Decoding Nature's Molecules: A Modern Guide to HR-ESIMS and NMR for Natural Product Structure Elucidation
This comprehensive guide explores the integrated application of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the definitive structural characterization of natural products.
Decoding Nature's Molecules: A Modern Guide to HR-ESIMS and NMR for Natural Product Structure Elucidation
Abstract
This comprehensive guide explores the integrated application of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the definitive structural characterization of natural products. Tailored for researchers, scientists, and drug development professionals, the article systematically covers foundational principles, advanced methodological workflows, critical troubleshooting strategies, and comparative validation approaches. We detail how the synergistic combination of these techniques provides unambiguous molecular formula determination, functional group identification, and complete stereochemical assignment, which is essential for advancing drug discovery from natural sources. The content addresses current best practices, common analytical pitfalls, and emerging trends, providing a practical framework for efficient and accurate structure elucidation in complex biological matrices.
The Essential Toolkit: Understanding HR-ESIMS and NMR Fundamentals for Natural Product Analysis
Application Notes and Protocols: HR-ESIMS in Natural Product Structure Elucidation
Within the integrated framework of a thesis employing HR-ESIMS and NMR for natural product discovery, HR-ESIMS provides the first critical layer of structural information: the unambiguous determination of the molecular formula. This serves as the essential foundation upon which NMR experiments build to deduce full connectivity and stereochemistry. The following notes and protocols detail the core principles and practical applications.
Quantitative Parameters for Molecular Formula Confirmation
The confidence in a proposed molecular formula hinges on three measurable, orthogonal parameters from the HR-ESIMS spectrum.
Table 1: Key HR-ESIMS Metrics for Formula Determination
| Parameter | Definition & Ideal Target | Role in Formula Assignment | Acceptable Error Tolerance (for Confirmation) |
|---|---|---|---|
| Mass Accuracy (Δ ppm) | Difference between measured and theoretical mass, expressed in parts per million. | Filters possible formulas from a candidate list. Higher accuracy dramatically reduces the number of possibilities. | Typically < 5 ppm, with < 3 ppm being good and < 1 ppm excellent for confident assignment. |
| Resolution (R) | Ability to distinguish two adjacent peaks (R = m/Δm, where Δm is peak width at 50% height). | Enables separation of isobaric ions (e.g., [M+H]+ from adducts) and resolution of isotopic fine structure. | R > 20,000 (FWHM) is considered "high resolution." For natural products (m/z ~< 1200), R > 50,000 is often desirable. |
| Isotopic Pattern Fidelity | Match between the observed and theoretical distribution of isotopic peaks (e.g., [M]+, [M+1]+, [M+2]+). | Provides a "fingerprint" dependent on the number and type of atoms (C, Cl, Br, S, etc.). The most definitive single filter. | A high spectral similarity match (e.g., > 90%) between experimental and simulated patterns is required. |
Detailed Experimental Protocols
Protocol 2.1: High-Resolution Mass Spectrometry Data Acquisition for Natural Product Extracts Objective: To obtain accurate mass and isotopic pattern data for a purified natural product or a complex mixture fraction.
Materials & Reagents:
- Purified natural product compound or HPLC fraction (concentration ≥ 0.1 mg/mL in suitable solvent).
- LC-MS grade solvents: Methanol, Acetonitrile, Water.
- Volatile acid/base modifiers: Formic acid, Ammonium hydroxide.
- Calibrant solution: ESI-L Low Concentration Tuning Mix (or equivalent, e.g., sodium formate cluster ions).
Procedure:
- Instrument Calibration: Perform external mass calibration using the calibrant solution, covering the expected m/z range (typically 100-2000). For the highest accuracy, internal calibration or post-acquisition lock mass correction is recommended.
- Sample Preparation: Dilute the sample to an appropriate concentration (avoiding detector saturation) in a solvent compatible with ESI (e.g., MeOH:H₂O, 1:1, v/v). For ion suppression assessment, analyze in both positive and negative ionization modes.
- Parameter Setup:
- Set the mass spectrometer to its maximum resolving power setting (e.g., ~60,000-100,000 FWHM at m/z 200-400).
- Configure the source: Capillary voltage (3-4 kV), source temperature (300-400°C), desolvation gas flow.
- Set scan range to encompass the expected ion(s) and potential adducts (e.g., m/z 150-1500).
- Acquire data with sufficient transients/scan to ensure a high signal-to-noise ratio for isotopic peaks.
- Data Acquisition: Inject the sample via direct infusion (for pure compounds) or LC-HRMS (for mixtures). Acquire data for 1-3 minutes in infusion mode or over the entire LC run.
- Data Processing: Extract the accurate m/z for the ion of interest (e.g., [M+H]⁺, [M+Na]⁺, [M-H]⁻). Apply lock mass correction if available. Generate an averaged mass spectrum across the peak apex.
Protocol 2.2: Molecular Formula Determination from HR-ESIMS Data Objective: To derive and confirm the molecular formula using mass accuracy and isotopic pattern matching.
Procedure:
- Ion Identification: From the averaged spectrum, identify the most abundant ion signal from the compound. Note all potential adducts ([M+Na]⁺, [M+K]⁺, [M+NH₄]⁺, [M-H]⁻, [M+FA-H]⁻) for cross-verification.
