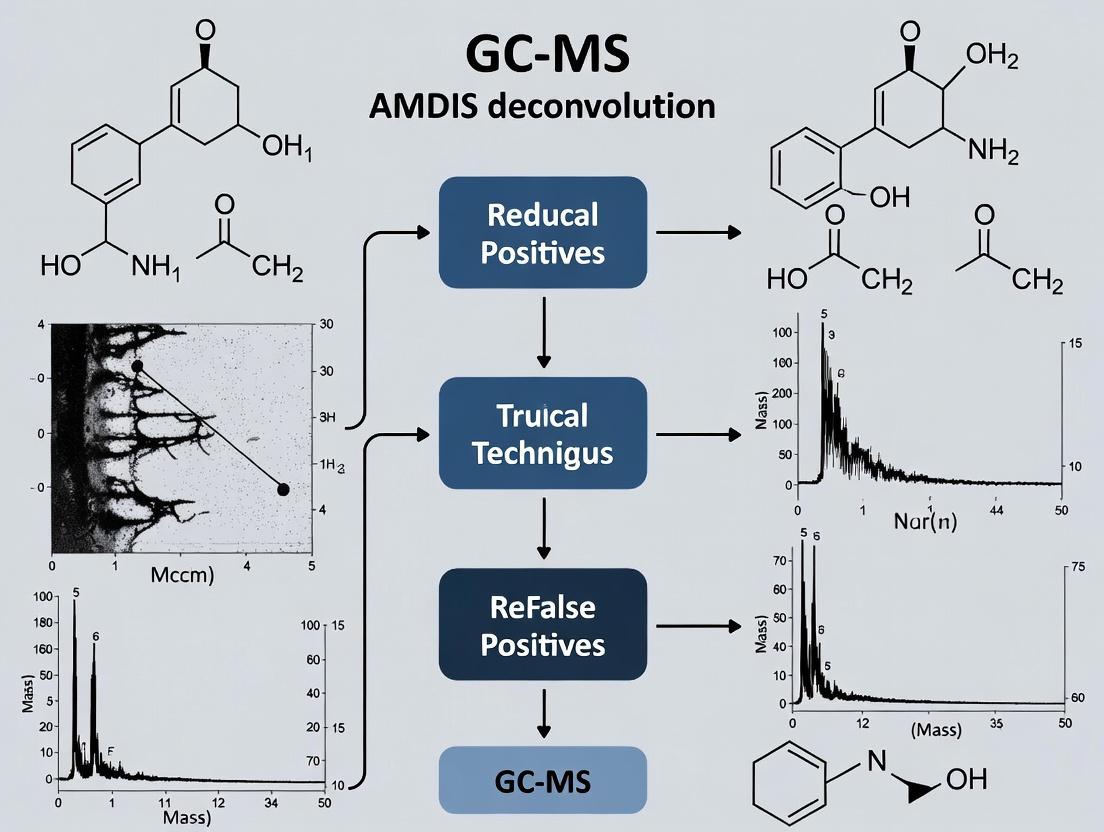
Minimizing False Positives: A Practical Guide to Optimizing AMDIS Deconvolution for Reliable GC-MS Metabolomics
For researchers, scientists, and drug development professionals, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a critical but error-prone tool in GC-MS data analysis, often generating false positives...
Minimizing False Positives: A Practical Guide to Optimizing AMDIS Deconvolution for Reliable GC-MS Metabolomics
Abstract
For researchers, scientists, and drug development professionals, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a critical but error-prone tool in GC-MS data analysis, often generating false positives that compromise data integrity. This article provides a comprehensive guide to reducing these errors, covering the foundational causes of false positives, practical methodologies for parameter optimization and custom library creation, targeted troubleshooting for peak detection, and rigorous validation through comparative analysis and complementary chemometric tools. By synthesizing current research and proven strategies, this guide aims to equip users with the knowledge to significantly enhance the reliability and accuracy of metabolite identification in complex biological samples.
Understanding the Challenge: Why AMDIS Generates False Positives in GC-MS Data
The Critical Role and Inherent Challenge of Spectral Deconvolution in GC-MS Metabolomics
Technical Support Center: Navigating AMDIS Deconvolution
This support center provides targeted guidance for researchers employing spectral deconvolution in GC-MS metabolomics, with a focused aim on reducing false positives—a central challenge in Automated Mass Spectral Deconvolution and Identification System (AMDIS) research. The following FAQs, troubleshooting guides, and protocols are designed to enhance the reliability of your data within this critical context.
Understanding Spectral Deconvolution & AMDIS
Q1: What is spectral deconvolution in GC-MS, and why is it critical for metabolomics? Spectral deconvolution is a mathematical process that separates overlapping chromatographic peaks to extract the pure mass spectrum of each individual chemical compound. In GC-MS metabolomics, complex biological samples often contain hundreds of metabolites that cannot be fully separated by the chromatography column, leading to co-elution. Deconvolution is critical because it allows for the accurate identification and quantification of these co-eluting compounds, which is foundational for discovering true biological signals. Without effective deconvolution, metabolite identification is prone to error, directly contributing to false positives and false negatives in your dataset [1] [2].
Q2: How does AMDIS work, and what are its known strengths and weaknesses concerning false positives? AMDIS operates by analyzing the GC-MS data file, identifying peaks, and using algorithms to separate ion profiles belonging to different compounds. Its strengths include high sensitivity for peak detection, the ability to resolve peaks where intensity ratios exceed 5:1, and widespread availability as freeware [2]. However, its primary weakness in the context of false positive reduction is its tendency to report a higher number of false identifications compared to other software. This occurs because AMDIS aggressively matches deconvoluted spectra against its library. Without strict constraints, it can mistakenly assign library compounds to spectral noise or fragments of other molecules, compromising the reliability of the results [2].
Q3: What is the single most effective step to reduce false positives with AMDIS? The most effective step is to use a customized, targeted user library specific to your research domain. A general commercial library (e.g., full NIST) contains hundreds of thousands of spectra, increasing the chance of random, incorrect matches. A targeted library limits search space to compounds relevant to your study. Research demonstrates that a custom library can reduce potential false hits dramatically (by 200 in one study) and cut analysis processing time significantly [2].
Troubleshooting Guide: Common AMDIS Challenges & Solutions
Problem 1: High Incidence of Incorrect Compound Identifications (False Positives)
- Potential Causes & Solutions:
- Cause: Using an overly broad spectral library.
- Solution: Create and use a project-specific user library. For example, a study on strawberry volatiles built a library of 104 specific compounds, which drastically improved targeting [2].
- Cause: Poorly optimized "Match Factor" settings.
- Cause: Inadequate chromatographic separation or excessive background noise.
- Solution: Review and optimize sample cleanup and GC methods to improve baseline separation. Employ algorithms like Multivariate Curve Resolution (MCR) for challenging co-elutions, which can use cross-validation to determine the correct number of components automatically, reducing model error [3].
- Cause: Using an overly broad spectral library.
Problem 2: Failure to Detect or Deconvolve Low-Abundance Metabolites
- Potential Causes & Solutions:
- Cause: AMDIS sensitivity settings are too stringent.
- Solution: Lower the "Sensitivity" parameter in the analysis method. Perform this adjustment systematically alongside match factor changes to avoid an explosion of false positives.
- Cause: Signal is obscured by chemical noise or background.
- Solution: Utilize extracted ion chromatograms (EICs) for specific m/z values of interest to visualize the compound's profile before deconvolution. Advanced deconvolution methods like DI-MS², which modulates the isolation window, have proven effective at deconvoluting chimeric spectra from low-abundance isobars [4].
- Cause: AMDIS sensitivity settings are too stringent.
Problem 3: Inconsistent Results Across Sample Batches
- Potential Causes & Solutions:
- Cause: Drift in GC retention time.
- Solution: Incorporate retention time alignment tools (often available in downstream data analysis platforms like MZmine). Use retention index markers for robust, instrument-independent calibration [1].
- Cause: Variations in sample matrix or derivatization efficiency.
- Solution: Implement a rigorous, standardized quality control (QC) protocol. Include pooled QC samples and standard mixtures in every batch to monitor system stability and deconvolution performance [1].
- Cause: Drift in GC retention time.
Experimental Protocol for Robust Deconvolution
This protocol outlines a best-practice workflow for GC-MS metabolomics with integrated steps to minimize deconvolution errors.
1. Sample Preparation & Derivatization:
- Extraction: Use a standardized solvent system (e.g., methanol:acetonitrile:water) suitable for your metabolite class [1]. For volatile analysis, consider headspace or SPME techniques [1].
- Clean-up: Perform a lipid removal step for fatty samples to prevent column and liner contamination, which creates background interference and harms deconvolution [1].
- Derivatization: For non-volatile metabolites, use trimethylsilylation. Ensure consistency in time, temperature, and reagent batches to minimize profile variability [1].
2. GC-MS Data Acquisition:
- System Suitability: Run a test mixture to check chromatographic resolution and mass calibration before analyzing experimental samples.
- Quality Controls: Inject pooled QC samples and blank solvents at regular intervals throughout the sequence.
- Data Format: Save data in open, accessible formats (e.g., .mzML, .netCDF) to ensure compatibility with AMDIS and other software tools [5].
3. Data Processing & Deconvolution with AMDIS:
- Library Preparation: Build a targeted user library. Include known standards to record their mass spectra and retention indices (RI). RI is an orthogonal filter critical for rejecting false spectral matches [1] [2].
- Parameter Optimization: Do not use default settings blindly. On a representative subset of data, systematically test combinations of
Sensitivity,Resolution, andShape Requirementsto maximize true positive recovery from your target library. - Deconvolution: Process samples through AMDIS using the optimized method and targeted library.
- Result Filtering: Apply post-deconvolution filters. First, filter by a high match factor (e.g., ≥75). Second, and crucially, filter by a retention index window (e.g., ± 5-10 RI units of the library standard). This two-step filter is highly effective against false positives [2].
4. Validation & Downstream Analysis:
- Manual Verification: Manually inspect integrated peaks for key metabolites, checking for accurate baseline placement and peak shape.
- Statistical Analysis: Export compound data for multivariate statistical analysis. Use data visualization techniques like Hierarchical Clustering Analysis (HCA) heatmaps to identify patterns and spot potential outliers that may stem from deconvolution artifacts [6].
Visual Guide to the Deconvolution Workflow
The following diagram outlines the complete experimental and computational workflow, highlighting critical checkpoints for false positive control.
Diagram 1: GC-MS Deconvolution & False Positive Reduction Workflow (100 chars)
Key Data & Parameter Tables
Table 1: Impact of a Custom Target Library on AMDIS Performance [2]
| Performance Metric | Using General NIST Library | Using Custom Strawberry VOC Library | Improvement |
|---|---|---|---|
| Potential False Hits | ~200+ | Minimized | Reduced by ~200 |
| Report File Size | Large (Baseline) | 0.98 MB | Reduced by >96% |
| Processing Time | 31 seconds | 9 seconds | ~71% faster |
Table 2: Recommended AMDIS Parameter Ranges for Balanced Sensitivity/Specificity
| Parameter | Purpose | Low Value (More Sensitive) | High Value (More Strict) | Recommended Starting Point |
|---|---|---|---|---|
| Sensitivity | Determines how small a peak can be detected. | High (e.g., 90) | Low (e.g., 30) | 70 |
| Resolution | Sets the required sharpness of a peak. | Low (e.g., 10) | High (e.g., 100) | 50 |
| Shape Factor | Defines the required fit to a Gaussian shape. | Low (e.g., 50) | High (e.g., 99) | 80 |
| Match Factor | Threshold for library identification. | Low (e.g., 50) | High (e.g., 90) | 75 (with RI filter) |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for GC-MS Metabolomics with Reliable Deconvolution
| Item | Function in Reducing Deconvolution Errors | Example/Note |
|---|---|---|
| Retention Index Marker Mix | Provides standardized retention anchors to calibrate retention times across runs, enabling the critical use of Retention Index filtering to reject false matches. | n-Alkane series (C8-C40) or fatty acid methyl ester (FAME) mix. |
| Chemical Derivatization Reagents | Converts non-volatile metabolites into volatile, stable derivatives for GC analysis. Consistent derivatization is key to reproducible spectra. | MSTFA (N-Methyl-N-(trimethylsilyl)trifluoroacetamide) with 1% TMCS. TMSH (Trimethylsulfonium hydroxide) for specific applications [3] [1]. |
| Targeted Analytical Standards | Used to build a custom user library. Essential for acquiring reference spectra and retention indices for metabolites of interest. | Purchase or synthesize pure compounds relevant to your biological system. |
| Quality Control (QC) Reference Material | A pooled sample from all experimental groups. Monitors instrument stability and data quality throughout the sequence, flagging batch effects. | Run repeatedly to assess technical variance in deconvolution results [1]. |
| Dedicated Spectral Library Software | For creating, managing, and formatting custom user libraries compatible with AMDIS (.MSL files). | NIST MS Search, AMDIS library creation tools, or other commercial library managers. |
This technical support center addresses a core challenge in analytical chemistry and systems biology: the generation of false positive identifications in Gas Chromatography-Mass Spectrometry (GC-MS) data analysis, specifically during the Automated Mass Spectral Deconvolution and Identification System (AMDIS) processing step. Within the broader thesis context of improving data fidelity in GC-MS research, false positives undermine the validity of metabolomic profiling, biomarker discovery, and compound identification. The primary culprits are co-elution, where two or more compounds exit the chromatography column at nearly the same time, and the inherent sensitivity and assumptions of deconvolution algorithms like AMDIS, which must interpret complex, overlapping spectral data [7]. This resource provides targeted troubleshooting guides, FAQs, and methodological advice to help researchers, scientists, and drug development professionals diagnose, mitigate, and prevent these issues in their experiments.
Technical Troubleshooting Guides
Issue 1: Persistent Co-elution Despite Method Adjustment
Problem Description: Two or more target analytes consistently elute together (e.g., with retention times of 3.21 min and 3.27 min), leading to a single, unresolved peak in the Total Ion Chromatogram (TIC). Attempts to resolve them by adjusting flow rate or mobile phase pH have failed [8].
Diagnosis Checklist:
- Check Retention Mechanism: Confirm your compounds are being retained on the column. Peaks eluting near the solvent front (at the column void time) indicate no meaningful chromatographic retention is occurring [8].
- Review Mobile Phase Chemistry: A complex mobile phase "soup" (e.g., containing ion-pairing reagents, buffers, and organic solvent) may not provide a controllable retention mechanism for your specific compounds [8].
- Assess Column Suitability: The selected stationary phase (e.g., a generic C18) may not offer the required selectivity for your analytes' chemical properties (e.g., pKa of 2.2 and 5.0) [8].
Step-by-Step Resolution Protocol:
- Shift Separation Mechanism: Abandon the current method. For acidic compounds, test a gradient method from 2% to 50% acetonitrile with 0.1% phosphoric acid over 10 minutes [8].
- Consider Orthogonal Chemistry: If the standard reversed-phase does not work, investigate alternative separation modes:
- Hydrophilic Interaction Liquid Chromatography (HILIC) for polar compounds.
- Mixed-mode columns that combine reversed-phase and ion-exchange mechanisms [8].
- Optimize for Sensitivity Post-Separation: Once separation is achieved, use mass spectrometry to regain sensitivity. Employ Selected Ion Monitoring (SIM) or Multiple Reaction Monitoring (MRM) on a triple quadrupole system to monitor unique ions for each co-eluting compound, providing digital resolution based on mass [9].
Issue 2: High False Positive Rate from AMDIS Deconvolution
Problem Description: AMDIS reports a high number of compound identifications, but manual validation reveals 70-80% are incorrect, often due to algorithm misassignment of fragments from co-eluting compounds or noise [10].
Diagnosis Checklist:
- Review Deconvolution Settings: The default AMDIS parameters (deconvolution width, component width, sensitivity) are likely too permissive for your complex sample matrix [10].
- Check Spectral Match Quality: Low Match Factors (MF) or poor agreement with library spectra for reported compounds indicate weak identifications.
- Inspect the Raw Data: Visual examination of the extracted ion chromatograms (EICs) may show peak shapes inconsistent with a pure component.
Step-by-Step Resolution Protocol:
- Systematically Optimize AMDIS Parameters: Use a design of experiments (DoE) approach to find the best settings for your specific instrument and sample type. Key parameters to adjust include:
- Deconvolution Width: Should approximate the widest peak in the chromatogram.
- Component Width: Adjust to model your typical peak shape.
- Sensitivity: Increase to find trace components, but balance against false positives [10].
- Apply a Heuristic Filter: Develop or apply a Compound Detection Factor (CDF) that weights the AMDIS Match Factor against other orthogonal data, such as retention index (RI) accuracy. This can significantly reduce false positive rates [10].
- Implement Complementary Deconvolution: For severely co-eluted peaks, use a second, mathematically distinct algorithm like Ratio Analysis of Mass Spectrometry (RAMSY). Process the problematic region with RAMSY to recover pure spectra for low-intensity, co-eluted ions that AMDIS may miss, then cross-validate identifications [10].
Issue 3: Retention Time Shift and Co-elution in Sequential Runs
Problem Description: The first injection in a sequence is satisfactory, but all subsequent injections show systematic retention time shifts and new co-elution events [11].
Diagnosis Checklist:
- Review Method Equilibration: The chromatographic method likely does not provide sufficient time for the column and system to return to initial conditions (e.g., mobile phase composition, pH) before the next injection [11].
- Check for System Volumes: Ensure the equilibration volume is sufficient to flush and re-equilibrate the entire fluidic path.
Step-by-Step Resolution Protocol:
- Extend the Run Time: Increase the post-run equilibration segment of your gradient method.
- Calculate Required Equilibration Volume: A good starting point is to program a wash volume equivalent to 3 times the system volume plus 5 times the column volume of the starting mobile phase [11].
- Verify Performance: After implementing the longer equilibration, run a sequence of standards to confirm retention time stability and the resolution of the previously co-eluted peaks.
Frequently Asked Questions (FAQs)
Q1: What is the fundamental cause of co-elution, and can it ever be beneficial? A1: Co-elution occurs when compounds have sufficiently similar physical and chemical interactions with the stationary phase of the GC or LC column. While it is generally a problem for identification, it can be exploited beneficially in specialized techniques like dual-isotope measurement. By forcing an analyte and its isotopically labeled internal standard to co-elute perfectly, their ionization efficiencies in the MS source become virtually identical, enabling highly precise and reproducible quantitative measurements [12].
Q2: Beyond AMDIS, what are my options for deconvoluting co-eluted GC-MS data? A2: Several algorithmic approaches exist, each with strengths. Bayesian Deconvolution methods model the data probabilistically, exploring several possible numbers of components in a peak and ranking identifications by probability, which can improve accuracy in high co-elution situations [13]. Ratio Analysis of Mass Spectrometry (RAMSY) is a complementary, non-empirical tool that can recover spectra from severe overlap [10]. For high-resolution accurate-mass (HRAM) GC-Orbitrap data, newer Bayesian pipelines have been shown to outperform traditional methods like AMDIS in correctly resolving compounds [13].
Q3: How can I proactively design experiments to minimize co-elution problems? A3: Invest time in orthogonal separation strategies. If your primary separation is reversed-phase liquid chromatography (RP-LC), consider adding a fractionation step using size-exclusion (SEC) or ion-exchange (IEX) chromatography under native conditions to pre-separate complexes [14]. In method development, always scout a wide range of gradients and mobile phase compositions. Using high-resolution accurate-mass (HRAM) instrumentation (e.g., GC-Orbitrap or Q-TOF) from the start provides more detailed data, making deconvolution algorithms more effective [13].
Q4: My deconvolution software identified a compound. What orthogonal evidence should I seek to confirm it and avoid reporting a false positive? A4: Never rely on spectral matching alone. Essential orthogonal verification includes:
- Retention Index/Time Match: Compare the observed retention time or calculated retention index against an authentic standard analyzed under identical conditions [10].
- Ion Ratio Verification: For SIM or MRM data, confirm the ratios of monitored ions match those of the standard [9].
- MS/MS Fragmentation: If using tandem MS, compare the full fragmentation spectrum.
- Literature/Database Cross-Check: Consult resources like the NIST database, PubChem, or METLIN to see if the putative identification is plausible in your sample context (e.g., a plant, human fluid) [10].
Comparative Analysis of Key Factors
Table 1: Comparison of Deconvolution Software and Strategies for Managing Co-elution
| Software/Strategy | Algorithmic Principle | Best Use Case | Key Advantage | Primary Limitation/Risk |
|---|---|---|---|---|
| AMDIS (Standard Use) | Empirical, model peak fitting based on ion chromatogram shapes [7]. | Routine screening of moderately complex samples. | Integrated, widely available, and relatively fast. | High false positive rate (70-80%) with complex samples or improper settings [10]. |
| AMDIS (Optimized) | Empirical, with parameters tuned via DoE and filtered with heuristics (e.g., CDF) [10]. | Targeted studies of specific, known complex matrices (e.g., plant metabolomics). | Significantly reduced false positives while maintaining workflow. | Optimization is time-consuming and matrix-specific. |
| RAMSY | Ratio analysis of mass spectra across multiple samples/channels [10]. | Resolving severe, intractable co-elution in critical peaks. | Can digitally resolve spectra where traditional peak-shape analysis fails. | Not a full workflow; best used as a complementary tool on problematic regions. |
| Bayesian Deconvolution | Probabilistic modeling of the number of components and their spectra [13]. | High-resolution (e.g., GC-Orbitrap) data with extreme co-elution. | Provides probability scores for identifications; explores multiple component numbers. | Computationally intensive; requires specialized software/implementation. |
| Chromatographic Optimization | Physical separation via adjusted mobile/stationary phase chemistry [8]. | Prevention of co-elution during method development. | Eliminates the problem at the source; most reliable. | Not always possible for all analytes; can be a lengthy process. |
Detailed Experimental Protocols
Objective: To empirically determine the set of AMDIS deconvolution parameters that maximizes true positive identifications and minimizes false positives for a specific GC-MS system and sample type. Materials: A representative pooled sample or quality control (QC) sample analyzed in triplicate; AMDIS software; statistical software (e.g., JMP, R, or Modde). Procedure:
- Select Critical Parameters: Choose 3-4 parameters most likely to influence results. Common choices are: Sensitivity, Resolution, Shape Requirement, and Deconvolution Width.