- Formula Generation: Input the following into a molecular formula generator software (e.g., Bruker SmartFormula, Thermo Fisher Elemental Composition):
- Accurate m/z of the ion.
- Permissible elements (e.g., C, H, N, O, P, S, Cl, Br, I, Na, K—based on biological context and NMR hints).
- Heuristic rules: Apply the Nitrogen Rule, check for reasonable Double Bond Equivalents (DBE = 4-40 for typical natural products), and set limits for element counts (e.g., H/C ratio between 0.1 and 3).
- Mass error tolerance (set to 5-10 ppm for initial search).
- Formula Ranking & Filtering:
- The software will output a list of candidate formulas. Primary Filter: Sort candidates by mass accuracy (lowest ppm error).
- Secondary Filter: Compare the experimental isotopic pattern (relative abundances of [M], [M+1], [M+2] peaks) with the theoretical pattern for each top candidate. Use the software's isotopic pattern matching algorithm (providing a similarity score, e.g., mSigma on Orbitrap).
- Final Confirmation: The correct molecular formula will have BOTH a low mass error (< 3-5 ppm) AND a high isotopic pattern fit (e.g., mSigma < 20). Cross-check the formula by confirming it is consistent with all observed adduct masses.
The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for HR-ESIMS Analysis
| Item | Function in HR-ESIMS Analysis |
|---|---|
| LC-MS Grade Solvents | Ultra-pure solvents (MeOH, ACN, H₂O) minimize chemical noise and background ions, ensuring clean spectra and accurate baseline for isotopic patterns. |
| Volatile Ion-Pairing Modifiers | Formic Acid (positive mode) / Ammonium Hydroxide (negative mode) promote efficient and consistent ionization of analytes without leaving residues. |
| Mass Calibration Solution | A precise mixture of known compounds (e.g., ESI Tuning Mix) provides reference peaks across the m/z range for periodic instrument calibration, essential for maintaining mass accuracy. |
| Lock Mass Solution | A reference compound (e.g., phthalates, siloxanes) introduced concurrently with the sample provides a real-time internal m/z correction, achieving sub-ppm mass accuracy. |
| Purified Natural Product Standard | A compound of known structure and formula is used as a system suitability check to validate instrument performance (resolution, accuracy) prior to analyzing unknown samples. |
Visualizations
Title: HR-ESIMS Molecular Formula Determination Workflow
Title: HR-ESIMS and NMR Integrated Structure Elucidation
Within the structural elucidation workflow of natural products, Nuclear Magnetic Resonance (NMR) spectroscopy is indispensable. Following initial profiling by High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS), which provides molecular formula and preliminary functional group hints, NMR offers a definitive, atom-level blueprint. This application note details the core 1D and 2D NMR experiments that form the backbone of this process, providing protocols and contextual interpretation for researchers in natural product and drug discovery.
Key 1D Experiments: The Foundation
1.1 ¹H NMR
- Informational Content: Provides information on the number, type, and electronic environment of hydrogen atoms. Key parameters are chemical shift (δ, ppm), signal integration (proportional to the number of protons), multiplicity (s, d, t, q, m, indicating the number of coupled neighboring protons via the n+1 rule), and coupling constant (J, Hz, indicating dihedral angles and coupling pathways).
- Protocol:
- Sample Preparation: Dissolve 1-5 mg of purified natural product in 0.6 mL of deuterated solvent (e.g., CDCl₃, DMSO-d₆, CD₃OD).
- Acquisition Parameters: Temperature: 298 K. Spectral width: 12-16 ppm. Pulse program: zg. Number of scans (NS): 16-128. Relaxation delay (D1): 1-2 seconds. Acquisition time (AQ): 2-4 seconds.
- Processing: Apply Fourier Transform (FT). Apply automatic or manual phase correction and baseline correction. Reference to residual proto solvent peak (e.g., CHCl₃ at 7.26 ppm).
1.2 ¹³C NMR (Broadband Proton-Decoupled)
- Informational Content: Reveals the number and types of carbon atoms (sp³, sp², sp, carbonyl). Chemical shifts indicate hybridization and electron density. Decoupling from protons yields singlets for each chemically distinct carbon.
- Protocol:
- Sample Preparation: Same as ¹H, but typically requires higher concentration or more scans due to low natural abundance (1.1%).
- Acquisition Parameters: Pulse program: zgpg30 (inverse-gated decoupling to avoid NOE enhancement for quantitative analysis) or zg. NS: 1024-8192. Spectral width: 220-240 ppm. D1: 2-3 seconds (longer due to longer T1 of ¹³C).
- Processing: FT, phase, and baseline correction. Reference to solvent signal (e.g., CDCl₃ central peak at 77.16 ppm).
Table 1: Summary of Key 1D NMR Experiments
| Experiment | Nucleus Observed | Key Information Provided | Typical Acquisition Time |
|---|---|---|---|
| ¹H NMR | ¹H | Proton count, chemical environment, coupling networks, stereochemistry (J). | 1-5 minutes |
| ¹³C NMR | ¹³C | Carbon count, hybridization, functional groups. | 30 minutes - 12 hours |
Key 2D Experiments: Establishing Connectivity
2.1 COSY (Correlation Spectroscopy)
- Informational Content: Identifies scalar (J) couplings between protons that are 2-3 bonds apart (¹H-¹H COSY). Cross-peaks reveal proton-proton connectivity within a spin system (e.g., -CH-CH₂-).
- Protocol:
- Pulse Program: cosygpppqf or cosygs.
- Parameters: Spectral width (F2, F1): Same as ¹H spectrum (e.g., 12 ppm). NS: 4-16 per t1 increment. Number of increments (TD1): 256-512. D1: 1-2s.
- Processing: Square sine-bell window function in both dimensions. Double Fourier Transform. Phase correction to pure absorption mode for diagonal and cross-peaks.
2.2 HSQC (Heteronuclear Single Quantum Coherence)
- Informational Content: Correlates a proton with the carbon to which it is directly bonded (¹J~CH~). Each cross-peak represents a direct C-H pair. Distinguishes protonated carbons (CH₃, CH₂, CH) and shows their chemical shifts.
- Protocol:
- Pulse Program: hsqcetgpsisp2.2 (sensitivity-enhanced, phase-sensitive with adiabatic pulses for ¹³C).
- Parameters: F2 (¹H) width: 12 ppm. F1 (¹³C) width: 160-220 ppm. NS: 4-16. TD1: 256-512. D1: 1-2s.
- Processing: QSINE or sine-bell window functions. Linear prediction in F1. FT. Phase correction.
2.3 HMBC (Heteronuclear Multiple Bond Correlation)
- Informational Content: Correlates a proton with a carbon that is 2-4 bonds away (²,³J~CH~). Key for linking molecular fragments through quaternary carbons and across heteroatoms (e.g., O, N).
- Protocol:
- Pulse Program: hmbcgplpndqf (optimized for long-range couplings, typically ~8 Hz).
- Parameters: Similar to HSQC, but with longer evolution delay for long-range coupling (~60-80 ms). F1 (¹³C) width often wider to capture carbonyls. NS: 8-32.
- Processing: Similar to HSQC. Absolute value mode display is common due to phase modulation.
Table 2: Summary of Key 2D Through-Bond Correlation Experiments
| Experiment | Correlation Type | Key Connectivity Revealed | Primary Use in Structure Elucidation |
|---|---|---|---|
| COSY | ¹H - ¹H (²,³J~HH~) | Proton-proton vicinal/geminal coupling networks. | Building proton spin systems. |
| HSQC | ¹H - ¹³C (¹J~CH~) | Direct C-H bonds. | Assigning all protonated carbons. |
| HMBC | ¹H - ¹³C (²,³J~CH~) | Long-range proton to carbon (including quaternary). | Linking fragments, placing quaternary carbons and heteroatoms. |
2.4 NOESY & ROESY (Through-Space Correlations)
- Informational Content: NOESY (Nuclear Overhauser Effect Spectroscopy) correlates protons that are close in space (<5 Å), regardless of the number of bonds separating them. Critical for determining relative stereochemistry and 3D conformation. ROESY (Rotating frame Overhauser Effect Spectroscopy) is preferred for mid-sized molecules (MW ~1000-2000 Da) where the regular NOE is weak or zero.
- Protocol (NOESY):
- Pulse Program: noesygpphpp (phase-sensitive with water suppression).
- Parameters: Mixing time (d8): Critical; 300-800 ms for small molecules. Spectral width: ¹H width. NS: 16-32. TD1: 512.
- Processing: Sine-bell window, FT, phase correction. Cross-peaks (same sign as diagonal) indicate spatial proximity.
- Protocol (ROESY):
- Pulse Program: roesygpphpp.
- Parameters: Spin-lock mixing time (d8): 150-400 ms.
- Processing: Similar to NOESY. Cross-peaks have opposite sign to diagonal.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for NMR-Based Natural Product Elucidation
| Item | Function & Description |
|---|---|
| Deuterated Solvents (CDCl₃, DMSO-d₆, CD₃OD, Acetone-d₆, D₂O) | Provides a lock signal for the NMR spectrometer and dissolves the sample without adding large interfering proton signals. |
| NMR Tubes (5 mm, 7", 528-PP) | High-quality, matched tubes ensure consistent spinning and spectral resolution. |
| TMS or DSS Reference Standard (Tetramethylsilane, Sodium 2,2-dimethyl-2-silapentane-5-sulfonate) | Primary internal chemical shift reference compound (0.00 ppm). DSS is preferred for aqueous solutions. |
| Shigemi Tubes | Matched microtubes for limited sample quantities (< 1 mg), reducing solvent volume and increasing effective concentration. |
| Anhydrous Solvents & Molecular Sieves | For rigorous sample drying to prevent exchangeable protons (OH, NH) from broadening signals or complicating spectra. |
| NMR Data Processing Software (MestReNova, TopSpin, ACD/Spectrus) | For processing, analyzing, assigning, and reporting 1D/2D NMR data. |
Visualized Workflows
Title: Natural Product Structure Elucidation Workflow
Title: 2D NMR Experiment Selection Logic
Within the broader thesis on advanced spectroscopic techniques for natural product research, the complementary roles of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy form the cornerstone of modern structure elucidation. HR-ESIMS provides precise molecular formula and fragment ion data, while NMR offers definitive atomic connectivity and stereochemistry. This application note details the integrated workflow, protocols, and materials required to harness their synergy for unambiguous characterization of novel compounds.
Application Notes: The Complementary Data Paradigm
Information Domain Comparison
HR-ESIMS and NMR interrogate different molecular properties. Their combined data creates a comprehensive structural picture.