- Design the Experiment: Use a fractional factorial design (e.g., a 2^(4-1) design) to define 8-12 unique parameter sets to test.
- Create and Run Batch Jobs: Process the same representative data file through AMDIS using each predefined parameter set.
- Define and Measure Response: Manually curate the results for a challenging region of the chromatogram. For each parameter set, calculate a response metric, such as: % False Positives = (1 - (Verified True IDs / Total Reported IDs)) * 100.
- Statistical Analysis & Modeling: Input the results into the statistical software. Use the model to identify which parameters have a significant effect and to predict the optimal parameter set that minimizes the % False Positives.
- Validation: Apply the predicted optimal parameters to a new, independent data set and verify the improvement.
Objective: To add a robust filtering layer to AMDIS outputs, reducing false positives by requiring agreement between spectral match and chromatographic retention data. Materials: AMDIS result file (.ELU); a retention index (RI) standard mixture (e.g., alkane series for GC) analyzed on the same method; a database of target compounds with known RIs. Procedure:
- Generate RI Calibration: Analyze the alkane standard. Record the retention times and calculate a linear calibration curve of RI vs. retention time.
- Calculate Experimental RI: For each compound reported by AMDIS, use its retention time to calculate its experimental RI from the calibration curve.
- Define the CDF: Develop a heuristic score. A simple, effective CDF can be a weighted sum: CDF = (Match Factor / 100) * (1 - (|ΔRI| / RI_Tolerance)).
Match Factoris from AMDIS (0-100).ΔRIis the absolute difference between the experimental and database RI.RI_Toleranceis an acceptable window (e.g., 10-20 RI units).
- Apply the Filter: Set a CDF threshold (e.g., >0.7). Discard all AMDIS identifications with a CDF below the threshold. This single step can dramatically reduce false positives caused by good spectral matches from co-eluting interferents that have the wrong retention behavior.
Visualizing the Problem and Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Resources for Method Development and Deconvolution
| Item / Resource | Function / Purpose | Key Application in False Positive Reduction |
|---|---|---|
| Mixed-Mode or HILIC Chromatography Columns [8] | Provide alternative retention mechanisms (e.g., ion-exchange + reversed-phase) to separate compounds that co-elute on standard C18 columns. | Prevents co-elution at the source by changing the fundamental separation chemistry, making deconvolution unnecessary. |
| Retention Index Standard Mixtures (e.g., n-Alkane series for GC) [10] | Allows calculation of a system-independent retention index (RI) for each compound, orthogonal to mass spectral data. | Enables orthogonal verification of AMDIS identifications; a compound with a good spectral match but wrong RI is likely a false positive. |
| Isotopically Labeled Internal Standards [12] | A chemically identical version of the target analyte with heavy isotopes (e.g., ^13C, ^2H), used for precise quantification. | When forced to co-elute perfectly with the native analyte, they correct for ionization variances and can help validate the analyte's presence by ratio. |
| AMDIS Software [7] | The standard algorithm for deconvoluting, identifying, and quantifying components in GC-MS data. | Its parameter optimization (via DoE) and post-processing filters (like CDF) are the primary tools for improving its own output fidelity [10]. |
| Bayesian Deconvolution Software / Scripts [13] | Advanced algorithms that model the number of components and their spectra probabilistically. | Provides a probability score for each identification, offering a more robust measure of confidence than a simple match factor, especially for high-res data. |
| NIST / Wiley / Fiehn Mass Spectral Libraries [10] | Comprehensive databases of reference mass spectra for compound identification. | The quality of the reference spectrum is critical. Using a well-curated, application-specific library (e.g., the Fiehn Metabolomics library) improves correct matching. |
In gas chromatography-mass spectrometry (GC-MS) analysis, particularly in metabolomics and forensic toxicology, the accurate deconvolution of co-eluting peaks is paramount. Deconvolution software separates overlapping signals to extract pure component spectra for reliable identification [15]. However, a persistent challenge across platforms is the generation of false positive identifications, which can compromise data integrity and lead to erroneous biological or chemical conclusions [15] [2].
This technical support center is framed within a focused thesis on reducing false positives in GC-MS AMDIS deconvolution research. AMDIS (Automated Mass Spectral Deconvolution and Identification System) is widely used freeware known for its powerful deconvolution engine and user-friendly interface [2] [16]. Yet, comparative studies consistently note its tendency to report a higher number of false positives compared to some commercial alternatives [15] [2]. The following guide provides a comparative analysis, troubleshooting, and best practices to help researchers, scientists, and drug development professionals optimize their deconvolution workflows, mitigate false identifications, and generate more reliable data.
The performance of deconvolution software is typically evaluated based on its sensitivity (ability to detect true compounds), specificity (ability to avoid false identifications), and robustness to parameter settings. The following table summarizes key findings from comparative studies involving AMDIS, ChromaTOF, AnalyzerPro, and other tools.
Table 1: Comparative Performance of GC-MS Deconvolution Software
| Software | Provider/Availability | Reported Strength | Reported Weakness (Re: False Positives) | Key Differentiating Feature |
|---|---|---|---|---|
| AMDIS | NIST (Freeware) | Excellent deconvolution of severely co-eluting peaks; high sensitivity; supports user libraries [2] [16]. | Highest propensity for false positives; requires careful library and parameter tuning [15] [2]. | Free, versatile, and highly sensitive, but results require rigorous vetting. |
| ChromaTOF | LECO (Commercial) | Tight integration with LECO instruments; automated processing [15] [17]. | Can produce a large number of false positives [15] [18]. | Vendor-specific solution offering high-throughput automation. |
| AnalyzerPro (Legacy)/Analyze | SpectralWorks (Commercial) | Advanced statistical and workflow tools for false positive reduction [19]. | May produce false negatives (miss true compounds) [15]. | Incorporates tools like PCA and target ion filtering to gate identifications [19]. |
| ADAP-GC 3.0 | Open Source (R/C) | Improved sensitivity for low-concentration compounds; robust peak detection [18]. | Performance can vary with complex biological matrices [18]. | Open-source pipeline using wavelet transforms for robust peak detection [18]. |
| PARADISe | Open Access | High robustness to user settings; handles severe overlap and low S/N peaks well [20]. | - | Based on PARAFAC2 algorithm; claims fewer non-detects and easier parameter setup than AMDIS/ChromaTOF [20]. |
Detailed Experimental Protocol for Comparative Evaluation
The foundational conclusions in Table 1 are drawn from controlled experimental comparisons. The following methodology, adapted from a key comparative study, outlines how such performance data are generated [15].
Objective: To evaluate and compare the deconvolution performance, including false positive rates, of AMDIS, ChromaTOF, and AnalyzerPro using a standardized metabolite mixture.
1. Sample Preparation:
- Prepare stock solutions (approx. 20 mM) of 36 endogenous metabolites (e.g., amino acids, organic acids, sugars) in a 50:50 water:acetonitrile mixture [15].
- Derivatize samples using a standard method (e.g., methoximation and silylation) to make metabolites volatile for GC-MS analysis [15].
- Create four test solutions by mixing and diluting stock solutions to achieve a wide range of relative concentration ratios, mimicking biological complexity [15].
2. Instrumental Analysis (GC-TOF-MS):
- Instrument: Use a GC system coupled to a Time-of-Flight Mass Spectrometer (e.g., LECO Pegasus III) [15].
- Column: DB-5ms capillary column (30 m × 250 µm I.D., 0.25 µm film thickness) [18].
- GC Program: Initial oven temperature (e.g., 80°C for 2 min), followed by ramps (e.g., 10°C/min to 220°C, then 5°C/min to 240°C, then 25°C/min to 290°C), with a final hold [18].
- MS Settings: Electron Impact ionization (70 eV); full scan mode (e.g., m/z 40-600); acquisition rate of 20 spectra per second [18].
3. Data Processing & Analysis:
- Process the same raw data file with each software (AMDIS, ChromaTOF, AnalyzerPro) using default or optimized settings for each [15].
- For identification, use a custom library containing mass spectra and retention indices (RI) of the expected metabolite derivatives [15].
- Manually validate all software-reported identifications against known sample composition to classify results as True Positives, False Positives, or False Negatives [15].
- Compare software based on the total number of components identified, the accuracy of identifications, and the number of false positives/negatives reported [15].
Technical Support & Troubleshooting Guides
Issue 1: Excessive False Positives in AMDIS Results
- Problem: AMDIS reports many compounds not present in the sample, especially in complex matrices [15] [2].
- Root Cause: High sensitivity and a low default threshold for spectrum matching can cause noise or background ions to be incorrectly matched to library spectra [2] [16].
- Solution:
- Implement a Custom Target Library: The most effective strategy is to use a dedicated library containing only the compounds relevant to your study. A custom strawberry VOC library reduced false hits by 200 compared to a commercial library [2].
- Adjust Match Factor: Increase the Minimum Match Factor (e.g., from 60 to 70 or 80) in the Analysis Settings/Identification tab to require a higher spectral similarity for reporting [16].
- Leverage Retention Index: Always use retention index (RI) filtering alongside spectral matching. In Analyze/Settings, specify a calibration file and set an appropriate RI window (e.g., ±10 units). This adds a critical orthogonal filter for identity [15] [16].
- Post-Processing Review: Manually inspect low-probability hits. Use the "Show Component on Chromatogram" feature to assess peak shape and purity [16].
Issue 2: Managing Co-elution and Poor Peak Resolution
- Problem: Compounds are not fully separated chromatographically, leading to mixed spectra and failed deconvolution.
- Root Cause: Complex samples or suboptimal GC methods cause peaks to overlap [15].
- Solution:
- Optimize AMDIS Deconvolution Parameters: In the Deconvolution Parameters settings, adjust the "Component Width" parameter. This is the most critical parameter for predicting deconvolution accuracy and must be set to approximate the width of peaks in your chromatogram [15].
- Adjust Sensitivity & Resolution: Modify the "Sensitivity" (high for low-concentration compounds) and "Resolution" (high for better separation of shoulder peaks) settings [16].
- Use the Right Tool for Severe Overlap: For extremely challenging co-elution, consider using software based on advanced algorithms like PARAFAC2 (e.g., PARADISe), which is specifically designed to handle highly overlapping and embedded peaks with minimal user input [21] [20].
Issue 3: False Positives from Structurally Similar Compounds (e.g., MDMA/MDA)
- Problem: Software misidentifies a compound as its structural analog, a common issue in forensic and pharmaceutical analysis [19].
- Root Cause: Analogues share most fragment ions, leading to high spectral similarity scores [19].
- Solution (Employed by AnalyzerPro):
- Utilize Orthogonal Data: If using MS methods with multiple functions (e.g., different collision energies), ensure the software compares data across all functions [19].
- Apply a Target Ion Filter: Implement a rule that requires the presence of a unique ion (like a distinct molecular ion) for a positive identification. This can gate and eliminate false positives from analogs [19].
- Statistical Correlation: Use principal component analysis (PCA) on the full spectral dataset to visualize and confirm separations between compound groups [19].
Diagram: GC-MS Deconvolution Workflow with Critical False Positive Reduction Filters. A robust workflow integrates custom libraries and orthogonal filters (Retention Index, Target Ions) after deconvolution and spectral matching to separate false from true identifications [15] [2] [19].
Frequently Asked Questions (FAQs)
Q1: Why does AMDIS produce more false positives than AnalyzerPro or other software? A1: AMDIS is designed with a highly sensitive deconvolution algorithm to maximize component detection, which can extract spectra from minor shoulders or noise [2]. Without strict filtering via a custom target library and retention index, these extracted spectra can match incorrectly to a broad commercial library [15] [2]. In contrast, software like AnalyzerPro incorporates advanced statistical workflows and gating logic (e.g., requiring a specific molecular ion) that actively suppress chemically plausible false positives [19].
Q2: What is the single most important step to improve AMDIS accuracy? A2: Creating and using a custom, project-specific target library is paramount [2]. This library should contain the retention indices and mass spectra of your compounds of interest, ideally from analyzed standards. A study showed this reduced potential false hits by 200 and cut processing time by 71% [2]. This limits the search space, dramatically reducing opportunities for incorrect matches.
Q3: Are newer or open-source tools like ADAP-GC 3.0 or PARADISe better than AMDIS? A3: "Better" depends on the need. For robustness and ease of use, PARADISe requires far fewer user-defined parameters and is less user-dependent, making it excellent for standardized processing [20]. For sensitivity to trace compounds, ADAP-GC 3.0 uses wavelet transforms for improved peak detection at low concentrations [18]. However, AMDIS remains highly valuable due to its proven deconvolution power, flexibility, and zero cost. The optimal choice may involve using AMDIS with stringent settings or as part of a multi-tool validation pipeline.
Q4: How can I validate my deconvolution results to be confident they aren't false positives? A4: Employ a multi-confirmation strategy: 1. Retention Index Match: Confirm the RI matches your standard within a tight window (e.g., ±5-10 units) [15]. 2. Spectral Purity: Inspect the deconvoluted spectrum in AMDIS. A clean, low-noise spectrum with a high match factor (e.g., >80) is more reliable [16]. 3. Orthogonal Verification: If possible, confirm identifications using a different analytical technique (e.g., different GC column, LC-MS, or standard addition). 4. Statistical Consistency: Check the identification consistency across biological or technical replicates; false positives often appear sporadically.
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagents and Materials for GC-MS Deconvolution Studies
| Item | Function in Protocol | Critical Notes for Reducing False Positives |
|---|---|---|
| Derivatization Reagents (e.g., MSTFA, BSTFA with TMCS) | Increases volatility and thermal stability of polar metabolites (e.g., acids, sugars) for GC-MS analysis [15]. | Incomplete or inconsistent derivatization creates multiple derivatives for a single metabolite, complicating the chromatogram and increasing risk of misidentification [15]. |
| Retention Index Standard Mix (e.g., n-Alkane series) | Used to calculate temperature-programmed retention indices (RI) for each analyte [15]. | Essential for creating a reliable custom library. RI provides a second, independent identification point that filters out false spectral matches [15] [2]. |
| Custom Target Library (in .MSL or .ELU format) | Contains the mass spectra and known RIs of the specific compounds targeted in the study [2]. | The most critical tool for false positive reduction in AMDIS. A focused library limits search scope and improves both accuracy and processing speed [2]. |
| Analytical Standard Compounds | Used to generate reference spectra and retention times/indices for the custom library [15]. | Pure, high-quality standards are necessary to build a definitive library. Analyze them under the same instrumental conditions as your samples. |
| Quality Control (QC) Sample (e.g., pooled sample from all groups) | Monitors instrument stability and data reproducibility across batch runs. | Systematic drift in retention time in QCs can cause RI-based identification to fail, leading to false negatives or positives. Regular alignment is needed. |
Proven Strategies: Optimizing AMDIS Settings and Building Targeted Libraries
Technical Support Center: Troubleshooting Guides & FAQs
This Technical Support Center provides targeted guidance for researchers aiming to optimize the Automated Mass Spectral Deconvolution and Identification System (AMDIS) within GC-MS workflows. The following FAQs address common challenges directly related to reducing false positives in deconvolution, framed within a broader thesis on improving the reliability of metabolomics and exposomics data [7] [22].
FAQ 1: My AMDIS analysis reports a high number of false-positive compound identifications. Which parameters should I adjust first to improve specificity? A high false-positive rate is a common challenge, as AMDIS can misidentify noise or co-eluting fragments as true components [23] [24]. Your first adjustments should focus on the Component Width and Sensitivity settings.
- Action on Component Width: Increase the specified component width to more closely match the actual peak widths in your chromatogram. An inaccurately low setting causes AMDIS to mistakenly deconvolve a single, broad peak into multiple, false components. Start with a width value equal to your average peak width at half height.
- Action on Sensitivity: Do not simply maximize sensitivity to find more peaks. Instead, use a moderate setting and pair it with a higher Minimum Match Factor (e.g., ≥70) for library searches. This ensures only well-resolved peaks with strong spectral matches are reported. Furthermore, integrate a Retention Index (RI) filter if your library supports it. Using RI information can dramatically reduce false positives by adding a secondary, orthogonal confirmation to the spectral match [22] [25].
FAQ 2: I am missing low-abundance metabolites in complex samples, but increasing Sensitivity also increases noise and false positives. How can I resolve this? This is a classic sensitivity/specificity trade-off. Instead of relying solely on the software's Sensitivity parameter, optimize your experimental and data acquisition conditions.
- Pre-Instrument Optimization: For trace-level analysis, employ techniques like Programmed Temperature Vaporization (PTV) for large-volume injection, which can improve sensitivity by 10-100x compared to standard splitless injection [26]. Also, consider using a fast GC column (e.g., 10-15 m length, ≤0.18 mm inner diameter) with a thin film (0.1 µm). These columns produce sharper peaks, increasing signal-to-noise ratio and improving detection limits [26].
- Acquisition Mode Selection: For targeted analysis of known compounds, switch from full-scan mode to Selected Ion Monitoring (SIM). SIM dramatically reduces chemical noise by only monitoring specific ions for your target analytes, thereby increasing the signal-to-noise ratio and enabling detection of lower concentrations [27].
- AMDIS Settings: Use a two-pass analysis strategy. First, run AMDIS with a lower Sensitivity setting to confidently identify major components. Second, process the data again with a higher Sensitivity setting but target a specific, narrow Retention Time Window where your metabolite of interest is expected, minimizing interference from other regions.
FAQ 3: How do I set the Resolution settings when my chromatogram has both very narrow and very broad peaks? The "Resolution" parameter in AMDIS (sometimes called "Peak Sharpness Threshold") helps distinguish true peaks from background noise. A one-size-fits-all setting may not work for complex samples.
- Strategy: AMDIS can apply a single Resolution setting globally, which may not be optimal [7]. The most effective approach is to improve the chromatographic resolution before data processing.
- Chromatographic Optimization: Use the resolution equation (R = ¼ √N * [(α-1)/α] * [k/(k+1)]) to guide method development [28]. To separate compounds of varying widths:
- Adjust Selectivity (α): This has the greatest impact. Change the column stationary phase to increase differences in compound retention [28].
- Optimize Efficiency (N): Use a longer column or a thinner film to increase the number of theoretical plates for better separation of narrow peaks [28].
- Modify Retention (k): Adjust the temperature program. A slower ramp rate generally increases retention and peak width, improving separation of early-eluting, narrow peaks.
- Post-Hoc Solution: If re-running the sample is not possible, segment your chromatogram and process different time regions with different Resolution settings that are appropriate for the local peak widths.
Optimizing Core AMDIS Parameters for Reduced False Positives
Optimizing AMDIS requires a balanced understanding of how its key parameters interact with your specific chromatographic data. The following tables summarize quantitative guidelines and effects.
Table 1: AMDIS Parameter Optimization Guide
| Parameter | Primary Function | Recommended Starting Value | Effect on False Positives | Thesis Context: Action to Reduce False Positives |
|---|---|---|---|---|
| Component Width | Sets the expected width of chromatographic peaks. | Set to the average peak width (in scans or seconds) of well-resolved peaks in your method. | Too Low: One wide peak is split into multiple false components. Too High: Two co-eluting peaks are merged, causing misidentification. | Calibrate using a standard mix analyzed with your exact method. Prioritize accurate width over narrow peaks. |
| Sensitivity | Controls the threshold for distinguishing signal from noise. | Start with a moderate value (e.g., 50-70 in AMDIS). | Too High: Noise and background artifacts are reported as peaks. Too Low: Legitimate low-abundance analytes are missed (false negatives). | Use in conjunction with a high Minimum Match Factor and Retention Index filtering [22] [24]. |
| Resolution / Peak Sharpness | Determines the required sharpness for a signal to be considered a peak. | Default setting is often sufficient. Adjust if analyzing very sharp (e.g., fast GC) or very broad peaks. | Too High: Broad, real peaks (e.g., from heavily tailing compounds) are rejected. Too Low: Slow baseline drift is interpreted as a peak. | Focus on improving chromatographic resolution at the source using the resolution equation [28]. |
| Minimum Match Factor | The lowest spectral similarity score accepted for a library identification. | Increase to ≥70 for confident reporting; use ≥80 for high-confidence identifications. | Too Low: Poor spectral matches are reported as identifications. Too High: Correct identifications with moderate spectral variability are rejected. | This is a critical, post-deconvolution filter. Raising this threshold is one of the most direct ways to reduce false-positive annotations [24]. |
Table 2: Impact of Instrumental & Acquisition Parameters on Deconvolution
| Parameter | Typical Range / Options | Impact on Deconvolution & False Positives | Optimization Tip for Thesis Research |
|---|---|---|---|
| Scan Rate (ms) | 5-20 Hz (full scan) [27] | Too Slow: Results in too few data points across a peak, harming accurate deconvolution and quantitation [27]. Optimal: Aim for ≥10 scans/peak for reliable shape determination. | For fast GC peaks (<2s width), ensure your MS scan rate is high enough to capture peak shape. Consider SIM mode for more data points [27] [26]. |
| Acquisition Mode | Full Scan, SIM, MS/MS [27] | Full Scan: Universal but noisiest, leading to challenging deconvolution [27]. SIM/MS: Reduces noise, simplifying deconvolution and lowering false detection rates. | Use full scan for untargeted discovery. For targeted validation of key biomarkers, use SIM or MS/MS to provide cleaner data for confident identification [26]. |
| Column Inner Diameter (ID) | 0.1 - 0.32 mm [26] [28] | Narrower ID (e.g., 0.18 mm): Produces sharper, more intense peaks, improving S/N and deconvolution of close-eluting compounds [26]. | Switching from a 0.25 mm to a 0.18 mm ID column can improve resolution and peak height, directly aiding AMDIS's component perception. |
| Injection Technique | Splitless, PTV, On-column [26] | PTV Large-Volume Injection: Can improve sensitivity 10-100x for trace analytes, bringing them above the noise floor for reliable deconvolution [26]. | For exposomics research targeting trace environmental contaminants, PTV is essential for detecting low-level signals that would otherwise be lost in noise. |
Experimental Protocols for Key Validation Experiments
Protocol 1: Establishing System-Specific AMDIS Parameter Baselines This protocol calibrates AMDIS settings using a well-characterized standard mixture under your exact analytical conditions, establishing a benchmark for component width and sensitivity.
- Materials: Prepare a calibration mixture containing 10-15 compounds spanning your expected retention time and volatility range (e.g., alkane mix for RI, or a metabolomics standard mix like the FAME mix or MegaMix [25]).
- Analysis: Run the mixture using your standard GC-MS method in full-scan mode. Ensure the peak widths are chromatographically optimal (neither too narrow nor too broad).
- Measurement: In your data analysis software, measure the average peak width at half height (in seconds) for 5-7 well-resolved, symmetrical peaks across the chromatogram.
- AMDIS Calibration:
- Enter the average peak width (in seconds) into the Component Width parameter.
- Set Sensitivity to a moderate value (e.g., 65).
- Process the data and inspect the deconvolution report. AMDIS should correctly identify all compounds in the mix without splitting peaks or creating extras.
- Adjust Sensitivity incrementally until all expected compounds are found with minimal extra "unknown" components.
- Documentation: Record these optimized settings as your "method baseline." Re-calibrate if any major change is made to the chromatographic method (column, temperature program, flow rate).
Protocol 2: Validating Identifications with a Retention Index Filter to Reduce False Positives This protocol adds a mandatory retention index check to the standard spectral matching process, significantly increasing annotation confidence [22] [25].
- Prerequisites: You must use a retention index-calibrated spectral library (e.g., a library with Kovats or equivalent RI values stored for each compound). A standard alkane series (e.g., C8-C40) must be analyzed using the same method as your samples.
- Sample Analysis: Analyze your biological or environmental samples. Also, analyze the alkane standard separately to calculate the observed RI for each alkane.
- Data Processing with RI:
- Process your sample data with AMDIS using your optimized parameters.
- In the identification settings, enable the RI filter. Set an RI tolerance window (e.g., ±10 index units for a robust method; ±5 for a highly reproducible method) [25].
- When AMDIS performs library search, it will now require a match on both spectrum and RI (within the tolerance).
- Result Interpretation: Compounds that pass both spectral and RI matching are assigned high confidence (Level 2 in modern frameworks) [22]. Compounds with a good spectral match but an RI mismatch should be flagged as potential false positives and rejected or reported with low confidence.
Protocol 3: Implementing a Post-Deconvolution Confidence Scoring Framework For high-stakes research (e.g., biomarker discovery), implementing a formal confidence scoring framework like the one adapted for GC-HRMS is recommended [22].
- Define Confidence Levels: Adopt a standard schema. For example:
- Level 1: Confirmed by authentic standard (matched RT, RI, and spectrum).
- Level 2: Probable structure (matched spectrum & RI, or MS/MS evidence).
- Level 3: Tentative candidate (spectral match only, or formula match).
- Level 4: Unknown feature (distinct m/z and RT only).
- Process with AMDIS: Identify compounds using AMDIS with RI filtering.
- Apply Secondary Filters: Manually or using complementary software (e.g., MetaBox with its PScore algorithm [24]), check for additional evidence:
- Isotopic Pattern Match: Does the observed isotopic pattern match the proposed formula?
- Ion Abundance Ratios: Do the ratios of key fragment ions match the reference spectrum?
- Presence in Blanks: Is the compound also present in procedural blanks? (This can indicate contamination).
- Assign Final Confidence: Synthesize all lines of evidence to assign a final confidence level to each annotation. Report these levels explicitly in your research findings.
Visualization of Workflows and Relationships
Diagram 1: AMDIS Deconvolution and False Positive Reduction Workflow
Diagram 2: Multi-Evidence Confidence Scoring Framework for Annotation [22]
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials & Reagents for Optimized GC-MS Deconvolution Research
| Item | Function & Purpose in False Positive Reduction | Example / Specification |
|---|---|---|
| Retention Index Standard Mixture | Provides anchor points for calculating compound-specific RIs, enabling the powerful RI filter to distinguish between co-eluting isomers and false spectral matches [22] [25]. | n-Alkane Series (e.g., C8-C30 or C8-C40 in hexane). FAME Mixes for fatty acid analysis. |
| Well-Characterized Calibration/Quality Control Mix | Used to empirically determine optimal Component Width and Sensitivity parameters for your specific instrument and method, establishing a reliable baseline. | MegaMix (contains ~76 compounds) [25], Grobs Mix, or a custom mixture representing your analyte classes. |
| RI-Enabled Spectral Library | A searchable database containing not only mass spectra but also reference RI values for compounds on specific stationary phases. Essential for Protocol 2. | NIST GC Method/Retention Index Database [25], FiehnLib [24], or in-house libraries built with authentic standards. |
| Application-Specific GC Column | The choice of stationary phase is the primary factor affecting selectivity (α), which drives chromatographic resolution and reduces co-elution—the root cause of difficult deconvolution [28]. | Choose based on analyte polarity. E.g., Rtx-5ms (5% phenyl) for general use; Stabilwax (polyethylene glycol) for polar compounds; Rtx-200 for halogens [28]. |
| Deuterated or ¹³C-Labeled Internal Standards | Corrects for analyte losses during sample preparation and matrix effects during ionization. Improves quantitative accuracy, which aids in distinguishing true low-abundance signals from noise. | Use for targeted quantitation of key biomarkers. Select standards that are chemically identical to analytes but with distinct mass shifts. |
| Post-Deconvolution Validation Software | Tools that apply additional statistical checks, consolidate results from multiple files, or implement advanced scoring algorithms (like PScore [24]) to filter AMDIS output. | MetaBox R package [24], iMatch (for RI filtering) [25], or commercial vendor software with batch processing and advanced reporting. |
In gas chromatography-mass spectrometry (GC-MS) metabolomics, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a foundational tool for peak deconvolution in complex chromatograms. However, a significant and well-documented limitation of AMDIS is its tendency to produce false positives and leave missing values when peaks are detected in only a subset of samples within an analysis set [29] [23]. These errors introduce noise and uncertainty, complicating data interpretation and potentially leading to incorrect biological conclusions. For researchers and drug development professionals, where accuracy is paramount, this represents a critical bottleneck.
The implementation of a targeted custom analyte database directly addresses this core issue. By shifting from broad, untargeted library searches to a focused identification process using a verified, context-specific library, researchers can drastically reduce false identifications. A custom library serves as a precise filter, ensuring that the software compares experimental spectra against a curated set of known, relevant compounds. This targeted approach is a cornerstone methodology for enhancing the reliability, reproducibility, and overall power of GC-MS-based research in reducing false positives from AMDIS deconvolution.
Technical Support Center: Troubleshooting Custom Analyte Databases
This support center provides targeted solutions for common challenges encountered during the creation, implementation, and maintenance of custom GC-MS analyte databases.
Troubleshooting Guides
Guide 1: Resolving High Rates of False Positive Identifications
- Problem: After implementing a custom library, AMDIS continues to report a high number of false positive compound matches.
- Diagnosis & Solution: This typically indicates issues with library specificity or deconvolution parameters. Follow this logical troubleshooting pathway:
Guide 2: Addressing Missing Values or Inconsistent Peak Integration
- Problem: The custom library fails to identify compounds that are visually present in the chromatogram, or peak areas are inconsistent across samples.
Diagnosis & Solution: This problem often stems from alignment issues or inconsistent deconvolution. Systematic verification is key [29].
- Perform Manual Inspection: Use software tools (like those described by Behrends et al. [29]) to visually inspect the chromatographic peaks across all samples to confirm data quality and the presence of the missed analyte.
- Check Retention Time Alignment: Ensure all chromatograms are properly aligned. Slight shifts can cause the library to miss a match. Recalibrate using internal standards.
- Audit the Library Entry: Verify that the retention time or retention index for the missing compound in your custom library is correct for your current analytical method (column, temperature program).
- Investigate Integration Parameters: If a peak is found but not integrated consistently, examine and adjust the integration and peak-picking settings within AMDIS or your downstream processing software to ensure robust peak detection across varying baselines and signal-to-noise levels.
Frequently Asked Questions (FAQs)
Q1: What are the primary advantages of a custom library over a large commercial library for targeted studies?
- A: A custom library reduces search space, minimizing false positives from structurally similar but irrelevant compounds. It ensures all entries are directly applicable to your research context (e.g., specific drug metabolites, pathway intermediates), and allows you to incorporate proprietary compounds not found in commercial libraries.
Q2: How do I handle error reporting or validation within my custom database workflow?
- A: Implementing structured error-checking is crucial [30]. During database building, validate entries against known standards. During analysis, use manual inspection tools to confirm identifications [29]. For scripting or automated workflows, define clear error codes (e.g., for missing files, failed calibrations) and use logging (
console.logstatements or equivalent) to trace and diagnose issues in real-time [30].
- A: Implementing structured error-checking is crucial [30]. During database building, validate entries against known standards. During analysis, use manual inspection tools to confirm identifications [29]. For scripting or automated workflows, define clear error codes (e.g., for missing files, failed calibrations) and use logging (
Q3: My database performance has slowed down significantly after adding many entries. How can I optimize it?
- A: This mirrors general database optimization principles [31]. First, monitor to identify the bottleneck (e.g., query speed, memory). Ensure your data files and library index are organized efficiently. If using a relational database backend, analyze slow queries, optimize their structure, and implement proper indexing on frequently searched fields like
m/zorRetention_Indexto dramatically speed up searches [31].
- A: This mirrors general database optimization principles [31]. First, monitor to identify the bottleneck (e.g., query speed, memory). Ensure your data files and library index are organized efficiently. If using a relational database backend, analyze slow queries, optimize their structure, and implement proper indexing on frequently searched fields like
Q4: What is the most critical step to perform before making changes to an existing custom database?
- A: Always create a full backup of your database before making any structural or content changes [32]. Changes made directly to database files can be difficult or impossible to undo and may corrupt the library. If possible, test modifications on a copy or development version first.
Experimental Protocols & Data
Core Protocol: Building a Custom GC-MS Analyte Library
This protocol details the creation of a targeted, in-house library from analytical standards.
Materials: Pure analytical standards of target compounds, suitable GC-MS system, derivatization agents (if needed, e.g., MSTFA for trimethylsilylation), internal standard mixture, data processing software (AMDIS, NIST MS Search, etc.).
Procedure:
- Standard Preparation: Prepare individual and mixture solutions of analytical standards at known concentrations. Include a retention index marker mixture (e.g., n-alkanes for Kovats Index).
- GC-MS Analysis: Inject each solution using your standardized GC-MS method. Ensure optimal chromatographic separation and MS signal.
- Spectra Acquisition & Deconvolution: For each analyte peak, use AMDIS to deconvolute the mass spectrum from co-eluting compounds and background. Set the deconvolution parameters (component width, sensitivity, shape requirements) stringently to obtain a "pure" spectrum.
- Library Entry Creation:
- Essential Metadata: Enter the compound name, CAS number, chemical formula, and exact mass.
- Chromatographic Data: Record the observed retention time and the calculated retention index (RI) based on the alkane markers. The RI is more reproducible than absolute retention time.
- Spectral Data: Import the deconvoluted mass spectrum (including the full m/z range and relative abundances).
- Validation: Annotate the entry with the source concentration and a quality flag (e.g., "Verified by Pure Standard").
- Database Curation: Compile individual entries into a single library file (.msl or .msp format). Perform a final review to remove duplicate or low-quality entries.
Performance Metrics: Custom vs. General Library
The following table quantifies the typical impact of implementing a targeted custom database on data quality in a GC-MS metabolomics study.
Table 1: Comparative Performance of a Custom Targeted Library vs. a General Purpose Library in AMDIS Deconvolution
| Performance Metric | General Purpose Library (e.g., NIST) | Custom Targeted Library | Impact on Research |
|---|---|---|---|
| False Positive Rate | High (15-30% typical) | Low (<5% achievable) | Drastically reduces erroneous identifications, increasing data reliability [29] [23]. |
| Missing Value Rate | High for low-abundance/target compounds | Very Low | Minimizes gaps in data matrices, enabling more robust statistical analysis [29]. |
| Identification Speed | Slower (searches large library) | Faster (searches focused library) | Improves workflow efficiency. |
| Method Relevance | Low (contains many irrelevant compounds) | High (100% applicable to study) | Ensures identifications are biologically relevant to the specific research context. |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Custom Library Development and GC-MS Analysis
| Item | Function & Importance |
|---|---|
| Certified Pure Analytical Standards | The foundation of the library. Provides the reference spectra and retention index for unambiguous identification of target analytes. |
| Retention Index Marker Kit | A homologous series (e.g., C8-C40 n-alkanes) used to calculate compound-specific retention indices (RI). RI is more reproducible across instruments and over time than absolute retention time, making the library robust. |
| Derivatization Reagents | For analyzing non-volatile metabolites (e.g., amino acids, organic acids). Reagents like MSTFA (N-Methyl-N-(trimethylsilyl)trifluoroacetamide) increase volatility and thermal stability, enabling GC-MS analysis and generating reproducible mass spectra for the library. |
| Stable Isotope-Labeled Internal Standards | Added to every sample prior to processing. Corrects for variability in extraction, derivatization, and instrument response. Essential for achieving accurate quantitative data alongside identifications. |
| Quality Control (QC) Pooled Sample | A pooled aliquot of all experimental samples. Run repeatedly throughout the analytical sequence to monitor instrument stability, retention time drift, and overall data quality over time. |
System Optimization and Implementation Workflow
Successfully integrating a custom library into a research pipeline requires careful planning. The following diagram outlines the complete workflow from initial setup to validated implementation, highlighting optimization checkpoints.
In gas chromatography-mass spectrometry (GC-MS) analysis, particularly in metabolomics and volatile organic compound (VOC) profiling, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a widely used, free tool for separating co-eluting peaks and identifying components [2]. However, its high sensitivity is a double-edged sword: while it excels at deconvolution, it is also prone to generating a high number of false-positive identifications [2] [33]. This occurs when the software incorrectly matches spectral noise or fragment ions of one compound to a similar spectrum in a large, generic commercial library. Studies have noted that indiscriminate use of AMDIS can generate 70–80% false assignments [33].
This high false-positive rate has significant consequences within a research thesis focused on method validation. It compromises data integrity, leads to wasted time manually curating results, and obscures true biological or chemical signals. Therefore, a core strategy for robust GC-MS deconvolution research involves refining the identification library itself. This case study demonstrates how building a custom, application-specific VOC library for AMDIS directly addresses this thesis problem, dramatically improving accuracy and efficiency [2].
Core Case Study: The AMDIS Strawberry VOC User Library
A 2024 study on strawberry aroma profiling provides a quantifiable benchmark for the effectiveness of a custom library [2]. Researchers developed a bespoke "Strawberry VOC User Library" for AMDIS containing 104 specific volatile compounds known to be relevant to strawberry aroma, complete with mass spectra, retention indices, and odor descriptors.
The performance of this targeted library was directly compared against a broad commercial library. The results, summarized in the table below, show transformative improvements in data processing and reliability [2].
Table 1: Performance Comparison: Custom Library vs. Commercial Library
| Metric | Commercial Library | Custom Strawberry Library | Improvement |
|---|---|---|---|
| Reported False Hits | ~200 (estimated) | ~0 | Reduced by ~200 |
| Analysis Output File Size | Not specified, but large | >96% smaller | Reduced by >96% |
| AMDIS Processing Time per Sample | 31 seconds | 9 seconds | 71% reduction |
Experimental Protocol for Library Creation and Validation [2]:
- Sample Collection: VOC data were gathered from 61 different strawberry cultivars harvested in South Korea.
- GC-MS Analysis: Volatiles were collected and analyzed using GC-MS, with data saved in the standard .cdf (netCDF) format compatible with AMDIS.
- Library Curation: A target list of 104 strawberry-relevant VOCs was compiled. For each compound, its mass spectrum and experimentally determined Retention Index (RI) were entered into a user library file (.MSL format).
- Method Comparison: The same set of sample data files (.cdf) was processed twice in AMDIS: once using only the large commercial library and once using the custom strawberry library.
- Output Analysis: The resulting reports from both runs were compared. The custom library eliminated hundreds of irrelevant "hits," produced a concise report containing only the target compounds, and processed the data over three times faster.
Technical Support Center
3.1 Troubleshooting Guide: Common AMDIS Deconvolution Issues
Issue: High False Positive Identification Rate
- Cause: AMDIS is matching spectra against an overly broad library (e.g., NIST) containing many irrelevant compounds [2] [33].
- Solution: Create a custom user library. Limit the search space to compounds relevant to your sample matrix (e.g., metabolites in your organism, VOCs in your food type). Always include experimentally measured Retention Indices (RI) for each compound to provide a second orthogonal filter, greatly increasing confidence in identifications [2] [33].
Issue: Inconsistent or Missed Detections Across Samples
- Cause: Instrumental drift over time (changes in column performance, ion source cleanliness) alters retention times and response factors [34].
- Solution: Implement a quality control (QC) protocol. Analyze a pooled QC sample at regular intervals throughout your batch run. Use this data to correct for retention time shifts and signal drift using algorithms like Random Forest regression, which has been shown to provide stable long-term correction [34].
Issue: Poor Deconvolution of Severely Co-eluting Peaks
- Cause: AMDIS's empirical algorithms may struggle to resolve peaks with near-identical retention times and overlapping spectra [33].
- Solution: Employ a complementary chemometric tool. Use techniques like Multivariate Curve Resolution - Alternating Least Squares (MCR-ALS) or Ratio Analysis of Mass Spectrometry (RAMSY). These methods can be applied to the same data to deconvolute complex peak clusters that AMDIS may not fully resolve, recovering low-intensity ions from co-eluted compounds [21] [33].
Issue: Analysis is Excessively Time-Consuming
- Cause: Manual, expert-led deconvolution and review of large datasets can take 60-120 minutes per sample [35].
- Solution 1: Adopt a targeted custom library, which drastically cuts AMDIS processing and report review time [2].
- Solution 2: Explore machine learning (ML) automation. Emerging deep learning approaches, such as convolutional neural networks (CNNs), can be trained to recognize specific VOC patterns directly from raw GC-MS data, bypassing traditional deconvolution and offering rapid, automated screening [35] [36].
3.2 Frequently Asked Questions (FAQs)
Q: My custom library eliminated false positives but also missed some compounds I know are present. What happened?
- A: This is likely due to improper Retention Index (RI) matching tolerance. The RI in your library must be calibrated to your specific GC method. Ensure you are using the correct homologous series of alkanes (or FAMEs) and that the RI tolerance window in AMDIS settings is set appropriately (e.g., ±10 units). An overly strict tolerance will reject correct matches [33].
Q: Can I use a custom library for non-targeted screening?
- A: A custom library is inherently a targeted screening tool. For true non-targeted analysis (NTA), you must use comprehensive commercial libraries and advanced data processing workflows, often involving high-resolution MS (HRMS) and machine learning models to prioritize unknown features [37]. However, a well-curated custom library can serve as an excellent first-pass filter in an NTA workflow to quickly annotate known compounds before investigating unknowns.
Q: How do I handle batch effects and instrument drift in a long-term study?
- A: This is a critical step for reproducible research. The most robust method is to use pooled Quality Control (QC) samples. Analyze a QC repeatedly throughout your batch sequence. Then, apply data correction models (e.g., based on Random Forest or Support Vector Regression) that use batch number and injection order to normalize peak areas across the entire dataset, compensating for instrumental drift [34].
Q: Are there automated alternatives to AMDIS for VOC detection?
- A: Yes, the field is moving toward greater automation. Machine learning strategies, particularly Convolutional Neural Networks (CNNs), show great promise. These can be trained on raw GC-MS data (treated as images) to automatically detect target VOCs, offering high specificity and speed while reducing subjective human intervention [35] [36]. Chemometric models like PARAFAC2 (e.g., implemented in PARADISe software) also offer automated, model-based deconvolution [21].