Table 1: Complementary Data Domains of HR-ESIMS and NMR
| Technique | Primary Information | Key Metrics | Throughput | Sample Requirement |
|---|---|---|---|---|
| HR-ESIMS | Exact mass, molecular formula, fragment ions, isotope patterns. | Mass accuracy (< 5 ppm), resolution (> 30,000), MS/MS spectra. | High | Low (µg to ng) |
| 1D/2D NMR | Atomic connectivity, functional groups, stereochemistry, dynamics. | Chemical shift (δ, ppm), coupling constant (J, Hz), integration, NOE/ROE. | Low | High (mg) |
Integrated Elucidation Workflow
The synergistic workflow proceeds iteratively, with data from one technique guiding experiments in the other.
Diagram Title: Integrated HR-ESIMS & NMR Elucidation Workflow
Detailed Experimental Protocols
Protocol A: HR-ESIMS for Molecular Formula Determination
Objective: Obtain exact mass and fragment ions to propose a molecular formula.
Materials & Sample Prep:
- Dissolve purified compound (0.1-1.0 µg) in 1 mL of LC-MS grade methanol or acetonitrile.
- Add 10 µL of 0.1% formic acid (for positive mode) or ammonium hydroxide (for negative mode).
- Dilute serially to a final concentration of ~1-10 ng/µL for infusion.
Instrument Parameters (Example for Q-TOF):
- Ion Source: ESI, positive/negative mode.
- Capillary Voltage: 3.0 kV.
- Desolvation Gas Flow: 800 L/hr at 350°C.
- Source Temperature: 120°C.
- Scan Range: m/z 50-2000.
- Lock Mass Correction: Use reference compound (e.g., leucine enkephalin, m/z 556.2771 for [M+H]⁺).
- MS/MS Acquisition: Collision energy ramped from 10-40 eV for fragmentation.
Data Analysis:
- Use instrument software to calibrate spectrum and identify [M+H]⁺, [M+Na]⁺, or [M-H]⁻ ions.
- Apply elemental composition tool with constraints: C (0-100), H (0-200), O (0-50), N (0-20), common elements.
- Accept formulas with mass error < 3 ppm and isotopic pattern match (iFit) > 90%.
- Interpret key fragment ions to propose substructures.
Protocol B: Comprehensive NMR Analysis for Structural Assignment
Objective: Elucidate full connectivity and stereochemistry based on HR-ESIMS formula.
Sample Preparation:
- Weigh 1-5 mg of purified compound into a clean NMR tube.
- Dissolve in 0.6 mL of deuterated solvent (CDCl₃, DMSO-d₆, CD₃OD). Ensure sample is fully dissolved and free of particulates.
Acquisition Sequence (for a 600 MHz NMR):
- ¹H NMR: Number of scans (NS) = 16-32, relaxation delay (D1) = 1.0 s.
- ¹³C NMR (BB-decoupled): NS = 1024-4096, D1 = 2.0 s. Use the molecular formula from HR-ESIMS to predict the number of expected carbon signals.
- 2D Experiments (Key for Connectivity):
- COSY (Correlation Spectroscopy): Identifies ²J/³J H-H couplings.
- HSQC (Heteronuclear Single Quantum Coherence): Directly correlates each proton to its bonded carbon (¹JCH).
- HMBC (Heteronuclear Multiple Bond Correlation): Correlates protons to carbons 2-4 bonds away (²JCH, ³JCH), establishing key linkages.
- NOESY/ROESY (Nuclear Overhauser Effect Spectroscopy): Identifies protons close in space (< 5 Å), critical for stereochemistry and conformation.
Data Interpretation Workflow:
Diagram Title: NMR Data Interpretation Logic Flow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Integrated HR-ESIMS/NMR Workflow
| Item | Function & Critical Role | Example/Specification |
|---|---|---|
| Deuterated NMR Solvents | Provide a field-frequency lock for stable NMR acquisition; minimize solvent proton signals. | CDCl₃, DMSO-d₆, CD₃OD (99.8% D minimum). |
| LC-MS Grade Solvents | Ultra-pure solvents for ESIMS to minimize background ions and prevent source contamination. | Methanol, Acetonitrile, Water (with 0.1% Formic Acid). |
| Internal Mass Calibrants | Provide real-time lock mass correction for sub-5 ppm mass accuracy in HR-ESIMS. | Leucine Enkephalin, Sodium Formate Cluster. |
| NMR Reference Standards | Calibrate chemical shift scales for accurate proton and carbon reporting. | Tetramethylsilane (TMS) or residual solvent peak (e.g., CHCl₃ at 7.26 ppm). |
| High-Purity Silica Gel | For purification of natural products prior to analysis to avoid interfering impurities. | 40-63 µm, 60 Å pore size for flash chromatography. |
| Microscale NMR Tubes | Enable high-quality NMR data acquisition with sample-limited natural products (≤ 1 mg). | 3 mm or 1.7 mm Capillary NMR Tubes. |
| Solid Phase Extraction (SPE) Cartridges | Rapid desalting and buffer exchange of samples prior to ESIMS analysis. | C18 reversed-phase cartridges. |
Application Notes: Integrating HR-ESIMS and NMR for De Novo Structure Elucidation
Within the broader thesis that modern natural product (NP) research requires the synergistic integration of high-resolution mass spectrometry (HR-ESIMS) and advanced nuclear magnetic resonance (NMR) spectroscopy, this protocol addresses the core challenges of molecular complexity, stereochemical determination, and limited sample availability. The workflow is designed to maximize information gain from sub-milligram quantities.