Visualizing the Workflow Improvement
The following diagrams contrast the standard and optimized workflows, highlighting where the custom library intervenes to enhance efficiency and accuracy.
Standard vs. Custom Library Workflow for AMDIS
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Reliable GC-MS Deconvolution Research
| Item | Function & Role in Reducing False Positives | Key Considerations |
|---|---|---|
| AMDIS Software | The primary, free deconvolution tool. Its performance is directly enhanced by paired custom libraries [2]. | User-friendly but requires parameter optimization and a good library for best results [33]. |
| Custom User Library (.MSL) | The core solution. Limits database search to relevant compounds, using Retention Index (RI) as a critical second filter [2]. | Must be built with experimentally derived RI and high-quality spectra from standards or verified samples. |
| Retention Index Standards | A homologous series (e.g., n-alkanes C8-C30 or FAME mix) used to calculate compound-independent RIs for library building and validation [33]. | Essential for aligning data across different methods, instruments, and over time, correcting for retention time drift. |
| Derivatization Reagents | Chemicals like MSTFA (for trimethylsilylation) modify polar metabolites for stable, volatile GC-MS analysis [33]. | Standardized derivatization is crucial for reproducible spectra that match library entries. |
| Quality Control (QC) Sample | A pooled sample representing the study's composition, run repeatedly to monitor and correct for instrumental drift [34]. | Enables the use of advanced data correction algorithms (e.g., Random Forest) to ensure long-term data stability [34]. |
| Chemometric Software (e.g., for MCR-ALS, PARAFAC2) | Provides advanced, model-based deconvolution for separating complex, co-eluting peaks that challenge traditional methods [21] [33]. | Used as a complementary tool to AMDIS to resolve difficult peak clusters and verify identifications. |
Integrating Retention Indices (RI) as Orthogonal Filters for Enhanced Specificity
Technical Support Center
This technical support center provides troubleshooting guides, FAQs, and detailed protocols to help researchers effectively integrate Retention Index (RI) filtering into their GC-MS AMDIS workflows. This integration is a critical strategy for reducing false positive identifications in complex mixture analysis, such as in metabolomics and natural product research [10] [33].
Troubleshooting Guides
Common Issue 1: Unstable or Drifting Retention Times (RT) and Retention Indices (RI) A stable RI system is foundational for reliable filtering. Shifts in RT compromise RI calculations and the validity of your orthogonal filter.
- Symptoms: Consistent drift in compound RTs across runs; calculated RIs for standard alkanes do not match expected values; increased false negative identifications after RI filtering.
- Diagnosis & Solution:
- Check System Conditioning: After instrument idle periods (e.g., overnight, weekends), RTs can be unstable. Perform 2-3 conditioning injections of your solvent or a standard at the beginning of a sequence to equilibrate the system [38].
- Verify Carrier Gas Flow: Ensure carrier gas is flowing continuously at a stable rate. Consider using instrument "sleep modes" that maintain minimal flow during idle times to preserve column conditioning [38].
- Inspect and Maintain Hardware: Check for leaks, clean or replace the syringe if injection is not smooth, and ensure the inlet liner and seal are in good condition. Contamination can cause RT shifts mid-sequence [38].
Common Issue 2: High False Positive Rate Persists After Applying RI Filter If RI filtering does not sufficiently reduce false calls from AMDIS, the filter parameters or calibration may be misapplied.
- Symptoms: AMDIS reports many identifications with poor spectral matches that are not filtered out; manual review shows implausible compounds for the sample type.
- Diagnosis & Solution:
- Calibrate RI System with Correct Standards: Use a homologous series (e.g., n-alkanes, FAME mix) that brackets your analyte RT range [10] [39]. Ensure peaks are correctly identified and integrated.
- Apply Validated ΔRI Thresholds: Use evidence-based thresholds, not arbitrary windows. For high confidence, require |ΔRI| ≤ 20 between the experimental and library value. Tentatively accept identifications with 20 < |ΔRI| ≤ 50, and reject those with |ΔRI| > 50 [40].
- Use a Targeted Library: Employ a custom, sample-relevant user library with AMDIS. One study showed a custom strawberry VOC library reduced potential false hits by 200 compared to a commercial library [2].
Common Issue 3: RI Filter Rejects Correct Identifications (False Negatives) Overly stringent filtering can discard correct identifications, especially for compounds where the reference RI is poorly defined.
- Symptoms: Known compounds spiked into the sample or expected metabolites are rejected by the RI filter.
- Diagnosis & Solution:
- Review Library RI Quality: Check the source and number of data points for the reference RI in your database. NIST libraries now include AI-predicted RIs to fill coverage gaps [39]. Prefer library entries with experimental RI values.
- Widen ΔRI Threshold for Critical Compounds: For compounds of key interest, use a less stringent ΔRI threshold (e.g., ≤ 50) and flag them for confirmation via other means (e.g., standard injection).
- Verify Calibration Curve Fit: Ensure the regression fit (e.g., linear, quadratic) for your RI calibration standards is accurate across the entire RT range, especially at the extremes.
Common Issue 4: Poor Deconvolution of Co-eluting Peaks by AMDIS AMDIS can struggle with severely co-eluting peaks, leading to missed or misidentified compounds, which then affects downstream RI filtering [10].
- Symptoms: Broad or asymmetrical peaks; AMDIS reports low Match Factors (MF) or fails to deconvolute peaks; RAMSY or PARAFAC2 analysis reveals hidden components [10] [21].
- Diagnosis & Solution:
- Optimize AMDIS Parameters: Systematically adjust deconvolution parameters (width, shape, sensitivity) using a design of experiments approach for your specific sample type and instrument method [10].
- Employ Complementary Algorithms: Use a second deconvolution method like Ratio Analysis of Mass Spectrometry (RAMSY) for problematic peak clusters. RAMSY can recover low-intensity, co-eluted ions that AMDIS misses [10].
- Consider Advanced Chemometric Tools: For complex datasets, explore algorithms like PARAFAC2 (e.g., via PARADISe software) or Multivariate Curve Resolution-Alternating Least Squares (MCR-ALS), which are designed for automated resolution of complex mixtures [21].
Table: Troubleshooting Common RI and AMDIS Issues
| Problem | Likely Cause | Immediate Action | Long-term Solution | ||
|---|---|---|---|---|---|
| RT Drift at Start of Run | Column not equilibrated [38] | Run 2-3 conditioning injections | Implement automated conditioning sequence | ||
| RI Filter Ineffective | Incorrect ΔRI threshold [40] | Apply | ΔRI | ≤ 20 rule | Build custom RI library for your column/conditions |
| AMDIS High False Positives | Library too broad [2] | Use a targeted user library | Optimize AMDIS settings & combine with RAMSY [10] | ||
| Poor Peak Deconvolution | Severe co-elution [10] | Manually integrate problem region | Use advanced chemometrics (PARAFAC2, MCR-ALS) [21] |
Frequently Asked Questions (FAQs)
Q1: What is a Retention Index (RI), and why is it more reliable than absolute retention time? An RI is a unitless number that expresses a compound's retention relative to a series of standard compounds (e.g., n-alkanes) analyzed under the same conditions. It is normalized against the scale defined by the standards, where alkanes are assigned an RI of 100 times their carbon number. RI is more reliable than absolute retention time because it is less sensitive to minor fluctuations in carrier gas flow, temperature gradients, and column degradation, providing a more reproducible metric for compound identification across different instruments and over time [10].
Q2: How does RI filtering specifically reduce false positives in AMDIS deconvolution? AMDIS identifies compounds primarily by matching mass spectra. In complex samples, spectra can be similar or mixed due to co-elution, leading to false matches. An RI filter adds a second, independent (orthogonal) identification parameter. After AMDIS proposes an identity based on spectrum, the system checks if the experimentally measured RI of the peak matches the known RI for that compound within a defined tolerance. Mismatches are flagged or rejected, significantly increasing identification confidence. One tool, iMatch, automates this by statistically filtering AMDIS results based on RI databases [41].
Q3: What are the recommended thresholds for accepting an RI match? Research on a large dataset of confirmed compounds provides clear guidance: A difference (|ΔRI|) of 20 units or less between experimental and library RI strongly supports the identification. A |ΔRI| of greater than 50 indicates a very low probability of a correct match and the identification should be rejected. Differences between 20 and 50 are ambiguous and the identification should be considered tentative and uncorroborated by RI [40].
Q4: My sample is very complex. AMDIS still gives many uncertain identifications even with RI. What are my options? You can adopt a tiered deconvolution strategy:
- Optimize & Filter: First, optimize AMDIS parameters for your sample and apply strict RI filtering [10].
- Complementary Deconvolution: For unresolved peak clusters, apply a second, complementary algorithm like RAMSY, which uses ratio analysis of mass spectra across samples to pull apart co-eluting components [10].
- Advanced Chemometrics: Consider next-generation tools like PARAFAC2 (implemented in PARADISe) or MCR-ALS, which are designed for automated resolution of complex data and are increasingly seen as the road to automation in GC-MS analysis [21].
Q5: Are there automated or machine learning approaches to improve this workflow? Yes, the field is rapidly advancing. Key developments include:
- AI-Augmented Libraries: New versions of mass spectral libraries (e.g., NIST23) use AI models to predict RIs for compounds lacking experimental data, greatly expanding RI filter coverage [39].
- Automated Calibration: Tools are being developed to automatically locate and assign n-alkane peaks in chromatograms, even with background interference, simplifying RI system calibration [39].
- Retention Time Prediction: Advanced multimodal machine learning models can now predict GC retention times with high accuracy (R² > 0.99) using molecular structure and temperature program data. This can guide method development and provide a predicted RI for unknown compounds [42].
Table: Quantitative Guidelines for RI Filtering Based on ΔRI [40]
| Absolute Difference | ΔRI | Interpretation | Action | |
|---|---|---|---|---|
| ≤ 20 | High identification precision. | Accept as RI-corroborated. | ||
| 20 to 50 | Indiscriminate/Uncertain. | Tentative identity (not RI-corroborated). | ||
| > 50 | Very low identification precision. | Reject as a false positive. |
Experimental Protocols
Protocol 1: Sample Preparation for Plant Metabolomics with RI Standard Addition
This protocol is adapted from GC-MS metabolomics studies on plant extracts and includes steps for adding Retention Index markers [10] [33].
1. Derivatization (Methoximation and Silylation):
- Materials: Dried plant extract, O-methylhydroxylamine hydrochloride (in pyridine), N-Methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA) with 1% Trimethylchlorosilane (TMCS), pyridine.
- Procedure: a. Add 10 μL of methoxyamine hydrochloride solution (40 mg/mL in pyridine) to the dried sample. b. Vortex and incubate at 30°C for 90 minutes (methoximation). c. Add 90 μL of MSTFA + 1% TMCS. d. Vortex and incubate at 37°C for 30 minutes (trimethylsilylation).
2. Retention Index Standard Addition:
- Materials: Fatty Acid Methyl Ester (FAME) mix (C8-C30) or n-alkane mix.
- Procedure:
a. Add 2.0 μL of the FAME mixture (or a similar RI standard mixture) directly to the derivatized sample.
b. Vortex thoroughly to mix.
- Purpose: This adds known compounds at regular intervals across the chromatogram. Their known RIs (e.g., FAME-C8 = 800, FAME-C12 = 1200, etc.) are used to construct a calibration curve for converting sample peak retention times to retention indices.
Protocol 2: Establishing and Validating an RI Filter for AMDIS Results
This protocol details the post-processing steps to apply an RI filter.
1. RI System Calibration:
- Run a standard mixture containing your RI markers (e.g., FAME mix) using your analytical GC-MS method.
- In your data analysis software, record the absolute retention time (RT) of each standard.
- Generate a calibration curve by plotting the known RI of each standard (y-axis) against its RT (x-axis). Fit a linear or polynomial regression. The equation of this curve will be used to convert RT to RI for all subsequent sample runs.
2. AMDIS Analysis with Targeted Library:
- Analyze your sample data with AMDIS using an optimized parameter set [10].
- Utilize a targeted user library if possible. Creating a custom library of expected compounds for your sample type (e.g., a strawberry VOC library) has been shown to dramatically reduce false positives and processing time compared to using a broad commercial library [2].
3. Apply the RI Filter:
- For each compound identified by AMDIS, use the calibration curve to calculate its experimental RI from its RT.
- Retrieve the reference RI for the proposed compound from a reliable database (e.g., NIST, a custom library).
- Calculate the absolute difference (|ΔRI|).
- Apply the decision thresholds:
- Accept if |ΔRI| ≤ 20.
- Flag for manual review if 20 < |ΔRI| ≤ 50.
- Reject if |ΔRI| > 50 [40].
Workflow Visualizations
Workflow for Integrating RI Filtering with AMDIS Results
The Scientist's Toolkit
Table: Key Research Reagent Solutions for RI-Enhanced GC-MS Metabolomics
| Reagent / Material | Function / Purpose | Key Note |
|---|---|---|
| O-methylhydroxylamine hydrochloride | Methoximation agent. Protects carbonyl groups (aldehydes, ketones) in metabolites by converting them to methoximes, preventing ring formation in sugars and improving chromatographic behavior and stability [10] [33]. | Typically used in a solution with pyridine. First step of a two-step derivatization. |
| MSTFA with 1% TMCS | Silylation agent. Replaces active hydrogens (e.g., in -OH, -COOH, -NH groups) with trimethylsilyl (TMS) groups. This volatilizes polar metabolites, making them amenable to GC analysis [10] [33]. | TMCS acts as a catalyst. Second step of derivatization. |
| FAME Mixture (C8-C30) | Retention Index (RI) calibration standards. A series of fatty acid methyl esters of known RI eluting across the chromatographic run time. Used to construct the RT-to-RI calibration curve [10]. | Added directly to the sample post-derivatization. Enables calculation of experimental RI for all detected peaks. |
| Targeted/User Library | Custom spectral/RI database. A curated library containing mass spectra and reference RIs for compounds expected in a specific sample type (e.g., strawberry VOCs, human serum metabolites) [2]. | Dramatically improves AMDIS specificity and speed versus a universal library [2]. |
| n-Alkane Mixture | Alternative RI calibration standards. A homologous series of straight-chain alkanes (e.g., C7-C30). Define the classic Kovats RI scale where RI = 100 × carbon number [39]. | Common alternative to FAME mix. Used to calibrate the RI system independently of the sample. |
Advanced Troubleshooting: Fine-Tuning Detection and Managing Match Factors
Welcome to the GC-MS Deconvolution Technical Support Center
This resource is designed for researchers focused on reducing false positives in GC-MS data analysis using the Automated Mass Spectral Deconvolution and Identification System (AMDIS). The following guides and FAQs address common experimental challenges, framed within a thesis on optimizing the balance between detection sensitivity and analytical specificity.
Troubleshooting Guides & FAQs
FAQ 1: My AMDIS analysis is producing too many false positives (>70% of hits). How can I improve specificity without missing true low-abundance compounds? This is a common issue often stemming from non-optimized deconvolution settings or complex co-elution [33].
- Diagnosis: High false positive rates frequently occur when AMDIS parameters are too permissive, incorrectly deconvoluting noise or interfering signals as compounds.
- Solution - Systematic Parameter Optimization:
- Employ a Factorial Design: Do not adjust parameters arbitrarily. Use a fractional factorial design of experiments to systematically test combinations of key AMDIS settings (Component Width, Adjacent Peak Subtraction, Resolution, and Sensitivity) against a standard mixture [33].
- Apply a Heuristic Filter: Develop or apply a compound detection factor (CDF). This metric, calculated from match factor (MF) and retention index (RI) deviation, helps filter out low-probability hits. For example, setting a threshold of
CDF = (MF/1000) / RI_deviationcan effectively suppress false IDs [33]. - Implement a Complementary Algorithm: For severely co-eluted peaks, use AMDIS output as a first pass. Then, apply a statistical deconvolution tool like Ratio Analysis of Mass Spectrometry (RAMSY) to the raw data for problematic regions. RAMSY excels at extracting pure spectra from overlapping peaks by analyzing intensity ratios across samples, recovering signals AMDIS may miss [33].
- Preventative Practice: Always use Retention Time Index (RI) libraries instead of absolute retention times and ensure your calibration is performed with every batch.
FAQ 2: How do I choose the right threshold for peak detection in a noisy chromatogram to avoid false negatives (missed peaks)? Choosing a fixed threshold is often insufficient for variable baselines. The goal is to maximize true positives while controlling false positives [43].
- Diagnosis: A single, static signal-to-noise (S/N) threshold may be too high for low-concentration analytes (causing false negatives) or too low in noisy regions (causing false positives).
- Solution - Adaptive Thresholding & Advanced Algorithms:
- Use Savitzky-Golay or Wavelet Filters: Smooth the chromatogram using a Savitzky-Golay filter (which preserves peak shape and height) or a wavelet transform (which isolates signal from noise across frequencies) before peak detection [44].
- Apply an Adaptive Threshold: Calculate the threshold dynamically as a multiple (e.g., 3x to 5x) of the local noise, estimated from a moving window of the chromatogram's baseline regions [44].
- Leverage Multivariate Methods: For complex datasets, employ chemometric techniques like Multivariate Curve Resolution - Alternating Least Squares (MCR-ALS). MCR-ALS models the total data as a sum of individual component profiles, effectively resolving co-eluted compounds and improving detection limits in noisy data [21].
- Verification: Spike a known standard at a concentration near the expected limit of detection (LOD) into your sample matrix. Your optimized method should consistently detect this standard.
FAQ 3: What is the most effective strategy to validate my deconvolution results and provide a reliable false positive estimate? Validation is critical for credible results. A single method is prone to systematic errors [43].
- Diagnosis: Relying solely on the match factor from a single library search does not statistically validate an identification.
- Solution - Orthogonal Verification & Decoy Strategies:
- Use Orthogonal Data: Whenever possible, confirm identifications by analyzing the sample with a different technique (e.g., GC-MS with a different stationary phase, or LC-MS) or by obtaining MS/MS spectra if your instrument is capable.
- Employ a Decoy Database Approach: Borrow a powerful concept from proteomics. Create a "decoy" spectral library by reversing or scrambling the spectra in your target library. Process your data against the combined target/decoy library. True identifications should only hit the target library, while false positives will hit both equally. The number of decoy hits provides a direct estimate of the false discovery rate (FDR) in your results [45].
- Adopt Advanced Automated Systems: For high-throughput work, consider platforms like PARADISe (PARAFAC2-based Deconvolution and Identification System), which uses the PARAFAC2 model to provide mathematically rigorous deconvolution with inherent validation of the multilinear model structure [21].
FAQ 4: My sample matrix is suppressing analyte signals and causing variable recovery. How can I improve sensitivity and robustness? Matrix effects are a major source of sensitivity loss and irreproducibility [46].
- Diagnosis: Co-extracted matrix components can suppress or enhance ionization, affect chromatographic peak shape, and clog the instrument.
- Solution - Enhanced Sample Preparation & Internal Standards:
- Optimize Sample Cleanup: Move beyond simple dilution. For complex plant extracts, rigorous techniques like solid-phase extraction (SPE) are often necessary to remove interfering pigments, lipids, and resins [46] [33].
- Use Matrix-Matched Calibration & Internal Standards: Prepare calibration standards in a blank matrix extract. Use deuterated or other stable isotope-labeled analogs of your target analytes as internal standards (IS). The IS corrects for losses during sample preparation and ionization suppression/enhancement during analysis.
- Derivatization Efficiency: For GC-MS, ensure derivatization (e.g., silylation) is complete and reproducible. Use fresh reagents, control moisture, and consider adding a derivatization standard to monitor the reaction [33].
Experimental Protocol: Optimized GC-MS Metabolite Profiling with AMDIS/RAMSY
This protocol details a method for analyzing plant metabolites, combining optimized AMDIS deconvolution with RAMSY analysis to maximize reliable identifications [33].
1. Sample Preparation (Derivatization for GC-MS):
- Materials: Dry plant extract, pyridine (silylation grade), methoxyamine hydrochloride, N-Methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA) with 1% trimethylchlorosilane (TMCS), fatty acid methyl ester (FAME) RI standards, internal standard (e.g., ribitol or deuterated compound) [33].
- Procedure:
- Add 50 µL of methoxyamine solution (40 mg/mL in pyridine) to the dried extract. Vortex and incubate at 30°C for 90 minutes.
- Add 70 µL of MSTFA (+1% TMCS). Vortex and incubate at 37°C for 30 minutes.
- Add 2.0 µL of FAME mixture and your chosen internal standard. Vortex thoroughly and transfer to a GC vial [33].
2. GC-MS Data Acquisition:
- Instrument: Agilent 7890A GC coupled to a 5975C MSD (or equivalent).
- Column: DB-5MS (30 m × 0.25 mm i.d., 0.25 µm film thickness).
- GC Program: Hold at 70°C for 5 min, ramp at 5°C/min to 310°C, hold for 5 min.
- Inlet: 250°C, splitless mode.
- MS Source: 230°C, quadrupole at 150°C, electron ionization at 70 eV, scan range m/z 50-600 [33].
3. Data Analysis Workflow:
1. AMDIS Parameter Optimization: Use a standard mixture of known metabolites. Run a factorial design (e.g., testing Component Width: low/medium/high; Sensitivity: low/medium/high). Select the parameter set yielding the highest correct identifications and match factors for the standards.