Table 1: Quantitative Benchmarks for HR-ESIMS and NMR in NP Analysis
| Technique | Key Parameter | Target Performance | Purpose in NP Elucidation |
|---|---|---|---|
| HR-ESIMS | Mass Accuracy | < 2 ppm (routinely < 1 ppm) | Determines elemental composition (C, H, O, N, S, etc.). |
| HR-ESIMS/MS | Resolution (FWHM) | > 50,000 | Isolates precursor ions for fragmentation, provides structural motifs. |
| NMR (Cryoprobe) | Sample Requirement | 10 – 100 µg (1H) | Enables data acquisition on trace quantities. |
| MicroCryoprobe | Sensitivity Gain | 4-5x over room temp probe | Critical for dilute samples or limited isolations. |
| 1D NMR | Experiment Time | 2-5 mins (1H, 50 µg) | Quick assessment of purity, proton count, and major functional groups. |
| 2D NMR | Experiment Time (HSQC) | 30-60 mins (50 µg) | Establishes 1H-13C direct connectivity backbone. |
Detailed Protocols
Protocol 1: HR-ESIMS-Based Molecular Formula Determination and Dereplication
Objective: To obtain an exact molecular formula and screen against NP databases prior to resource-intensive NMR analysis.
- Sample Preparation: Dissolve the purified NP in LC-MS grade methanol or acetonitrile to a concentration of approximately 1-10 ng/µL. Include 0.1% formic acid (for positive mode) or ammonium hydroxide (for negative mode) to promote ionization.
- Instrument Calibration: Calibrate the ESI-TOF or Orbitrap mass spectrometer using a standard mixture (e.g., sodium formate) immediately prior to analysis to ensure sub-2-ppm mass accuracy.
- Data Acquisition: Acquire full-scan HR-ESIMS data in both positive and negative ionization modes (m/z 100-2000). For MS/MS, isolate the [M+H]+ or [M-H]- ion with a 1-2 m/z window and fragment using stepped Collision-Induced Dissociation (CID) energies (e.g., 20, 40, 60 eV).
- Data Analysis:
- Process the centroid data to obtain exact m/z values for the molecular ion and key fragments.
- Use software (e.g., Molecular Formula Calculator) to generate all possible elemental compositions within a 2-ppm error tolerance, applying heuristic rules: LEWIS and SENIOR chemical rules, isotopic pattern matching (A+1, A+2), and consideration of expected elements (C, H, O, N, P, S, Halogens).
- Submit the molecular formula and MS/MS spectra to databases (e.g., GNPS, DNP, Reaxys) for rapid dereplication.
Protocol 2: Microscale NMR Structure Elucidation Workflow
Objective: To determine the planar structure and relative stereochemistry of a novel NP using < 100 µg of sample.
- Sample Preparation for MicroCryoprobe:
- Transfer the purified, dried NP (20-100 µg) into a 1.0 mm or 1.7 mm NMR tube.
- Dissolve in 5-20 µL of deuterated solvent (e.g., CD3OD, DMSO-d6). Use a volatile solvent to allow for recovery after analysis.
- NMR Data Acquisition Sequence (Order is Critical):
- 1H NMR: Single scan or a few scans to check sample solubility and signal-to-noise. Optimize shims.
- 1D Selective NOESY or 1D ROESY: Acquire early on the same sample, as the transient nuclear Overhauser effect (NOE) builds up over time and can be erased by subsequent pulsing.
- 1H-13C gHSQC: Map all protonated carbons.
- 1H-13C gHMBC: Establish 2-3 bond 1H-13C correlations to link structural fragments and locate quaternary carbons.
- 1H-1H COSY or TOCSY: Establish vicinal and geminal proton coupling networks.
- Stereochemical Analysis:
- Relative Configuration: Use 1D selective NOESY/ROESY experiments on key proton pairs to measure through-space correlations. For rigid systems, coupling constant (J-value) analysis from 1H NMR can define dihedral angles.
- Absolute Configuration: If sample allows (>50 µg), prepare derivatives with chiral anisotropic reagents (e.g., Mosher's esters) and compare 1H NMR chemical shift differences (ΔδSR). Alternatively, use computational prediction of Electronic Circular Dichroism (ECD) spectra.