2. Primary Deconvolution: Process all sample chromatograms through AMDIS using the optimized settings. Search against a target library (e.g., NIST, FiehnLib) with a Retention Index window (e.g., ±10 units).
3. Heuristic Filtering: Apply a Compound Detection Factor (CDF) to the AMDIS results. Example: CDF = (Match Factor / 1000) / abs(ΔRI). Filter out hits below a validated CDF threshold (e.g., < 0.5) [33].
4. Secondary Deconvolution with RAMSY: For chromatographic regions where AMDIS reported poor deconvolution (low MF) or suspected co-elution (broad/shouldering peaks), apply the RAMSY algorithm. RAMSY analyzes intensity ratios across multiple related samples (e.g., biological replicates) to resolve pure component spectra [33].
5. Validation: Compare and integrate identifications from steps 3 and 4. Use a decoy library approach to estimate the FDR for the final metabolite list [45].
Table 1: Impact of Deconvolution Strategies on Identification Accuracy [33]
| Deconvolution Method | Reported False Positive Rate | Key Advantage | Primary Use Case |
|---|---|---|---|
| AMDIS (Default Settings) | Can be 70-80% | Fast, automated, widely available | Initial screening of simple mixtures |
| AMDIS (Optimized Parameters + CDF Filter) | Significantly reduced | Balances speed with improved specificity | Routine profiling of complex extracts |
| RAMSY Algorithm | Low (extracts pure spectra) | Superior for resolving co-eluted peaks | Targeted analysis of problematic chromatographic regions |
| PARAFAC2 (e.g., PARADISe) | Mathematically constrained | Provides unique, mathematically rigorous solution | High-value samples requiring maximum reliability [21] |
Table 2: Troubleshooting Matrix for Common Issues
| Symptom | Likely Cause | Immediate Action | Long-term Solution |
|---|---|---|---|
| High false positives | Poor S/N, library overfitting, co-elution | Apply stricter MF/RI filters; review chromatogram | Optimize AMDIS params; use RAMSY; apply decoy FDR [33] [45] |
| Missed peaks (false negatives) | Threshold too high, signal suppression | Lower detection threshold; check recovery of IS | Optimize sample cleanup; use adaptive thresholding [46] [44] |
| Unreliable quantification | Matrix effects, incomplete derivatization | Use matrix-matched IS for correction | Optimize derivatization protocol; use isotope-labeled IS [46] [33] |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents for GC-MS Metabolomics Sample Preparation
| Item | Function/Description | Critical Note |
|---|---|---|
| Methoxyamine hydrochloride | Protects carbonyl groups (aldehydes/ketones) via methoximation, preventing cyclic forms of sugars. | Must be prepared fresh in dry pyridine for consistent reaction [33]. |
| N-Methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA) | A silylation agent that replaces active hydrogens (-OH, -COOH, -NH) with a trimethylsilyl group, increasing volatility. | Using MSTFA with 1% TMCS (catalyst) improves derivatization efficiency [33]. |
| Retention Index Standard (FAME mix) | A homologous series of fatty acid methyl esters eluting across the chromatographic run. | Allows calculation of Kovats Retention Indices, essential for compound identification independent of small retention time shifts [33]. |
| Deuterated Internal Standards | Isotopically labeled analogs of target compounds (e.g., d27-myristic acid). | Corrects for analyte losses during preparation and matrix effects during ionization; crucial for accurate quantification [46] [33]. |
| Pyridine (Silylation Grade) | Anhydrous solvent for derivatization reactions. | Must be kept anhydrous; water will quench the silylation reaction and degrade the derivatizing agent. |
Visual Guides to Workflows and Decision Processes
AMDIS Optimization and Decision Workflow
Integrated Experimental and Computational Workflow
Technical Support Center: Troubleshooting AMDIS Deconvolution & False Positives
This guide provides targeted solutions for researchers facing challenges in setting Match Factor (MF) thresholds and reducing false positives during compound identification with Automated Mass Spectral Deconvolution and Identification System (AMDIS) in GC-MS analysis.
Frequently Asked Questions (FAQs)
1. Q: A high percentage of my AMDIS identifications are false positives. What is the primary cause and initial corrective action? A: The indiscriminate use of AMDIS's default parameters is a major cause, with studies reporting false-positive rates of 70–80% [10] [33]. The most effective initial action is to optimize and raise your Match Factor threshold. Instead of using a single universal value, implement tiered thresholds (e.g., 70 for indication, 80 for tentative identification, 90 for confident identification) and always require orthogonal confirmation from retention indices (RI) [47] [48].
2. Q: How do I optimally set the Match Factor threshold to balance sensitivity and reliability? A: There is no single optimal value; it depends on your library and analysis goals. For a broad commercial library like NIST, start with a high threshold (e.g., ≥85). For a curated, application-specific custom library, a lower threshold (e.g., ≥70) can be used safely [2]. The key is to determine the threshold experimentally using a validation set of known compounds relevant to your sample matrix and to use Retention Index (RI) filtering to discard high-MF matches with poor RI agreement [47] [49].
3. Q: Even with a high Match Factor, I get incorrect identifications from co-eluting compounds. How can I resolve this? A: Co-elution is a fundamental challenge that AMDIS alone cannot always resolve [10]. Implement a complementary deconvolution strategy:
- Use the Reverse Search function in NIST MS Search or similar tools to penalize spectra with extra peaks not in the library spectrum [47].
- Apply a secondary, statistical deconvolution tool like Ratio Analysis of Mass Spectrometry (RAMSY) on problematic peaks. RAMSY can recover low-intensity ions from overlapped peaks that AMDIS may miss, improving spectral purity for matching [10] [33].
4. Q: My sample contains novel or library-absent compounds. How can I avoid false positives and still gain useful information? A: For non-targeted analysis of unknowns, reduce reliance on the MF alone.
- Use the Hybrid Similarity Search (available in NIST software) to identify compounds related to, but not exactly matching, library entries [47].
- Employ a multi-criteria intelligent workflow. A proposed structure should satisfy several checks: a reasonable MF, RI prediction within tolerance (e.g., ≤70 units on a non-polar phase), a matching molecular ion from soft ionization data, and a plausible molecular formula from high-resolution MS [49]. This cross-validation drastically reduces false assignments.
5. Q: Can I create a custom library to improve identification accuracy for my specific field? A: Yes, and it is highly recommended. A targeted, custom library is one of the most effective ways to reduce false positives and processing time.
- As demonstrated in strawberry VOC research, a custom library containing 104 target compounds reduced false hits by 200, decreased report file size by over 96%, and cut AMDIS processing time by 71% (from 31s to 9s) compared to a commercial library [2].
- The library should include clean spectra, validated Retention Indices, and chemical information for your compounds of interest [2].
Table 1: Recommended Match Factor Thresholds Based on Identification Confidence & Library Type
| Confidence Level | Recommended MF Threshold | Required Orthogonal Evidence (e.g., RI) | Typical Use Case & Library Type |
|---|---|---|---|
| Indication | 70 - 79 | Not required for initial screening | Preliminary screening with a custom, targeted library [2]. |
| Tentative Identification | 80 - 89 | Mandatory. RI deviation ≤ 20-30 units [47]. | Routine non-targeted analysis with commercial libraries (NIST, Wiley). |
| Confident Identification | ≥ 90 | Mandatory. RI deviation ≤ 10-15 units [47] [48]. | Reporting identified compounds. Requires validation with standards where possible. |
Table 2: Performance Metrics of Different Deconvolution & Library Strategies
| Strategy | Reported False Positive Rate | Key Performance Benefit | Reference / Application |
|---|---|---|---|
| AMDIS with default settings | 70-80% [10] [33] | Fast, automated deconvolution. | Baseline for comparison. |
| AMDIS + Optimized MF & RI Filtering | Significantly reduced (error rates <10% achievable) [48] | Balances reliability and comprehensiveness. | General non-targeted profiling [48]. |
| AMDIS + Custom Target Library | Reduction of ~200 false hits in a case study [2] | Dramatically speeds up processing, increases target accuracy. | Targeted metabolomics (e.g., strawberry VOCs) [2]. |
| AMDIS + RAMSY Deconvolution | Lower than AMDIS alone [10] [33] | Better recovery of pure spectra from severe co-elution. | Complex plant metabolite extracts [10] [33]. |
Table 3: Retention Index (RI) Tolerance Thresholds for False Positive Rejection
| Chromatographic Phase | Recommended RI Tolerance (ΔRI) | Context & Notes |
|---|---|---|
| Standard Non-Polar | ≤ 10 units (stringent) ≤ 20 units (routine) [47] | For high-confidence matching. Median absolute deviation for high-score (>750) IDs was 9 units [47]. |
| Polar | ≤ 100 units [49] | Prediction and measurement are less accurate on polar phases. Used as a secondary filter [49]. |
| AI-Predicted RI (AIRI) | ≤ 15-30 units [47] | For compounds without experimental RI. Mean error of AIRI is ~15 units [47]. |
Detailed Experimental Protocols
Protocol 1: Integrated RAMSY-AMDIS Workflow for Complex Plant Extracts [10] [33] This protocol uses statistical deconvolution (RAMSY) to complement AMDIS for difficult co-elutions.
Sample Derivatization:
- Perform methoximation by adding 10 μL of O-methylhydroxylamine hydrochloride in pyridine (40 mg/mL) to the dry extract. Incubate at 30°C for 90 min.
- Follow with silylation by adding 90 μL of MSTFA with 1% TMCS. Incubate at 37°C for 30 min.
- Add a retention index marker (e.g., FAME mixture C8-C30) before GC-MS analysis.
GC-TOF-MS Analysis:
- Use a non-polar or semi-standard non-polar capillary column (e.g., DB-5MS).
- Operate the mass spectrometer in electron ionization (EI) mode at 70 eV.
- Acquire data in full-scan mode over an appropriate mass range (e.g., m/z 50-600).
AMDIS Processing (Initial):
- Use a factorial design to optimize AMDIS deconvolution parameters (Component Width, Adjacent Peak Subtraction, Resolution, Sensitivity) for your specific instrument and sample type.
- Process data with a target library. Apply a Compound Detection Factor (CDF)—a heuristic filter based on match factor, peak shape, and signal-to-noise—to reduce initial false positives.
RAMSY Processing (Targeted):
- Export the unresolved or poorly deconvoluted peak regions from AMDIS.
- Process these regions with the RAMSY algorithm. RAMSY analyzes intensity ratios across multiple spectra to resolve co-eluted ions by identifying distinct chromatographic profiles [10] [33].
- Extract the purified mass spectra for each component resolved by RAMSY.
Identification & Validation:
- Search the RAMSY-purified spectra against your mass spectral library.
- Compare the identifications and Match Factors with the initial AMDIS results.
- Confirm all identifications by matching the experimental Retention Index against a reliable RI database within a strict tolerance (e.g., ±20 units).
Protocol 2: Creation and Use of a Custom AMDIS Target Library [2] This protocol outlines steps to build a project-specific library, significantly reducing false positives.
Data Collection for Library Entries:
- Analyze authentic chemical standards under your standard GC-MS conditions.
- For compounds without standards, curate high-quality spectra from reliable sources (e.g., peer-reviewed literature, dedicated metabolite databases) that match your analytical conditions.
Spectrum and Metadata Entry:
- Use the AMDIS "New Library" function or a compatible library manager.
- For each entry, input: a) The pure, clean mass spectrum. b) The experimentally determined or reliably sourced Kovàts Retention Index for your specific column type. c) Additional metadata: Chemical name, CAS number, molecular formula, chemical class, and odor descriptors if applicable [2].
Library Formatting and Application:
- Save the library in the AMDIS user library format (.LBR).
- In AMDIS, set your custom library as the primary target library for the "Identify" step.
- You can use a lower Match Factor threshold (e.g., 70) with higher confidence because the search space is restricted to relevant compounds [2].
- The software will only report matches from this focused list, eliminating irrelevant false hits from large commercial libraries.
Visual Workflow and Relationship Diagrams
Decision Workflow for Intelligent Match Factor Threshold Application
Integrated AMDIS and RAMSY Deconvolution Workflow Protocol
How Custom Target Libraries Reduce False Positives
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Software, Libraries, and Reagents for Reliable AMDIS Analysis
| Item Name / Category | Specific Product / Example | Primary Function in Reducing False Positives |
|---|---|---|
| Deconvolution & Identification Software | AMDIS (NIST) [10] [2] [33] | Core deconvolution algorithm. Must be parameter-optimized. |
| Complementary Deconvolution Tool | RAMSY (Ratio Analysis of MS) [10] [33] | Statistical resolution of severe co-elution not fully handled by AMDIS. |
| Mass Spectral Library (Commercial) | NIST EI Mass Spectral Library [47] [49] | Broad search space for non-targeted analysis. Requires high MF thresholds and RI filtering. |
| Mass Spectral Library (Custom) | User-created AMDIS Library (.LBR) [2] | Restricts search to relevant compounds, allowing lower thresholds and minimizing false hits. |
| Retention Index Database | NIST Retention Index Library [47] | Provides experimental and AI-predicted RI values for RI-based match filtering. |
| Derivatization Reagent | MSTFA + 1% TMCS [10] [33] | Silylates polar metabolites for GC-MS analysis, impacting RI and spectrum. Essential for reproducibility. |
| Retention Index Standards | n-Alkane Series (C8-C40) or FAME Mix (C8-C30) [10] [48] [33] | Allows calculation of experimental Kovàts Retention Index for compound identification. |
| Method Validation Reference | Certified Reference Materials (CRMs) | Enables empirical determination of optimal MF/RI thresholds for your specific method and matrix. |
This technical support center is designed for researchers and scientists working with GC-MS data, particularly within metabolomics and drug development. A core challenge in these fields is the reliable identification of compounds when chromatographic peaks overlap, a phenomenon known as co-elution. Automated deconvolution software, such as the widely used Automated Mass Spectral Deconvolution and Identification System (AMDIS), is essential for processing complex datasets but is not infallible. Studies indicate that indiscriminate use of automated tools can generate false positive identification rates as high as 70-80% [33].
The content here is framed within the critical thesis of reducing false positives in GC-MS AMDIS deconvolution research. The guides and protocols provided are built on the principle that strategic manual review is not a failure of automation but a necessary, expert-led step to validate results, ensure data integrity, and produce publication-quality findings. This center provides clear, actionable guidance on when to intervene and how to do so effectively.
Troubleshooting Guides & FAQs
Q1: How do I know when to initiate a manual review of my GC-MS deconvolution results instead of relying solely on AMDIS? You should initiate a manual review when automated flags or data quality indicators suggest unreliable results. Key triggers include:
- Software Warnings: The deconvolution report indicates a high "Match Factor" but with a low "Purity" score, or flags a peak as "unresolved."
- Spectral Anomalies: The extracted spectrum appears noisy, shows unexpected ion ratios, or lacks a clear molecular ion or characteristic fragment series.
- Chromatographic Indicators: Visual inspection of the chromatogram reveals obvious shoulder peaks, asymmetrical peak shapes, or a discrepancy between the peak apex and the reported retention index.
- Contextual Discrepancies: A compound is identified that is biologically implausible for the sample type or its concentration seems inconsistent with internal standards.
- Post-Processing Flags: Results from complementary software (like a Purity Angle test in Empower PDA data) exceed the calculated threshold, suggesting co-elution [50].
Q2: What is a step-by-step protocol for manually reviewing and validating a suspected co-eluting peak? Follow this detailed protocol to investigate a peak flagged for potential co-elution:
- Chromatographic Inspection: Visually examine the total ion chromatogram (TIC) and extracted ion chromatograms (EICs) for key ions of the target compound. Look for peak asymmetry, shoulders, or a widening baseline.
- Spectral Fidelity Check: Extract the mass spectrum at the peak apex, leading edge, and trailing edge. Compare these spectra for consistency. A pure compound will have nearly identical spectra across the peak [50].
- Library Match Interrogation: Critically assess the AMDIS library match. Do not rely solely on the overall match factor. Check if the major fragment ions (especially base peaks) align perfectly. Significant mismatches in key ions indicate impurity or misidentification.
- Deconvolution Parameter Adjustment: Re-process the peak region in AMDIS with adjusted parameters. Manually increase the "Component Width" and "Resolution" settings, as this parameter is critical for accurate deconvolution of overlapping signals [15]. Observe if a clean spectrum for the target compound emerges.
- Apply Complementary Algorithms: For persistently challenging peaks, export the raw data and process it with an orthogonal deconvolution algorithm, such as Ratio Analysis of Mass Spectrometry (RAMSY). Using RAMSY as a "digital filter" on AMDIS results has been shown to recover low-intensity ions and improve identification confidence in complex plant extracts [33].
- Final Validation: If a co-eluent is suspected, attempt to find a unique fragment ion for the interfering compound. Use this ion to reconstruct its chromatographic profile and confirm the overlap.
Q3: What experimental and data processing strategies can I implement upfront to minimize false positives from co-elution? Proactive method optimization is the best defense. Implement these strategies:
- Chromatographic Optimization: Prioritize improving physical separation. Lengthen the GC gradient, use a slower temperature ramp, or consider a different column phase to increase resolution before analysis.
- Systematic Deconvolution Optimization: Do not use AMDIS default settings blindly. For each new sample matrix or method, perform a factorial design of experiments to optimize AMDIS deconvolution parameters (like component width, sensitivity, and resolution) using a well-characterized standard mix [33].
- Use Orthogonal Identification Criteria: Require dual confirmation for compound identity. A high-quality identification should match both the mass spectrum (e.g., NIST library) and the retention index (RI) within a defined window (e.g., ±10 RI units) against a trusted standard library [15] [33].
- Implement a Heuristic Filter: Develop and apply a Compound Detection Factor (CDF). This can be a simple scoring system that weights the match factor, RI match, and peak shape purity. Discard identifications that fall below a strict, pre-defined threshold [33].
Key Data on Deconvolution Software Performance
Understanding the limitations of automated tools is crucial for deciding when manual review is essential. The following table summarizes findings from a comparative study of deconvolution software [15].
Table: Comparative Performance of GC-MS Deconvolution Software
| Software | Key Strength | Major Limitation | False Positive Rate (Context) | Optimal Use Case |
|---|---|---|---|---|
| AMDIS (NIST) | Freely available; good spectrum deconvolution; uses RI libraries. | Highly sensitive to parameter settings; can miss subtle co-elution. | High (Produces a large number of false positives) [15]. | Initial screening of known compounds; requires rigorous parameter optimization and manual review. |
| ChromaTOF (LECO) | Tight instrument integration; automated peak finding. | Algorithm can be overly aggressive in declaring pure components. | High (Produces a large number of false positives) [15]. | High-throughput environments where results are routinely validated with standards. |
| AnalyzerPro (SpectralWorks) | Robust deconvolution for complex overlaps. | Can be overly conservative in declaring components. | Lower but may produce false negatives [15]. | Critical applications where confidence in reported identifications is paramount, accepting that some minor components may be missed. |
Table: Impact of a Combined Deconvolution Strategy (AMDIS + RAMSY)
| Metric | AMDIS Alone | AMDIS + RAMSY | Improvement & Explanation |
|---|---|---|---|
| False Positive Rate | High (70-80% reported in some studies) [33]. | Significantly Reduced | RAMSY acts as a statistical filter, removing spurious matches by analyzing ion intensity ratios across the peak [33]. |
| Ability to Deconvolve Severe Overlap | Limited, often low Match Factors for co-eluted peaks. | Enhanced | RAMSY recovers low-intensity, co-eluted ions that AMDIS may assign to noise, leading to cleaner extracted spectra [33]. |
| Metabolite Identification in Plant Extracts | May miss metabolites due to overlap. | More Comprehensive | The combined approach provides improved spectral deconvolution, leading to more reliable identifications in complex biological samples [33]. |
Detailed Experimental Protocols
Protocol 1: Optimizing AMDIS Deconvolution Parameters via Factorial Design This protocol is adapted from methods used to improve metabolite identification in plant extracts [33].
- Prepare a Standard Test Mixture: Create a solution containing 10-15 known metabolites covering a range of chemical classes and concentrations relevant to your study.
- Define Key Parameters: Select the AMDIS parameters to optimize (e.g., Component Width, Resolution, Sensitivity).
- Design the Experiment: Use a fractional factorial design (e.g., a 2^3 design) to test high and low values for each parameter. This minimizes the number of required runs.
- Process Data: Run the same standard mixture data file through AMDIS multiple times, each time with a different parameter set as per the experimental design.
- Evaluate Output: For each run, record the number of true positives (correctly identified standards), false positives, and false negatives.
- Determine Optimal Settings: Identify the parameter set that maximizes true positives while minimizing false positives. Use these settings for your subsequent sample analyses.
Protocol 2: Manual Peak Purity Assessment Using Photodiode Array (PDA) Data This protocol, based on Waters Empower software guidelines, provides orthogonal evidence of co-elution [50].
- System Suitability: Perform six replicate injections of a pure analytical standard. Ensure the peak's Maximum Spectral Absorbance (MSA) is less than 1.0 AU for optimal sensitivity.
- Establish Baseline Purity: Process the data with the "AutoThreshold" function enabled. For the pure standard peak, confirm that the calculated Purity Angle is consistently less than the Purity Threshold.