Visualizations
Title: NP Structure Elucidation Workflow
Title: HRMS & NMR Data Synergy
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in NP Research |
|---|---|
| LC-MS Grade Solvents (MeOH, ACN, H2O) | Minimize background noise and ion suppression during HR-ESIMS analysis. |
| Deuterated NMR Solvents (CD3OD, DMSO-d6, CDCl3) | Provide a field-frequency lock and a deuterium signal for shimming; essential for NMR. |
| Microscale NMR Tubes (1.0 mm, 1.7 mm) | Enable analysis of sub-100 µg samples by reducing sample volume, maximizing concentration in the active probe volume. |
| Cryogenic NMR Probe | Increases sensitivity by cooling the detection electronics, reducing thermal noise; essential for low-abundance NPs. |
| Chiral Derivatization Kits (e.g., α-Methoxy-α-(trifluoromethyl)phenylacetic acid (MTPA) chlorides) | Used to convert chiral alcohols/amines into diastereomers for absolute configuration determination via NMR. |
| Solid Phase Extraction (SPE) Cartridges (C18, Diol, Ion Exchange) | For rapid desalting, concentration, or fractionation of crude extracts prior to detailed analysis. |
| Computational Chemistry Software (for ECD/ORD prediction, DFT-NMR calculation) | To compare calculated spectroscopic data with experimental results for stereochemical assignment. |
Application Notes and Protocols within the Context of HR-ESIMS and NMR for Natural Product Structure Elucidation
Sample Purity Assessment
Accurate structure elucidation of natural products via HR-ESIMS and NMR is contingent upon sample purity. Impurities can lead to spectral overlap, mis-assigned signals, and incorrect molecular formula determination.
Quantitative Assessment Methods
Table 1: Common Purity Assessment Techniques and Their Metrics
| Technique | Key Quantitative Metric | Purity Threshold for HR-ESIMS/NMR | Typical Analysis Time |
|---|---|---|---|
| Analytical HPLC-UV/ELSD | Peak Area % (λ=210-254 nm) | >95% (single major peak) | 20-40 min |
| UHPLC-HRMS | (i) UV Peak Homogeneity, (ii) Isotopic Pattern Fit | >90% (for challenging NPs) | 10-20 min |
| 1H NMR (qNMR) | Integral Ratio of Analyte vs. Certified Reference Standard | Quantitative % purity possible | 5-10 min per scan |
| TLC with Densitometry | Spot Intensity/Pixel Density | >90% (single spot, multiple eluents) | 30-60 min |
Protocol: Analytical HPLC for Purity Assessment Pre-NMR
Materials: Purified natural product sample, HPLC-grade solvents, C18 reverse-phase column (150 x 4.6 mm, 3.5 µm), HPLC system with DAD/ELSD.
- Prepare Sample: Dissolve 0.5-1.0 mg of sample in appropriate solvent (e.g., MeOH) to a concentration of ~0.5 mg/mL. Filter through a 0.22 µm PTFE syringe filter.
- Chromatographic Conditions:
- Mobile Phase A: 0.1% Formic acid in H2O.
- Mobile Phase B: 0.1% Formic acid in Acetonitrile.
- Gradient: 5% B to 100% B over 25 minutes.
- Flow Rate: 1.0 mL/min.
- Column Temp: 35°C.
- Detection: DAD (200-400 nm), ELSD.
- Data Analysis: Process chromatogram. The primary peak should represent >95% of the total integrated peak area at 210-254 nm for high-confidence NMR analysis.
Solvent Selection for HR-ESIMS and NMR
Solvent choice directly impacts spectral quality, solubility, and chemical shift referencing.
Critical Selection Criteria
Table 2: Solvent Selection Guide for Natural Product Analysis
| Technique | Preferred Solvents | Key Considerations | Avoid (Interference) |
|---|---|---|---|
| HR-ESIMS (+ve mode) | MeOH, ACN, H2O (+ 0.1% FA) | Volatility, conductivity, adduct formation. | Non-volatile buffers (e.g., phosphate), polymers. |
| HR-ESIMS (-ve mode) | MeOH, ACN, H2O (+ NH4OH) | Deprotonation efficiency. | Acidic modifiers. |
| 1H/13C NMR | CDCl3, DMSO-d6, CD3OD, D2O | Sample solubility, residual solvent peaks, chemical shift, hygroscopicity. | Protonated solvents; impurities. |
| 2D NMR (e.g., COSY, HSQC) | Same as 1D NMR | Must be identical to 1D NMR solvent for consistency. | Solvent mixtures. |
Protocol: Sample Preparation for NMR Spectroscopy
Materials: Highly pure NP sample, deuterated solvent (e.g., CDCl3), NMR tube (5 mm), micropipettes.
- Weighing: Accurately weigh 0.5-2.0 mg of purified compound into a clean vial. Mass accuracy is critical for concentration calculation.
- Solvation: Add ~0.6 mL of selected deuterated solvent via micropipette. Cap and vortex thoroughly for 1-2 minutes. For poorly soluble compounds, gentle warming in a water bath (<40°C) or brief sonication may be used.
- Transfer: Using a Pasteur pipette, transfer the solution to a clean, dry 5 mm NMR tube. Ensure no solid particulates are transferred.
- Concentration Check: For a 1D 1H NMR experiment on a 500 MHz instrument, 0.5-2.0 mg in 0.6 mL typically yields an acceptable S/N ratio for overnight 13C or 2D experiments.
Concentration Requirements
Optimal concentration balances signal-to-noise (S/N) with artifacts like aggregation or viscosity broadening.
Table 3: Recommended Concentration Ranges by Experiment
| Analytical Experiment | Ideal Sample Concentration | Minimum Sample Amount (500 MHz) | Rationale |
|---|---|---|---|
| 1D 1H NMR | 1-10 mM | 0.1-0.5 mg | Sufficient S/N for proton detection in <5 min. |
| 1D 13C NMR (noise decoupled) | 10-50 mM | 2-5 mg | Overcome low natural isotopic abundance. |
| 2D HSQC/HMBC | 2-20 mM | 1-2 mg | Balance of sensitivity and experiment time (1-4 hrs). |
| HR-ESIMS (direct infusion) | 1-10 µM (in MS-compatible solvent) | <1 µg | Avoid ion suppression; suitable for adduct observation. |
Protocol: Determining Optimal NMR Concentration
- Calculate Molarity: Molarity (M) = (mass in g / molecular weight in g/mol) / volume in L. Target 5-10 mM for initial 1H NMR.