- Analyze Unknowns: Process your sample chromatograms using the validated method.
- Interpret Results: For any peak in your sample, a Purity Angle greater than the Purity Threshold is evidence of spectral impurity, indicating a co-eluting compound. This peak must be investigated further, even if the MS deconvolution appears successful [50].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table: Key Reagents for GC-MS Metabolomics and Deconvolution Validation
| Item | Function in Experiment | Critical Application in Co-elution Review |
|---|---|---|
| Fatty Acid Methyl Ester (FAME) Mixture (C8-C30) | Provides retention index (RI) markers for precise, system-specific calibration of retention times. | Enables use of retention index matching as a mandatory second filter for compound ID, catching false positives where MS matches but RI is wrong [33]. |
| O-Methylhydroxylamine Hydrochloride | Derivatizing agent for methoximation; protects carbonyl groups (ketones, aldehydes) in metabolites. | Standardizes analyte chemistry, improving chromatographic behavior and spectral reproducibility, which aids deconvolution [33]. |
| N-Methyl-N-trimethylsilyltrifluoroacetamide (MSTFA) with 1% TMCS | Derivatizing agent for trimethylsilylation; adds TMS groups to acidic protons (e.g., -OH, -COOH). | Increases volatility and thermal stability of metabolites for GC-MS. Consistent derivatization is key for reliable library spectrum matching [33]. |
| Chemically Pure Analytical Standards | Unambiguous reference materials for target compounds. | Essential for: 1) Creating in-house RI/MS libraries; 2) Optimizing deconvolution parameters (Protocol 1); 3) Validating purity thresholds (Protocol 2) [15] [50]. |
| Retention Index / Mass Spectral Library | Curated database pairing known mass spectra with validated retention indices. | The cornerstone of reliable identification. Using RI as a second constraint dramatically reduces false positives from MS similarity alone [15] [33]. |
Workflow & Decision Diagrams
Diagram Title: Decision Workflow for Initiating Manual Review of GC-MS Deconvolution Results
Diagram Title: Proactive Workflow to Minimize False Positives Before Manual Review
In gas chromatography-mass spectrometry (GC-MS) based metabolomics and natural product research, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a cornerstone tool for extracting pure component spectra from complex chromatographic data [10]. However, a critical challenge persists: the indiscriminate use of its parameters can generate false positive identification rates as high as 70–80% [10]. This high rate of erroneous assignments consumes valuable research resources, delays discovery, and compromises the integrity of downstream analyses.
This article is framed within a broader thesis aimed at systematically reducing false positives in GC-MS AMDIS deconvolution research. We posit that the integration of a data-driven, heuristic Compound Detection Factor (CDF) provides a robust solution to this problem. The CDF acts as a statistical filter, applied post-deconvolution to separate high-confidence identifications from spurious matches based on a holistic assessment of match quality, peak purity, and retention index fidelity [10]. This technical support center provides researchers and drug development professionals with the practical protocols, troubleshooting guidance, and foundational knowledge required to implement and benefit from this workflow enhancement.
Core Methodology: The CDF Heuristic Filter
The Compound Detection Factor (CDF) is a heuristic, multi-parameter score designed to evaluate the reliability of a compound identification made by AMDIS. It moves beyond a simple spectral match factor by incorporating orthogonal data points that collectively indicate a true positive.
Calculation of the CDF
The CDF is typically a weighted or logical function of several criteria:
- Spectral Match Factor (MF): The traditional AMDIS match score against a reference library (e.g., NIST).
- Reverse Match Factor (RMF): Confirms the purity of the deconvoluted spectrum.
- Retention Index (RI) Deviation: The absolute difference between the experimental Linear Retention Index (LRI) of the analyte and the library RI for the proposed compound. A larger deviation suggests a misidentification.
- Peak Shape and Purity Metrics: Parameters assessing the quality of the deconvolution, such as the peak width symmetry and the resolution from co-eluting compounds.
A simplified CDF logic can be represented as a high-confidence identification if:
(MF > Threshold_A) AND (RMF > Threshold_B) AND ( |ΔRI| < Threshold_C )
Experimental Validation Protocol
The development and validation of the CDF heuristic were demonstrated in a study on plant metabolomics [10]. The core experimental protocol is as follows:
Sample Preparation:
- Plant material (leaves/stems) is dried and ground.
- Extraction is performed using pressurized liquid extraction (e.g., Dionex ASE) with ethanol at 60°C and 1500 psi for 15 minutes.
- Extracts are dried under vacuum.
- Derivatization: The dried extract undergoes methoximation (with O-methylhydroxylamine hydrochloride in pyridine) followed by silylation (with MSTFA + 1% TMCS) to render metabolites volatile for GC-MS analysis [10].
GC-MS Analysis:
- System: Standard GC-MS system with electron ionization (EI) at 70 eV.
- Column: A non-polar or low-polarity capillary column (e.g., DB-5MS).
- Temperature Program: A gradient suitable for metabolomics (e.g., 60°C to 330°C).
- Internal Standards: Fatty acid methyl ester (FAME) mixtures are used to calculate experimental Linear Retention Indices (LRI) for each analyte [10].
Data Processing with AMDIS & CDF Application:
- Raw data files are processed in AMDIS.
- A factorial design of experiments is used to optimize AMDIS deconvolution parameters (component width, resolution, sensitivity) for each sample type.
- The optimized AMDIS output, containing compound identifications with MF, RMF, and peak metrics, is exported.
- Experimental LRIs are calculated for all detected peaks.
- The CDF algorithm is applied: each proposed identification is checked against the library LRI. Identifications failing the CDF criteria (e.g., |ΔRI| > 20 units) are flagged as low-confidence or rejected.
- For peaks with poor deconvolution (low MF), complementary algorithms like Ratio Analysis of Mass Spectrometry (RAMSY) can be applied to recover spectra of co-eluting components [10].
Key Quantitative Results
The application of a heuristic CDF filter has demonstrated significant improvements in identification accuracy.
Table 1: Impact of a Heuristic Filter (CDF) on AMDIS Deconvolution Performance
| Performance Metric | Standard AMDIS | AMDIS + CDF Filter | Improvement |
|---|---|---|---|
| Reported False Positive Rate [10] | 70-80% | Not explicitly quantified, but described as a "decrease" | Significant reduction |
| Key Filtering Criteria | Spectral Match (MF) only | MF + Reverse Match + Retention Index (RI) Deviation | Adds orthogonal verification |
| Data Fidelity | High risk of misidentification due to co-elution & spectral similarity | High-confidence identifications with consistent RI | Enhanced reliability for downstream analysis |
Table 2: Parameter Optimization for False Positive Reduction (General Principles from MS Analysis)
| Parameter | Overly Permissive Setting | Effect (More False Positives) | Recommended Optimization Action |
|---|---|---|---|
| Retention Time (RT) Window [51] | Wide (e.g., -1 to +1 min) | Matches ions with incorrect RT shift are accepted | Narrow window based on observed systematic shift (e.g., 0 to +1 min) [51] |
| Signal Intensity Threshold [51] | Too low (e.g., 50) | Noise is interpreted as signal | Increase threshold to ignore low-abundance noise [51] |
| Isotopic Peak Requirement (N value) [51] | Too few (e.g., 3 peaks) | Incomplete ion envelopes are accepted | Require more isotopic peaks (e.g., 4 or 5) for valid ion identification [51] |
Technical Support Center: Troubleshooting and FAQs
Troubleshooting Guide
Table 3: Common AMDIS/CDF Workflow Issues and Solutions
| Problem | Potential Causes | Diagnostic Steps | Recommended Solutions |
|---|---|---|---|
| High False Positive Rate after CDF | 1. Incorrect LRI calculation.2. Library RIs are inaccurate or from a different method.3. CDF thresholds are too lenient. | 1. Verify FAME standard peaks and LRI calculation formula.2. Check source of library RI values (experimental vs. predicted).3. Manually inspect a subset of flagged "positives." | 1. Re-process FAME standard data.2. Use a validated, method-matched RI library or generate in-house library.3. Tighten CDF thresholds (e.g., reduce max ΔRI). |
| Low Number of Identifications | 1. CDF thresholds are too strict.2. AMDIS deconvolution is suboptimal.3. Spectral match threshold is too high. | 1. Check how many IDs are rejected at each CDF criterion.2. Inspect raw chromatogram for visible peaks not reported.3. Lower MF threshold and rely more heavily on RI filter. | 1. Loosen CDF thresholds iteratively and validate.2. Re-optimize AMDIS deconvolution parameters (width, sensitivity).3. Implement a tiered confidence system (e.g., "Tentative" vs. "Confirmed"). |
| Poor Retention Time Reproducibility | 1. GC column degradation.2. Inconsistent oven temperature.3. Variable derivatization efficiency. | 1. Monitor RT shift of internal standards over time.2. Check GC system calibrations.3. Check derivatization protocol consistency. | 1. Perform column maintenance or replacement.2. Service GC instrument.3. Standardize derivatization time, temperature, and reagent freshness. |
| AMDIS Fails to Deconvolute Overlapping Peaks | 1. Severe co-elution.2. Very different component concentrations.3. AMDIS parameters set incorrectly. | 1. Visually inspect the total ion chromatogram for shoulder peaks.2. Examine extracted ion chromatograms for specific masses. | 1. Modify GC method to improve chromatographic resolution.2. Use a complementary deconvolution tool like RAMSY for problematic regions [10]. |
Frequently Asked Questions (FAQs)
Q1: What exactly is a "heuristic factor" in this context, and why is it better than a fixed rule? A: A heuristic factor is a practical, data-driven rule of thumb that approximates a solution where a perfect algorithmic model is impractical [52]. In GC-MS, fixed rules like "accept all matches with MF > 80" fail because they ignore co-elution and RI consistency. The CDF is a heuristic because it intelligently combines multiple pieces of evidence (spectral match, peak purity, retention data) to make a better judgment on identification confidence, mimicking the decision-making process of an expert analyst [10] [52].
Q2: Can I use the CDF approach with any GC-MS data, or does it require specific experimental setup? A: The core principle is universal, but its success depends on method-dependent parameters. The most critical requirement is the use of Retention Indexing. You must analyze a series of alkane or FAME standards under the exact same GC conditions as your samples to calculate experimental LRIs. Without this, you cannot implement the crucial RI deviation check within the CDF [10].
Q3: How do I balance reducing false positives with avoiding false negatives? A: This is a fundamental trade-off [43]. Excessively strict CDF criteria (e.g., ΔRI < 5) will eliminate false positives but may also discard correct identifications of compounds with variable RI. The best practice is to validate and calibrate your thresholds using a set of known standards analyzed within your matrix. Start with literature-based thresholds (e.g., ΔRI < 20), then adjust based on your validation data. For critical applications, a tiered identification system (e.g., Level 1: Matched RI & Spectrum, Level 2: Matched Spectrum only) is recommended.
Q4: Are there software tools that automatically apply such heuristic filtering? A: While AMDIS itself does not have a built-in CDF function, the concept is integrated into some advanced metabolomics platforms and workflows. Furthermore, the next generation of mass spectral data tools is moving towards greater use of such heuristic and data-mining approaches [52]. Implementing a CDF filter typically requires scripting (e.g., in Python or R) to process the report file from AMDIS, calculate LRIs, and apply the filtering logic. Some academic workflows, like the FLARE pipeline for RNA-editing data, exemplify this automated statistical filtering approach [53].
Q5: My lab focuses on drug development screening. How relevant is this to High-Throughput Screening (HTS) assays? A: Extremely relevant. False positives are a major cost and time burden in HTS, including mass spectrometry-based screens [54]. While the specific CDF for GC-MS may not apply, the philosophy of using orthogonal, heuristic checks is directly transferable. For example, in an MRM-based screen, a heuristic could combine signal intensity, signal-to-noise ratio, and chromatographic peak shape to automatically flag potential false-positive hits for secondary review before committing resources to follow-up [54] [43].
Visual Workflow and Toolkit
Workflow Diagrams
Diagram 1: CDF-Enhanced AMDIS Deconvolution Workflow
Diagram 2: Decision Logic for the CDF Heuristic Filter
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Key Reagents and Materials for GC-MS Metabolomics & CDF Validation
| Item | Function in the Workflow | Critical Notes for Reproducibility |
|---|---|---|
| FAME Standard Mixture (e.g., C8-C30) | Serves as the external or internal standard series for calculating experimental Linear Retention Indices (LRI). Essential for the RI check in the CDF [10]. | Use the same mixture and vendor for consistency. Prepare fresh dilutions regularly to avoid degradation. |
| Derivatization Reagents:• O-methylhydroxylamine HCl (MOX)• N-Methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA) with 1% TMCS• Pyridine (silylation grade) | Methoximation: Protects carbonyl groups (ketones, aldehydes) and reduces tautomerization.Silylation: Replaces active hydrogens (-OH, -COOH, -NH) with trimethylsilyl groups, increasing volatility and thermal stability for GC analysis [10]. | Reagents must be anhydrous. Use under inert atmosphere if possible. MSTFA+TMCS is hygroscopic; store properly and check performance regularly. |
| Alkane Standard Mixture (e.g., C7-C40) | An alternative to FAMEs for LRI calculation. Alkanes are inert and provide a universal retention scale. | Choose an alkane series that brackets your analyte elution range. |
| NIST Mass Spectral Library & RI Add-on | The primary reference for spectral matching (MF, RMF). The RI add-on library provides crucial reference retention index data for the CDF filter [52]. | Ensure the RI library was generated on a similar column (e.g., DB-5 equivalent) and using a comparable temperature program. |
| Quality Control (QC) Metabolite Standard Mix | A mixture of known metabolites covering various compound classes. Used to validate the entire workflow, including derivatization efficiency, instrument sensitivity, LRI calculation accuracy, and CDF filter performance. | Run the QC sample repeatedly at the start, throughout, and at the end of a batch to monitor system stability. |
Ensuring Accuracy: Validation Protocols and Complementary Chemometric Tools
In gas chromatography-mass spectrometry (GC-MS) analysis, particularly in non-targeted screening for forensic toxicology, metabolomics, and drug development, the Automated Mass Spectral Deconvolution and Identification System (AMDIS) is a widely employed tool [55] [56]. Its primary function is to separate overlapping peaks and identify compounds within complex biological matrices such as serum, urine, or tissue samples. However, a significant and well-documented challenge is its tendency to generate false positive identifications [56]. These inaccuracies can compromise research integrity, lead to erroneous biomarker discovery, and misdirect downstream experiments in drug development pipelines.
The core thesis of this technical support center is that robust, systematic validation experiments are not optional but essential for reducing false positives and ensuring reliable results. The most effective strategy for this validation involves the use of spiked standards within complex mixture backgrounds [57]. This approach creates a "ground truth" model system where the identities and quantities of target analytes are known, allowing researchers to objectively benchmark AMDIS parameters, tune deconvolution algorithms, and quantitatively assess the performance of their entire analytical workflow in terms of sensitivity, specificity, and false discovery rate [57].
Troubleshooting Guides & FAQs
This section addresses common, specific challenges users encounter when performing validation experiments and general GC-MS analysis with AMDIS.
Deconvolution & Identification Issues
Q1: AMDIS is reporting a high number of false positive identifications in my spiked validation sample. What parameters should I adjust first?
- Answer: A high false positive rate often indicates that the matching criteria are too lenient. Focus on adjusting the following AMDIS parameters in sequence:
- Match Factor/Reverse Match: Increase the threshold for both (e.g., to 80+). The Reverse Match is particularly critical as it measures how well the library spectrum matches your sample spectrum, penalizing for ions in the sample not present in the library.
- Sensitivity: Start with a "Medium" setting. "High" sensitivity can co-elute noise with signals, generating false peaks.
- Resolution: Ensure this is set appropriately for your instrument and column. Too high a setting can split true peaks, while too low can fail to separate co-eluting compounds.
- Shape Requirements: Increasing the shape requirement can filter out noisy, peak-like artifacts.
- Pro Tip: Use your spiked standard experiment. After each parameter change, reprocess the data and monitor the change in the ratio of correctly identified spikes (true positives) versus incorrect background identifications (false positives). The goal is to maximize true positives while driving false positives to near zero [55].
Q2: How can I validate that AMDIS is correctly quantifying my analytes, not just identifying them?
- Answer: This requires a spiked experiment with a calibration curve. Spike your target analytes at a minimum of 5 different, known concentrations into the complex matrix (e.g., yeast lysate, serum blank) [57]. Process all samples through AMDIS.
- Plot the concentration reported by AMDIS (based on peak area or height) against the known spiked concentration. Assess the linearity (R²), accuracy (% bias), and precision (% RSD) of the response.
- This will reveal if matrix effects are causing ion suppression or enhancement and whether AMDIS's quantification model is appropriate for your sample type. It directly tests the reliability of the "quantitative" output of your workflow.
Sample Preparation & Experimental Design
Q3: What is the best background matrix to use for creating a spiked validation standard?
- Answer: The ideal background matrix is one that closely mimics your real study samples but is guaranteed not to contain your target analytes. For example:
- For serum or plasma studies: Use charcoal-stripped or analyte-free surrogate serum.
- For cellular or tissue metabolomics: Use a protein extract or lysate from a control system (e.g., yeast cell lysate as used in proteomic benchmarks [57]).
- For forensic toxicology: Use drug-free serum or urine [55]. The complexity of the background (its endogenous proteins, lipids, salts, and metabolites) is what tests the deconvolution algorithm's ability to find the "signal in the noise."
Q4: How many compounds and what concentration range should I spike for a comprehensive benchmark?
- Answer:
- Number: Use a mixture of at least 10-50 compounds, if possible. Libraries like the UPS1 proteomic standard (48 proteins) provide a good model [57]. For small molecules, create a custom mix covering a range of chemical properties (polarities, molecular weights).
- Concentration: Spike at multiple levels relative to the background. Include low concentrations (near the limit of detection), mid-range (therapeutic or physiological), and high concentrations. This tests the workflow's dynamic range and ensures sensitivity is assessed at biologically relevant levels [55] [57].
Data Analysis & Interpretation
Q5: After validation, my AMDIS results for real samples still show some unlikely compounds. How do I perform a final manual review?
- Answer: Even a well-validated system requires critical review. For every identification flagged by AMDIS, manually inspect:
- Chromatogram Peak Shape: Is it a smooth, Gaussian-shaped peak, or a sharp spike or jagged artifact?
- Mass Spectrum Quality: Does the background-subtracted spectrum have a high signal-to-noise ratio? Do the key fragment ions co-elute precisely at the same retention time?
- Retention Index Match: Compare the observed retention time (or index) against a known standard run on the same method. A mismatch >0.1 min is a red flag.
- Bibliographic/Contextual Plausibility: Is the compound known to be present in your biological system? Is it a known artifact (e.g., phthalates from plastics, column bleed)? Establish a standard operating procedure (SOP) for this review to ensure consistency.
Detailed Experimental Protocols
Based on published methodologies for benchmarking analytical workflows [55] [57], here is a generalized step-by-step protocol for executing a validation experiment.
Protocol: Benchmarking AMDIS Performance Using Spiked Standards
Objective: To quantitatively determine the false positive rate, true positive rate (sensitivity), and quantification accuracy of an AMDIS-based GC-MS workflow for a defined set of target analytes in a complex matrix.
Materials:
- Complex background matrix (e.g., drug-free serum, yeast cell lysate).
- Stock solutions of target analyte standards.
- Internal standard (IS, preferably deuterated or otherwise isotopically labeled analogs of targets).
- Sample preparation reagents (extraction solvents, derivatization agents, etc.).
- GC-MS system with established method.
Procedure:
Prepare Spiked Validation Set:
- Create a dilution series of your target analyte mix (e.g., 5-8 concentration levels).
- Spike each concentration level into separate aliquots of the complex background matrix. Also prepare a "blank" background sample with no spike and a "solvent standard" with analytes in neat solvent.
- Add a constant amount of Internal Standard (IS) to every sample (blank, spiked, and solvent) to monitor process variability.
Sample Processing:
- Process all samples (including blanks and solvent standards) through your entire routine workflow—extraction, derivatization (if needed), and GC-MS analysis—in randomized order to avoid batch effects.
Data Acquisition & Deconvolution:
- Acquire data in full-scan mode (e.g., 50-550 m/z) to allow for non-targeted deconvolution.
- Process all data files through AMDIS using a consistent parameter set. Use a target library containing your spiked analytes.
Data Analysis & Benchmarking:
- For each spiked sample, compile a list of compounds identified by AMDIS.
- Categorize each identification:
- True Positive (TP): A spiked analyte correctly identified.
- False Positive (FP): An identification that is not one of the spiked analytes (e.g., a background component misidentified or a spectral artifact).
- False Negative (FN): A spiked analyte that was present but not identified by AMDIS.
- Calculate key performance metrics:
- False Positive Rate (FPR) = FP / (FP + TN). (TN or True Negative is defined here as background components correctly not identified as a target analyte).
- Sensitivity/Recall = TP / (TP + FN).
- Precision = TP / (TP + FP).
- For quantification, plot reported abundance vs. spiked concentration for each true positive to calculate linearity and accuracy.
Table 1: Key Performance Metrics from Validation Experiments
| Metric | Formula | Target Benchmark (Example) | Interpretation |
|---|---|---|---|
| False Positive Rate (FPR) | FP / (FP + TN) | < 5% | Measures how often the system reports an analyte that is not present. Lower is better. |
| Sensitivity/Recall | TP / (TP + FN) | > 90% | Measures the system's ability to find all true analytes. Higher is better. |
| Precision | TP / (TP + FP) | > 95% | Measures the reliability of a positive identification. Higher is better. |
| Quantification Accuracy | (Measured Conc. / Spiked Conc.) * 100% | 85-115% | Measures the correctness of the reported amount. |
Note: Target benchmarks are illustrative and should be defined based on project requirements. Data derived from principles in [55] [57].
Visual Workflow: Validation Experiment Process
Flowchart: Validation and Optimization of GC-MS AMDIS Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for Spiked Validation Experiments
| Item | Function | Critical Considerations |
|---|---|---|
| Certified Pure Analytical Standards | Provides the known "signal" to be recovered. Forms the basis of the "ground truth." | Purity (>98%). Stability in solution. Cover a range of chemical properties relevant to your study. |
| Complex Background Matrix (e.g., Charcoal-stripped serum, yeast lysate) | Provides the realistic "noise" and matrix effects. Tests deconvolution specificity. | Must be confirmed as free of target analytes. Should mimic the physicochemical properties of real study samples. |
| Isotope-Labeled Internal Standards (e.g., Deuterated analogs) | Monitors and corrects for variability in sample preparation, injection, and ionization. | Should be added at the very beginning of sample prep. Ideally, one IS per analyte class. |
| Quality Control (QC) Reference Material | A separate, consistent sample run throughout the batch to monitor instrument stability over time. | Can be a pooled study sample or a commercially available reference material. |
| Comprehensive, Curated Mass Spectral Library | The reference database against which unknown spectra are matched. | Library entries should include reliable retention index data. Must be compatible with AMDIS (.msl, .msp). |
Information synthesized from experimental designs in [55] [57].
Technical Support Center
This technical support center is designed within the context of thesis research focused on reducing false positives in GC-MS AMDIS deconvolution. The following guides address common challenges researchers face when validating AMDIS outputs, providing clear protocols to enhance the reliability of compound identification in complex samples such as biological matrices or environmental extracts [58] [24].
Frequently Asked Questions (FAQs)
Q1: Why does my AMDIS analysis produce a high number of false positive identifications, and how can I mitigate this? A1: AMDIS is known to generate a high rate of false positives, which is a central challenge in automated deconvolution [24]. This occurs due to co-elution, background noise, and imperfect spectral matching. To mitigate this:
- Increase the Match Factor Threshold: Raise the minimum match factor (e.g., from 60 to 80 or 90) in the Identification (Identif.) settings tab to enforce stricter spectral matching [16].
- Leverage Retention Time/Index: Use a calibration file and retention index information to filter candidates based on elution time, adding a critical confirmatory parameter beyond the mass spectrum [16].
- Employ Secondary Validation: Always plan to validate AMDIS findings using manual interpretation of spectra/chromatograms or an independent software algorithm [24].
Q2: What is the difference between AMDIS's ".ELU" and ".FIN" output files, and which should I use for validation studies?
A2: The ".ELU" file contains the raw deconvolution data (spectra, peak areas), while the ".FIN" file contains the final identification results after matching against the target library [58]. For robust comparative validation studies, it is recommended to use the "raw" .ELU deconvolution data. This allows you to apply consistent, study-specific identification criteria across all samples and compare the underlying spectral quality independently of AMDIS's built-in library matching thresholds [58].
Q3: How can I improve the detection of low-abundance or co-eluting compounds that AMDIS misses? A3: For challenging peaks, adjust the deconvolution parameters:
- Adjust Component Width: Modify the "Component Width" setting to better match the chromatographic peak shape of your method.
- Analyze in Segments: AMDIS may identify compounds correctly only when a smaller portion of the chromatogram is analyzed. Manually select a specific retention time window around the area of interest and re-run the deconvolution [24].
- Use Sensitive/Threshold Modes: Experiment with the "Sensitive" and "Threshold" settings in the deconvolution options to adjust the software's responsiveness to small peaks and background noise.
Q4: My manual review of the spectrum disagrees with AMDIS's identification. Which should I trust? A4: Manual interpretation by a skilled analyst is still considered the gold standard. AMDIS is an automated tool and can be misled by poor deconvolution or library spectra of variable quality. Proceed as follows:
- In AMDIS, examine the extracted (white) vs. raw (black) spectrum overlay to assess deconvolution quality [16].
- Check the match factor and the "Fit," "RMFit," and "Purity" values in the results window for confidence metrics [16].
- Manually compare the deconvoluted spectrum against the library entry, paying attention to key discriminant ions. If manual review raises doubts, flag the identification as unconfirmed.
Troubleshooting Guides
Issue: Inconsistent quantification of the same metabolite across multiple sample runs.
- Cause: AMDIS does not always use a common reference ion mass fragment (IMF) to quantify the same metabolite across different samples, affecting reproducibility [24].
- Solution:
- Manual Ion Selection: After identification, note the primary quantification ion used by AMDIS for a target compound in a representative sample.
- Verify Consistency: Check the "Quant Ion" column in the results window across all samples. If it varies, this is the source of inconsistency.
- External Processing: For rigorous studies, export the raw ion abundances and perform consistent peak integration or quantification using a separate software package or script that forces the use of a single, specific quantification ion for each compound [24].
Issue: High false negative rate (AMDIS fails to identify a compound I know is present).
- Cause: The compound's signal may be below the deconvolution threshold, or its spectrum may be overly obscured by co-eluting compounds.
- Solution:
- Lower the Match Factor: Temporarily reduce the minimum match factor to see if the identification appears.
- Review Deconvolution: Zoom in on the specific retention time. Use the right-click "Show Component on Chromatogram" option to visualize what AMDIS extracted as a pure component [16]. If the deconvolution is poor, adjust the "Resolution" and "Shape" settings.
- Check the Library: Ensure the target compound and its expected retention time are correctly entered in your custom library [16].
Issue: Data files are cumbersome to process in batch, and results require extensive manual curation.
- Cause: AMDIS's output layout is not immediately amenable to high-throughput statistical analysis, and manual curation is time-consuming [24].
- Solution:
- Batch Processing: Use the AMDIS "Batch Job" functionality to process multiple files consecutively with the same settings.
- Automate Post-Processing: Develop or use existing scripts (e.g., in R or Python) to parse AMDIS report (.FIN or .ELU) files, align compounds across samples, and format data for input into statistical tools like MetaboAnalyst [24]. The
MetaBoxR package is an example built for this purpose [24].
Performance Comparison of Deconvolution Tools
The following table summarizes key performance metrics from comparative studies, highlighting the trade-offs between different software approaches.
Table 1: Comparative Performance of GC-MS Deconvolution Software
| Software / Method | False Positive Rate | False Negative Rate | Key Strength | Primary Limitation | Reference |
|---|---|---|---|---|---|
| AMDIS (Default) | 33.2% | 9.8% | Widely used, integrated with NIST library, good for targeted analysis. | High false positive rate; quantification inconsistency across samples. | [24] |
| Manual Interpretation | ~5-10% (estimated) | Variable (user-dependent) | Gold standard for verification; high specificity. | Extremely time-consuming; not scalable for large datasets. | [24] |
| MetaBox (PScore Algorithm) | 12.7% | 4.3% | Lower error rates; automated, high-throughput R package. | Requires familiarity with R; less known than AMDIS. | [24] |
| In-house Scripts / PyMS | Highly variable | Highly variable | Fully customizable to specific research needs. | Requires significant programming expertise to develop and validate. | [58] [59] |
Experimental Protocol for Validating AMDIS Results
This protocol provides a framework for systematically evaluating the accuracy of AMDIS deconvolutions, which is essential for thesis research aimed at improving data fidelity.
Objective: To quantify the false positive and false negative rates of AMDIS deconvolution and identification under specific experimental conditions by comparison against manual interpretation and a secondary software algorithm.
Materials & Samples:
- Standard Mixture: A calibrated mixture of 20-50 known volatile organic compounds (VOCs) or metabolites at varying concentrations (e.g., 0.1-100 ng/µL) [58] [24].
- Test Samples: Representative biological samples (e.g., faecal extracts, breath collections) spiked with the standard mixture [24].
- Software: AMDIS (v2.73 or later), R statistical environment with MetaBox package (or alternative like PyMS) [59] [24].
Procedure:
- Data Acquisition:
- Analyze the standard mixture and test samples by GC-MS using your standard metabolomics/profiling method.
- Ensure consistent injection order with randomized blanks and quality control (QC) samples.
AMDIS Processing (Primary Analysis):
- Create a target library in AMDIS containing the compounds in your standard mixture, with their known retention times and spectra [16].
- Process all data files through AMDIS using a standardized set of deconvolution parameters (Component Width: Medium, Resolution: High, Shape Requirements: Medium). Set the initial Match Factor to 60 [16].
- Export both the
.ELU(deconvoluted spectra) and.FIN(identification results) files for each sample [58].
Manual Interpretation (Validation Benchmark):
- For the standard mixture sample, manually review the Total Ion Chromatogram (TIC) and corresponding mass spectra at each peak.
- For each expected compound, note its presence/absence and verify the spectrum against the reference library. This manual curation set serves as your "ground truth" [24].
Secondary Software Processing:
- Process the same raw data files using the secondary validation tool (e.g., MetaBox in R).
- Use the same target library used in AMDIS. Apply the algorithm's scoring function (e.g., PScore) to identify and quantify compounds [24].
Comparative Analysis & Calculation of Metrics:
- Align the compound lists from AMDIS, Manual Interpretation, and the Secondary Software.
- For the Standard Mixture: Calculate:
- False Positive Rate (AMDIS):
(Number of compounds ID'd by AMDIS but *not* in the known standard) / (Total ID's by AMDIS). - False Negative Rate (AMDIS):
(Number of known standard compounds *not* ID'd by AMDIS) / (Total compounds in standard). - Perform the same calculation for the Secondary Software.
- False Positive Rate (AMDIS):
- For Test Samples: Compare the lists of potential biomarkers or differentially abundant compounds generated by each method. Assess the degree of overlap.
The Scientist's Toolkit
Essential reagents and materials for conducting validation experiments.
Table 2: Key Research Reagent Solutions for Validation Experiments
| Item | Function in Validation | Example / Specification |
|---|---|---|
| Certified Standard Mixture | Serves as a ground truth sample with known identities and concentrations to calculate exact false positive/negative rates. | 30+ component VOC mix (e.g., from Restek or Supelco) at defined concentrations. |
| Derivatization Reagents | For metabolomics, renders non-volatile metabolites volatile for GC-MS analysis (e.g., MSTFA for silylation). | N-Methyl-N-(trimethylsilyl)trifluoroacetamide (MSTFA). |
| Internal Standard Mix | Corrects for injection volume variability and instrument drift; crucial for validating quantification consistency. | Stable isotope-labeled compounds (e.g., ¹³C-Glucose, D₈-Naphthalene) not native to the sample. |
| Retention Index Calibration Mix | Allows conversion of retention times to system-independent Kovats indices, enabling cross-study/library comparison. | n-Alkane series (C₈-C₄₀) analyzed under the same GC conditions. |
| Custom Target Library File | The curated list of compounds against which AMDIS searches; its quality directly impacts identification accuracy. | .MSL or .LBR file containing reference spectra and validated retention times for your target compounds [16]. |
| Quality Control (QC) Sample | A pooled sample from all test samples; analyzed repeatedly to monitor system stability and performance over the batch. | Pooled aliquot of all biological test extracts. |
Workflow for Validating AMDIS Deconvolution
The following diagram outlines the logical workflow for a rigorous comparative validation of AMDIS results, integrating manual review and secondary software to reduce false discoveries.
In Gas Chromatography-Mass Spectrometry (GC-MS) analysis, deconvolution is the critical computational process that separates the overlapping signals of co-eluting compounds to reconstruct a pure mass spectrum for each chemical component [7]. This step is foundational for accurate metabolite identification and quantification, especially in complex mixtures typical of metabolomics, forensic toxicology, and drug development research [7] [60].
The Automated Mass Spectral Deconvolution and Identification System (AMDIS), developed by the National Institute of Standards and Technology (NIST), has been a widely used and freely available tool for this task [7]. However, a known and significant limitation of AMDIS is its tendency to produce false positives, particularly when analyzing samples containing structurally similar compounds or in experiments where a peak is detected in only a subset of chromatograms [23]. For instance, in forensic analysis, samples containing 3,4-methylenedioxymethamphetamine (MDMA) frequently trigger a false positive report for its analog 3,4-methylenedioxyamphetamine (MDA), because their mass spectral fragments are nearly identical at higher collision energies [19].
This persistent issue underscores the thesis that relying on a single deconvolution algorithm can compromise data integrity. A strategic, multi-tool approach is necessary to validate findings and reduce erroneous identifications. This technical support center outlines a framework that leverages complementary software tools and statistical workflows to augment AMDIS, providing researchers with a robust methodology for tackling difficult deconvolutions and enhancing the reliability of their conclusions.
Core Principles: AMDIS and Complementary Tools
Successful deconvolution requires understanding the strengths and weaknesses of available tools. The following table summarizes the core principles and typical applications of AMDIS and key complementary solutions.
Table: Core Principles and Applications of Deconvolution Tools
| Tool Name | Core Algorithm/Principle | Primary Strength | Typical Use Case | Key Limitation |
|---|---|---|---|---|
| AMDIS | Model peak perception & spectral subtraction [7] [16] | Fast, automated; excellent for well-resolved, library-known compounds. | Initial, high-throughput screening of target compounds in complex samples [16]. | Prone to false positives from structurally similar compounds and co-elution [19] [23]. |
| PARADISe | PARAFAC2 (Parallel Factor Analysis 2) [61] | Handles severe co-elution and shifting retention times; provides a pure spectrum. | Resolving complex, untargeted metabolomics data where peaks are highly overlapped [61]. | Requires user-defined retention time windows; can be computationally intensive. |
| AnalyzerPro & Statistical Workflows | Principal Component Analysis (PCA), Target Ion Filtering [19] | Statistically validates identifications; gates results based on definitive ions (e.g., molecular ion). | Confirming identifications and eliminating false positives post-AMDIS or post-PARADISe [19]. | Requires additional data processing step; relies on a priori knowledge of discriminating ions. |
Technical Support & Troubleshooting FAQs
This section addresses common experimental challenges and provides guidance based on a multi-tool strategy.
Q1: My AMDIS report shows a high-confidence hit for a compound, but I suspect it is a false positive from a structurally similar compound in my mixture (e.g., MDA/MDMA). How can I confirm this? [19]
A: This is a classic deconvolution challenge. First, examine the molecular ion region (M+, M+H+) in the deconvoluted spectrum. For MDA/MDMA, the definitive difference is the molecular ion at m/z 180 for MDA, which is absent in MDMA [19]. If AMDIS does not clearly exclude based on this, apply a statistical confirmation workflow:
- Process the raw data through a tool like AnalyzerPro XD or similar software capable of multi-function data analysis.
- Apply a target ion filter in the low-energy function (where molecular ions are most prevalent). Set the filter to require the presence of the unique molecular ion for the suspected compound.
- A positive identification should show the correct fragment ion profiles across higher energy functions and the presence of the unique molecular ion. The absence of the specific molecular ion invalidates the AMDIS hit, allowing you to filter it out as a false positive [19].
Q2: I am working with untargeted metabolomics data where many peaks are severely co-eluted. AMDIS results seem incomplete or messy. What is a better approach? [7] [61]
A: For severely co-eluted peaks, a model-based algorithm like PARAFAC2, implemented in PARADISe, is more appropriate than AMDIS's model peak approach [61]. PARAFAC2 can mathematically resolve components even when their chromatographic profiles are highly overlapped and not perfectly aligned across samples.
- Protocol: Import your .CDF data files into PARADISe. Define retention time intervals covering the region of co-elution. The software will iteratively resolve the number of components, their pure mass spectra, and their concentration profiles across samples [61]. This output is more reliable for highly complex regions and is essential for discovering unknown compounds not in libraries.
Q3: I have processed a batch of samples with PARADISe and have a list of resolved components. How do I efficiently identify them and check for consistency across my sample set? [23] [61]
A: PARADISe excels at deconvolution but benefits from downstream validation.
- Identification: PARADISe can interface directly with NIST MSSEARCH libraries to propose identifications for each resolved pure spectrum [61]. Always check the match factor and visually inspect the spectral match.
- Cross-Sample Validation: To avoid "missing value" problems where a peak is detected in only some samples, use a complementary software script (e.g., in Matlab or R) designed for this purpose. Such a tool can align peaks across all samples in a set, integrate areas consistently, and flag outliers, ensuring a robust data matrix for statistical analysis [23].
Q4: In a high-throughput screening context, what is a practical workflow to maximize speed while minimizing false reports? [19] [16]
A: Implement a tiered confirmation strategy:
- Tier 1 (Screening): Use AMDIS with a moderately high match factor (e.g., 70-80) for rapid processing of all samples. This generates a candidate list [16].
- Tier 2 (Confirmation): Subject all positive findings from Tier 1 to a targeted statistical filter. Using the original raw data, confirm the presence of 1-2 unique and ions (preferably the molecular ion) for each compound. This step, which can be automated in software like AnalyzerPro, will eliminate the majority of structurally similar false positives [19].
- Tier 3 (Investigation): For any ambiguous results or novel compounds, use PARADISe for a deep-dive deconvolution of the specific retention time window.
Workflow for Difficult GC-MS Deconvolutions
Q5: Beyond software, what experimental steps can I take during data acquisition to make deconvolution easier and more accurate? [19] [7]
A: Optimize your chromatographic separation to reduce co-elution in the first place. When using ASAP-MS or other techniques with collision energy ramping, ensure your method includes a low-energy function (e.g., 15V) to preserve the molecular ion, which is the most critical differentiator for similar compounds [19]. For comprehensive screening, employ techniques like GC×GC-TOFMS which vastly increases peak capacity, making deconvolution inherently simpler [62].
Essential Experimental Protocols
Protocol 1: Statistical Confirmation of AMDIS Results to Eliminate False Positives
This protocol is based on work by SpectralWorks to distinguish MDMA from MDA using ASAP-MS [19].
- Objective: To apply a post-deconvolution statistical filter to remove false positives arising from structurally similar compounds.
- Materials: Waters RADIAN ASAP-MS (or equivalent), AnalyzerPro XD software (or equivalent statistical package).
- Method:
- Acquisition: Analyze samples using a multi-function scan method (e.g., m/z 50-650) with stepwise increasing cone voltages (e.g., 15V, 25V, 35V, 50V). The low voltage (15V) function is crucial for preserving molecular ions [19].
- Initial Deconvolution: Process the data file through AMDIS using a standard target library to generate initial identifications.
- Statistical Filtering: Import the raw data into AnalyzerPro XD. For each compound flagged by AMDIS, define a "target ion filter" rule. The rule must require that the unique molecular ion (e.g., m/z 180 for MDA) is present above a signal-to-noise threshold in the low-energy function data. Compounds failing this rule are rejected.
- Validation: Review the PCA plot of the data. True positives should show separation in the principal component dominated by the low-energy function, while false positives will cluster incorrectly [19].
Protocol 2: Resolving Severe Co-elution with PARADISe
This protocol follows the general guidelines for using PARADISe for untargeted GC-MS data [63] [61].
- Objective: To deconvolute highly overlapping chromatographic peaks and obtain pure component spectra.
- Materials: PARADISe software (Version 6.0.1 or higher), GC-MS data in .CDF format.
- Method:
- Data Import: Launch PARADISe and import your .CDF data files.
- Region Definition: Visually inspect the Total Ion Chromatogram (TIC). Define retention time intervals that encompass regions of complex, overlapped peaks.
- PARAFAC2 Modeling: For each interval, initiate the PARAFAC2 deconvolution. Specify an estimated number of components or let the software estimate it. The algorithm will iteratively resolve the data into mathematical components.
- Output & Identification: The output is a peak table containing the resolved pure spectrum and elution profile for each component. Use the integrated NIST MSSEARCH link or export spectra for library matching. The pure spectra are ideal for library searches as they are free of co-eluting interference [61].
Confirmation Strategy for Similar Compounds
The Scientist's Toolkit: Key Research Reagent Solutions
Table: Essential Materials for Advanced GC-MS Deconvolution Studies
| Item / Reagent | Function / Purpose | Critical Specification / Note |
|---|---|---|
| Deuterated Internal Standards | Corrects for instrument variability and matrix effects during quantification; essential for reliable peak area integration across samples. | Select compounds structurally analogous to your analytes (e.g., d5-MDMA for MDMA quantification). |
| NIST/EPA/NIH Mass Spectral Library | Reference database for compound identification via spectral matching. The cornerstone of GC-MS identification [7]. | Use the latest version. AMDIS and PARADISe can interface directly with the NIST MSSEARCH software [16] [61]. |
| Retention Index Marker Mix | Allows calculation of Kovats Retention Indices (RI), a system-independent identifier that complements spectral matching and reduces false positives. | A standard alkane series (e.g., C8-C40) analyzed under identical conditions as samples. |
| Analytical Grade Derivatization Reagents | Renders non-volatile metabolites (acids, sugars, etc.) volatile and thermally stable for GC-MS analysis, expanding metabolome coverage [7]. | Common reagents: MSTFA (for trimethylsilylation), methoxyamine hydrochloride. Purity is critical to avoid artifact peaks. |
| Quality Control (QC) Pooled Sample | A homogenized mix of all study samples run repeatedly throughout the sequence. Monitors instrument stability and validates deconvolution consistency. | Prepared from aliquots of all experimental samples. Essential for batch correction in untargeted studies. |
Establishing a Standardized Reporting Framework for Deconvolution Confidence
This technical support center is designed as a resource for researchers, scientists, and drug development professionals working with GC-MS and AMDIS deconvolution. A core challenge in this field is the risk of false positive identifications, where compounds are incorrectly reported due to co-elution, spectral similarity, or suboptimal analysis parameters [64] [7]. These false positives can compromise downstream analyses, leading to erroneous biological interpretations and costly validation efforts. This center provides focused troubleshooting guides, FAQs, and methodological frameworks aimed at systematically reducing these errors by enhancing the rigor, transparency, and reproducibility of deconvolution reporting.