- Run a Quick 1H NMR: Acquire a 1-2 scan 1H spectrum.
- Assess:
- If the solvent signal is clipped or analyte signals are saturated, the concentration is too high. Dilute 2-fold and repeat.
- If baseline noise is high and analyte signals are barely distinguishable, concentration is too low. Consider evaporating solvent and re-dissolving in a smaller volume.
- Adjust for 2D Experiments: For insensitive 2D experiments like HMBC, use the highest concentration possible without causing solubility or aggregation issues (often 15-30 mM).
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Pre-Analysis Sample Preparation
| Item | Function in Pre-Analysis |
|---|---|
| Deuterated NMR Solvents (CDCl3, DMSO-d6, CD3OD) | Provide a lock signal for NMR spectrometers, minimize interfering solvent proton signals. |
| HPLC-MS Grade Solvents (MeOH, ACN, H2O) | Ultra-pure solvents for MS and HPLC to minimize background ions and noise. |
| 0.22 µm PTFE Syringe Filters | Remove particulate matter that can clog HPLC systems or cause light scattering in NMR. |
| Certified Reference Standards (e.g., qNMR standards) | Quantitatively determine sample purity and concentration via 1H NMR. |
| Volatile Buffers/Additives (Formic Acid, Ammonium Acetate) | Modulate pH and ionization efficiency in HR-ESIMS without leaving residues. |
| Precision Analytical Balances (µg to mg range) | Accurately weigh sub-milligram quantities of precious natural products. |
| NMR Tube Cleaners/Drying Ovens | Ensure contaminant-free NMR tubes for reproducible results. |
Experimental Workflow Diagrams
Title: Pre-Analysis Workflow for NP Characterization
Title: Factors in Analytical Solvent Selection
From Raw Data to Confirmed Structure: A Step-by-Step HR-ESIMS and NMR Workflow
Within the broader framework of natural product structure elucidation, integrating High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy is foundational. The initial HR-ESIMS analysis serves as the critical first pass, providing the exact molecular mass and a shortlist of probable molecular formulas. This data directly informs subsequent isolation strategies and guides the detailed structural investigation by NMR, creating an efficient, iterative workflow for de novo identification of bioactive compounds.
Core Principles and Data Interpretation
HR-ESIMS determines the mass-to-charge ratio (m/z) of ions with high precision, typically within 5 ppm error or less. The exact mass allows for the calculation of potential elemental compositions by considering combinations of carbon, hydrogen, nitrogen, oxygen, and other elements relevant to natural products (e.g., S, P, Cl, Na, K). The isotopic pattern, particularly the relative abundance of the [M+1] and [M+2] peaks, further refines formula assignment.
Table 1: Key Quantitative Metrics for HR-ESIMS Data Validation
| Parameter | Typical Specification | Role in Formula Assignment |
|---|---|---|
| Mass Accuracy | < 5 ppm (often < 2 ppm) | Defines the allowable error window for candidate formulas. |
| Resolution (FWHM) | > 20,000 | Separates isobaric ions and provides clean isotopic clusters. |
| Isotopic Fidelity | < 5% deviation from theoretical | Confirms elemental composition via [M+1]/[M+2] peak ratios. |
| Double Bond Equivalents (DBE) | Integer or half-integer value | Indicates number of rings and π-bonds, constraining formulas. |
| Nitrogen Rule | Even mass for even # of N atoms | Basic filter for molecular ion validity. |
Detailed Experimental Protocols
Protocol 3.1: Sample Preparation for HR-ESIMS Analysis
Objective: To prepare a purified natural product extract or fraction for accurate mass measurement.
- Solubilization: Dissolve the dry sample in LC-MS grade methanol, acetonitrile, or a water/organic mixture (e.g., 50:50 MeOH:H₂O) to a final concentration of approximately 0.1-1 µg/µL.
- Additive for Ionization: For positive ion mode, add 0.1% (v/v) formic acid to promote [M+H]⁺ formation. For negative ion mode, add 0.1% (v/v) ammonium hydroxide or a neutral buffer (e.g., 10 mM ammonium acetate) to promote [M-H]⁻ formation.
- Filtration: Pass the solution through a 0.22 µm PTFE or nylon syringe filter to remove particulate matter that could clog the instrument capillary.
- Internal Mass Calibration: Introduce a known lock-mass compound (e.g., leucine enkephalin for ESI+ at m/z 556.2766) via a separate reference sprayer, or premix the sample with a calibrant appropriate for the measured m/z range.
Protocol 3.2: Instrument Tuning and Data Acquisition
Objective: To acquire high-fidelity, high-resolution mass spectra.
- Instrument Setup: Utilize a Q-TOF, Orbitrap, or FT-ICR mass spectrometer equipped with an ESI source.
- Parameter Optimization:
- Source Conditions: Adjust capillary voltage (2.5-4 kV), cone voltage (20-60 V), desolvation temperature (200-400°C), and desolvation gas flow (N₂, ~600 L/hr) for optimal ion yield and minimal in-source fragmentation.