Troubleshooting Guides: Addressing Common Deconvolution Errors
Issue 1: High Rate of False Positive Identifications in Complex Samples
- Problem: AMDIS or similar software reports many unlikely compounds, especially in regions of high co-elution.
- Root Cause: This often stems from an overly permissive deconvolution setting (low "Match Factor" threshold, wide deconvolution windows) or the use of an inappropriate or uncurated mass spectral library [64].
- Solution:
- Tighten Deconvolution Parameters: Incrementally increase the required "Match Factor" and "Reverse Match" thresholds. Adjust the "Component Width" and "Adjacent Peak Subtraction" settings to better match your chromatographic peak shape [7].
- Employ a Custom Library: Build or use a project-specific library containing only metabolites relevant to your sample type (e.g., human metabolome, plant volatiles). This drastically reduces spurious matches to irrelevant compounds [64].
- Implement Retention Index (RI) Filtering: Use a standardized RI system (e.g., alkane series). Configure your software to only accept identifications where the library match's predicted RI falls within a tight window (e.g., ±5 index units) of the observed RI [65].
- Manual Verification: For critical results, mandate visual inspection of the deconvoluted spectrum against the library spectrum and examination of the extracted ion chromatograms for purity.
Issue 2: Inconsistent Quantification of the Same Compound Across Samples
- Problem: Peak areas for a confirmed metabolite show high variability not explained by biology.
- Root Cause: Inconsistent peak modeling due to shifting baselines, background noise, or partial co-elution that changes from sample to sample.
- Solution:
- Standardize Pre-Processing: Apply consistent smoothing, baseline correction, and noise analysis algorithms to all files before deconvolution [7].
- Use an Internal Standard (IS): Spike a known amount of a non-endogenous compound into every sample during preparation. Normalize all peak areas to the IS response to correct for injection and instrument variability [66].
- Review Integration Bounds: For key compounds, manually verify and, if necessary, adjust the integration start and end points to ensure consistency across all chromatograms.
- Validate with Calibrators: Run a series of calibration standards to confirm the linearity and reproducibility of the quantification for your target analytes under the chosen deconvolution settings.
Issue 3: Failure to Detect or Deconvolve Low-Abundance or Co-eluting Peaks
- Problem: Known low-level metabolites are missed, or severely co-eluted peaks are reported as a single component.
- Root Cause: Insufficient signal-to-noise ratio (SNR) or deconvolution algorithms failing to distinguish components with highly similar spectra and retention times [64].
- Solution:
- Optimize Instrument Sensitivity: Ensure proper instrument maintenance (clean ion source, tune MS) and method optimization (injection technique, column condition) to maximize SNR [66].
- Adjust Noise and Sensitivity Settings: In AMDIS, lower the "Noise Factor" and adjust the "Minimum Match" and "Shape Requirements" settings to be more sensitive to smaller, narrower peaks [7].
- Targeted Ion Extraction: If a specific ion is unique to the low-abundance compound, use extracted ion chromatograms (EICs) for that specific mass to improve detection before attempting deconvolution.
- Leverage Advanced Algorithms: For critical applications, consider complementary software tools (e.g., PARAFAC, MCR-ALS) that use multivariate analysis for more powerful separation of co-eluted signals.
Frequently Asked Questions (FAQs)
Q1: What are the most critical parameters in AMDIS that influence false positive rates, and what are recommended starting values? A: The most critical parameters are the Match Factor (recommended start: ≥70), Reverse Match (recommended start: ≥70), and Deconvolution Width/Window settings. The width should be set slightly wider than your average chromatographic peak at half height. Using overly wide windows increases the chance of blending separate components, while overly narrow windows can split a single peak [7].
Q2: How can I statistically assess the confidence of my deconvolution results, rather than relying solely on software-reported match scores? A: Confidence can be assessed through a multi-parameter scoring system. Develop a framework that combines: 1) Spectral Match Score (from AMDIS/NIST), 2) Retention Index/Time Deviation (absolute difference from library standard), and 3) Peak Purity Metrics (e.g., symmetry, width at half-height relative to pure standards). Results should be binned into confidence tiers (e.g., High, Medium, Low) based on composite scores [67]. The MEAD framework provides a statistical model for quantifying uncertainty in deconvolution estimates [67].
Q3: Our lab analyzes diverse sample types. Should we use one universal deconvolution method or develop specific ones? A: Develop sample-type-specific methods. A universal method is often a compromise that increases false positives and negatives. Personalized or context-specific reference panels and parameters significantly improve accuracy [68]. Create and validate separate methods (with tailored libraries and parameters) for, e.g., plasma, urine, and plant extracts. The imply algorithm demonstrates the power of using personalized reference panels for different subject groups [68].
Q4: What is the best practice for documenting and reporting deconvolution methods to ensure reproducibility? A: Adopt a standardized reporting checklist. Every publication or report should explicitly state:
- Software & Version: e.g., AMDIS 2.73, NIST Library version.
- Critical Parameters: Noise factor, component width, match thresholds, RI tolerance.
- Library Details: Name and version of mass spectral library; if custom, describe its composition.
- Validation Protocol: Description of how deconvolution accuracy was assessed (e.g., using pure standards or spike-ins).
- Confidence Thresholds: Define the composite score cut-offs used for High/Medium/Low confidence identifications [64] [7].
Data Presentation: Performance Metrics of Deconvolution Approaches
Table 1: Comparison of Deconvolution Software and Confidence Metrics
| Software/Method | Key Principle | Reported Accuracy/Performance | Key Metric for Confidence | Advantage for Reducing False Positives |
|---|---|---|---|---|
| AMDIS (Standard) [7] | Model peak shape from pure ions, subtract from composite spectrum. | Widely used; performance varies with parameters. | Match Factor, Reverse Match, RI Deviation. | Good baseline tool; highly configurable. |
| PYQUAN Workflow [64] | AMDIS for ID, then custom Python script for quantification with visual QC. | >97% correct ID/quantification for peaks <10s apart. | Automated + Visual inspection of each peak. | Integrates automated and manual validation. |
| MEAD Framework [67] | Statistical error-in-variable model correcting for platform scaling & noise. | Provides confidence intervals for proportion estimates. | p-values, confidence intervals for estimates. | Quantifies uncertainty; robust for downstream stats. |
| Imply Algorithm [68] | Uses longitudinal data to create personalized reference panels. | Reduced bias vs. single-reference methods in simulations. | Improved correlation with ground truth. | Accounts for inter-individual heterogeneity. |
Table 2: Proposed Tiers for Reporting Deconvolution Confidence
| Confidence Tier | Spectral Match (NIST) | Retention Index Match | Peak Purity / Shape | Required Action for Reporting |
|---|---|---|---|---|
| Level 1 (High) | Match ≥ 80 & Reverse ≥ 80 | RI within ± 5 units | Symmetric, matches standard shape. | Can be reported as identified. |
| Level 2 (Medium) | Match ≥ 70 & Reverse ≥ 70 | RI within ± 10 units OR not available. | Mild asymmetry or broadening. | Report as "tentatively identified" or "putative". |
| Level 3 (Low) | Match < 70 OR spectral ambiguity. | RI deviation > 10 units. | Severe tailing, co-elution evident. | Report as "unknown feature" with m/z and RI. |
Experimental Protocols
Protocol 1: Systematic Optimization of AMDIS Parameters Using a Standard Mixture This protocol establishes a lab-specific, optimized deconvolution method.
- Prepare Calibration Mix: Create a mixture of 20-30 authentic standards covering a range of retention times and compound classes relevant to your work.
- Acquire GC-MS Data: Run the mixture in triplicate under your standard analytical conditions.
- Iterative Parameter Testing: Create multiple AMDIS analysis methods, varying one key parameter at a time (Component Width, Noise Factor, Match Threshold) over a reasonable range.
- Evaluate Performance: For each method, record: a) the true positive rate (correctly identified standards), b) the false positive rate (extra, incorrect identifications), and c) the quantification accuracy (peak area consistency across replicates).
- Select Optimal Settings: Choose the parameter set that maximizes true positives and quantification accuracy while minimizing false positives. This set becomes your lab's validated base method.
Protocol 2: Construction and Validation of a Custom In-House Mass Spectral Library A curated library is the most effective tool for reducing false positives [64].
- Analyze Pure Standards: Inject individual authentic chemical standards. Deconvolute peaks using stringent settings to obtain a clean spectrum.
- Append Metadata: To each spectrum, add essential metadata: a) Confirmed identity, b) Experimental Retention Time under your conditions, c) Calculated Retention Index vs. an alkane series, d) Chemical class.
- Library Curation: Combine these into a dedicated user library. Regularly review and remove entries with poor-quality spectra.
- Validation: Test the library by analyzing a separate, more complex standard mixture. Verify that it yields higher-confidence identifications and fewer spurious matches than a general-purpose library (e.g., full NIST).
Mandatory Visualization
Standardized GC-MS Deconvolution & Confidence Assignment Workflow
Statistical Inference to Control False Positives in Deconvolution
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Software for Robust Deconvolution Studies
| Item | Function & Role in Reducing False Positives | Example / Specification |
|---|---|---|
| Authentic Chemical Standards | To build custom spectral libraries and validate retention times/indices. Critical for grounding identifications in empirical data. | Commercial metabolite standards (e.g., from Sigma-Aldrich, Cayman Chemical). |
| Retention Index Marker Mix | Allows calculation of Kovats Retention Indices (RI), a system-independent identifier for filtering library matches. | n-Alkane series (C8-C40) or fatty acid methyl ester (FAME) mix. |
| Internal Standard (IS) Mixture | Corrects for analytical variability in sample prep and injection, improving quantification accuracy across samples. | Stable isotope-labeled analogs of target compounds or non-biological compounds (e.g., deuterated metabolites). |
| Customizable Deconvolution Software | Core tool for data processing. Software that allows detailed parameter control and custom library import is essential. | AMDIS (free) [65], MetaboliteDetector, or commercial tools (ChromaTOF). |
| Statistical Inference Package | To move beyond point estimates and quantify uncertainty in deconvolution results for rigorous group comparisons. | R/Bioconductor packages (e.g., ISLET for imply [68] or implementations of MEAD-like frameworks [67]). |
| High-Purity Solvents & Inert Supplies | Prevents chemical noise and background contamination that can be mis-identified as sample components. | GC-MS grade solvents, deactivated inlet liners, high-temperature septa [66]. |
Conclusion
Effectively reducing false positives in AMDIS deconvolution is not about a single fix but requires a systematic, multi-layered strategy. This begins with a foundational understanding of the software's algorithmic behavior and is built upon through meticulous optimization of deconvolution parameters and, most powerfully, the creation of application-specific custom libraries. Vigilant troubleshooting of match factors and peak detection, followed by rigorous validation against known standards and complementary chemometric methods like RAMSY or PARAFAC2-based tools, forms the final pillar of a reliable workflow. For biomedical and clinical research, implementing these practices translates to more trustworthy metabolomic profiles, which are essential for discovering robust biomarkers, understanding disease mechanisms, and assessing drug metabolism. The future points toward greater automation and integration of these optimization and validation steps directly into analysis pipelines, making high-fidelity deconvolution more accessible and further solidifying GC-MS as a cornerstone of quantitative metabolomics.
References
- 1. Metabolomics by Gas Chromatography-Mass Spectrometry [https://pmc.ncbi.nlm.nih.gov/articles/PMC4829120/]
- 2. The AMDIS Strawberry User Library from Korean Germplasm [https://www.mdpi.com/2311-7524/10/8/882]
- 3. A new method for the automated selection of the number ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267013011380]
- 4. Enhancing Chimeric Fragmentation Spectra Deconvolution ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12620602/]
- 5. Scalability of mass spectrometry-based metabolomics for ... [https://pubs.rsc.org/en/content/articlehtml/2025/np/d5np00034c]
- 6. Using the Three Cs of Data Visualization as a Life Raft ... [https://www.chromatographyonline.com/view/using-the-three-cs-of-data-visualization-as-a-life-raft-when-you-re-drowning-in-multidimensional-chromatography-peaks]
- 7. Spectral Deconvolution for Gas Chromatography Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3962095/]
- 8. Co-Elution of two peaks with almost same Retention Time [http://www.chromforum.org/viewtopic.php?t=19671]
- 9. - Gc /ms - ibbr-cnr ms [https://www.ibbr.cnr.it/ibbr/facilities/gc-ms-ms]
- 10. Dereplication of Natural Products Using GC-TOF Mass ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5044510/]
- 11. Retention time shift and co-elution after first injection on ... [https://support.waters.com/KB_Inst/Chromatography/WKB4104_Retention_time_shift_and_co-elution_after_first_injection_on_ACQUITY_UPLC_H-Class_System]
- 12. Quantitative measurements via co - elution and dual-isotope detection... [https://pubmed.ncbi.nlm.nih.gov/1783669/]
- 13. Bayesian approach to peak deconvolution and library ... [https://www.sciencedirect.com/science/article/pii/S000326701730750X]
- 14. Next-generation Interactomics: Considerations for the Use ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6944233/]
- 15. Comparative evaluation of software for deconvolution ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993607002439]
- 16. AMDIS: Setting Up and Running a Deconvolution and ... [https://www.sepscience.com/ms-solutions-20-amdis-setting-up-and-running-a-deconvolution-and-target-analysis-part-1-7280]
- 17. Data Deconvolution Techniques in Analytical Chemistry [https://www.leco.com/technique/deconvolution/]
- 18. ADAP-GC 3.0: Improved Peak Detection and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5544921/]
- 19. Statistical tools to eliminate false positives during rapid ... [https://spectralworks.com/2025/09/15/statistical-tools-to-eliminate-false-positives-during-rapid-testing-for-illicit-drugs-using-asap-ms/]
- 20. Gas chromatography – mass spectrometry data processing ... [https://www.sciencedirect.com/science/article/pii/S0021967317306489]
- 21. On the road to automation: a comparative review ... [https://www.sciencedirect.com/science/article/pii/S0165993625001542]
- 22. An actionable annotation scoring framework for gas ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9719826/]
- 23. A software complement to AMDIS for processing GC-MS ... [https://www.sciencedirect.com/science/article/abs/pii/S000326971100234X]
- 24. Identifying and quantifying metabolites by scoring peaks of GC ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4307155/]
- 25. Filtering Out False Positives [https://www.chromatographyonline.com/view/filtering-out-false-positives-0]
- 26. Gaining Sensitivity in Environmental GC–MS [https://www.chromatographyonline.com/view/gaining-sensitivity-environmental-gc-ms]
- 27. Fundamentals of Benchtop GC–MS Data Analysis and ... [https://www.chromatographyonline.com/view/fundamentals-of-benchtop-gc-ms-data-analysis-and-terminology]
- 28. Guide to GC Column Selection and Optimizing Separations [https://www.restek.com/articles/guide-to-gc-column-selection-and-optimizing-separations?srsltid=AfmBOoqB7qzL4zQdTG8_2gqRKLZlIMZevTFpvzbC1H4JPRk0_foQ3Vx8]
- 29. A software complement to AMDIS for processing GC-MS ... [https://pubmed.ncbi.nlm.nih.gov/21575589/]
- 30. Troubleshoot Custom Databases [https://auth0.com/docs/authenticate/database-connections/custom-db/error-handling]
- 31. Troubleshooting Database Issues Like a Pro [https://medium.com/@adamf_64691/troubleshooting-database-issues-like-a-pro-4984bfc3b77a]
- 32. Database Troubleshooting and How-to Guides [https://confluence.atlassian.com/kb/database-troubleshooting-and-how-to-guides-744326149.html]
- 33. Dereplication of Natural Products Using GC-TOF Mass ... [https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2016.00059/full]
- 34. Correction of gas chromatography–mass spectrometry long ... [https://www.nature.com/articles/s41598-025-24794-y]
- 35. Fast and automated biomarker detection in breath samples ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9004778/]
- 36. Towards robust medical machine olfaction: Debiasing GC-MS ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0314742]
- 37. Advancing non-target analysis of emerging environmental ... [https://www.sciencedirect.com/science/article/pii/S0160412025001552]
- 38. When GC Retention Times Shift: Practical Advice for ... [https://www.sepscience.com/when-gc-retention-times-shift-practical-advice-for-operators-10822]
- 39. Automating GC Retention Index Calibration to Enable more ... [https://labscievents.pittcon.org/event/pittcon-2025/planning/UGxhbm5pbmdfMjQ1MzI2Nw==]
- 40. Use of the Retention Index to Secure Correct Identities in ... [https://www.nelsonlabs.com/use-of-the-retention-index-to-secure-correct-identities-in-gc-ms/]
- 41. Filtering Out False Positives [https://www.chromatographyonline.com/view/filtering-out-false-positives]
- 42. gas chromatography retention time prediction and isomer ... [https://pubs.rsc.org/en/content/articlehtml/2025/dd/d4dd00369a]
- 43. How to Avoid False Positives and False Negatives in ... [https://www.acdlabs.com/blog/how-to-avoid-false-positives-and-false-negatives-in-analytical-chemistry/]
- 44. Real-Time Peak in Detection Sinusoidal... - GeeksforGeeks Noisy [https://www.geeksforgeeks.org/data-analysis/real-time-peak-detection-in-noisy-sinusoidal-time-series/]
- 45. On the estimation of false positives in peptide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3076744/]
- 46. LC–MS Sensitivity : Practical Strategies to Boost Your Signal and... [https://www.chromatographyonline.com/view/lc-ms-sensitivity-practical-strategies-boost-your-signal-and-lower-your-noise]
- 47. New Library-Based Methods for Nontargeted Compound ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11844893/]
- 48. Performance evaluation of non-targeted peak-based cross ... [https://www.sciencedirect.com/science/article/abs/pii/S0021967312006401]
- 49. Intelligent Workflow and Software for Non-Target Analysis ... [https://www.mdpi.com/1420-3049/28/8/3409]
- 50. How do you set up the Purity Thresholds in Empower 3? [https://support.waters.com/KB_Inf/Empower_Breeze/WKB11215_How_do_you_set_up_the_Purity_Thresholds_in_Empower_3]
- 51. Minimize the Detection of False Positives by the Software ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2749487/]
- 52. Mass Spectral Libraries: The Next Generation [https://www.spectroscopyonline.com/view/mass-spectral-libraries-next-generation]
- 53. FLARE: a fast and flexible workflow for identifying RNA editing ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-023-05452-4]
- 54. Overcoming a false-positive mechanism in RapidFire MRM ... [https://www.sciencedirect.com/science/article/pii/S2472555225000450]
- 55. Systematic forensic toxicological analysis by GC - MS in serum using... [https://pubmed.ncbi.nlm.nih.gov/26333204/]
- 56. Identifying and quantifying metabolites by scoring peaks of GC - MS data [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-014-0374-2]
- 57. Benchmarking Quantitative Label-Free LC-MS Data ... [https://pubmed.ncbi.nlm.nih.gov/26585461/]
- 58. A multiple-method comparative study using GC-MS, AMDIS and... [https://pubmed.ncbi.nlm.nih.gov/34523187/]
- 59. [PDF] A software complement to AMDIS for... | Semantic Scholar [https://www.semanticscholar.org/paper/A-software-complement-to-AMDIS-for-processing-GC-MS-Behrends-Tredwell/4fa81fbd7ae4154112eff26d92650850816dd2d7]
- 60. The Role of GC/MS in Forensic Toxicology [https://conquerscientific.com/the-role-of-gc-ms-in-forensic-toxicology-identifying-substances-in-biological-samples/]
- 61. PARADISe [https://ucphchemometrics.com/paradise/]
- 62. High-efficiency effect-directed analysis (EDA) advancing ... [https://www.sciencedirect.com/science/article/pii/S0160412024004410]
- 63. Tutorial on PARADISe PARAFAC2-based Deconvolution ... [https://www.protocols.io/view/tutorial-on-paradise-parafac2-based-deconvolution-36wgqdwdovk5/v2]
- 64. PYQUAN: A rapid workflow around the AMDIS deconvolution software for high throughput analysis of pyrolysis GC/MS data - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S0165237015302400]
- 65. AMDIS Literature and Downloads [http://www.amdis.net/What_is_AMDIS/AMDIS_Literature_and_Downloads/amdis_literature_and_downloads.html]
- 66. 10 Common Mistakes in Gas Chromatography [https://aelabgroup.com/10-common-mistakes-in-gas-chromatography/]
- 67. Statistical Inference for Cell Type Deconvolution [https://arxiv.org/html/2202.06420v5]
- 68. imply: improving cell-type deconvolution accuracy using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10557724/]