- Mass Analyzer Tuning: Calibrate the analyzer using a standard tuning mixture (e.g., sodium formate cluster ions) across the intended mass range (e.g., m/z 50-2000).
- Data Acquisition: Acquire data in centroid mode over a sufficient time (1-2 minutes) to ensure a good signal-to-noise ratio. Collect both full-scan MS data and, if applicable, low-collision-energy MS/MS data for subsequent fragmentation analysis.
Protocol 3.3: Data Processing and Molecular Formula Determination
Objective: To derive the exact mass and generate a ranked list of probable molecular formulas.
- Spectrum Processing: Deisotope the raw spectrum using instrument software. Identify the protonated/deprotonated molecular ion peak ([M+H]⁺ or [M-H]⁻).
- Exact Mass Extraction: Record the m/z value of the monoisotopic peak. Apply lock-mass correction if used.
- Formula Calculation: Input the exact mass into formula calculation software (e.g., Bruker SmartFormula, Thermo Fisher Compound Discoverer, or open-source tools like mMass). Set constraints:
- Elements: C, H, N, O (mandatory); plus S, P, Cl, F, Na, K as relevant.
- Heuristic Filters: Apply typical ranges for natural products: DBE (0-50), element count limits (e.g., C<100, H<200, O<50, N<20), and adherence to the Nitrogen Rule.
- Tolerance: Set mass error tolerance to ±5 ppm.
- Results Ranking & Validation: Rank candidate formulas by mass error. Cross-validate the top candidates by comparing the theoretical and observed isotopic distribution patterns (using Pearson's correlation or percentage difference). The formula with the lowest mass error and best isotopic fit is selected as the most probable.
Visualization of Workflows
Diagram Title: HR-ESIMS First Pass Workflow for Natural Products
Diagram Title: Molecular Formula Determination Logic Flow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for HR-ESIMS First Pass Analysis
| Item | Function & Rationale |
|---|---|
| LC-MS Grade Solvents (MeOH, ACN, H₂O) | Ultra-high purity minimizes background chemical noise and ion suppression, ensuring accurate mass measurement. |
| Volatile Additives (Formic Acid, NH₄OH, Ammonium Acetate) | Promotes efficient ionization in positive or negative mode and stabilizes precursor ions without persistent adducts. |
| PTFE Syringe Filters (0.22 µm) | Removes insoluble particulates that can cause source contamination and unstable spray, leading to mass drift. |
| Mass Calibration Standard (e.g., Sodium Formate, Agilent Tuning Mix) | Provides known m/z ions for internal or external calibration, guaranteeing specified mass accuracy. |
| Lock-mass Reference Compound (e.g., Leu-Enkephalin, HP-0921) | Continuously introduced to correct for minor instrument drift during long acquisition periods. |
| High-Purity Nitrogen Gas (>99.999%) | Serves as the desolvation and nebulizer gas; purity is critical for stable spray and low background. |
Within the comprehensive workflow for natural product structure elucidation, initial NMR profiling forms the critical bridge between HR-ESIMS-derived molecular formula and full structural assignment. Following Step 1 (HR-ESIMS for Formula Determination), Step 2 employs 1D ¹H and ¹³C NMR experiments to map the fundamental carbon skeleton and identify key functional groups. This phase is not about complete elucidation but about efficient triage and hypothesis generation. It answers preliminary questions: Is the compound predominantly aliphatic or aromatic? What are the major proton- and carbon-containing environments? The data from this step directly informs the selection of advanced 2D NMR experiments (Step 3) for full connectivity mapping.
Key Quantitative NMR Parameters & Data Interpretation
Table 1: Diagnostic ¹H NMR Chemical Shift Ranges for Common Functional Groups
| Functional Group | Approximate δH Range (ppm) | Characteristic Multiplicity & Coupling | Key Diagnostic Utility |
|---|---|---|---|
| Aliphatic CH₃, CH₂, CH | 0.5 – 2.5 | Multiplets, broad singlets | Skeleton saturation level, methyl group count |
| Allylic / α to C=O | 1.6 – 3.0 | Multiplet | Proximity to unsaturation/carbonyl |
| Alkynes | 1.7 – 3.1 | Singlet | Acetylenic proton identification |
| Ethers / Alcohols (ROH) | 3.0 – 4.5 | Singlet (broad for OH) | Oxygenated site identification |
| Alkenes | 4.5 – 6.5 | Doublets, triplets, multiplets | Olefinic proton count & substitution |
| Aromatics / Heteroaromatics | 6.0 – 8.5 | Multiplets | Aromatic ring presence, substitution pattern |
| Aldehydes | 9.0 – 10.0 | Doublet (J ~ 1-3 Hz) | Aldehyde group confirmation |
| Carboxylic Acids | 10.0 – 13.0 | Broad singlet | Carboxyl proton, exchangeable |
Table 2: Diagnostic ¹³C NMR Chemical Shift Ranges
| Carbon Type | Approximate δC Range (ppm) | DEPT-135 Signal | Key Diagnostic Utility |
|---|---|---|---|
| Alkyl (C, CH, CH₂, CH₃) | 0 – 60 | CH₃/CH: +ve, CH₂: -ve, C: null | Skeleton mapping, degree of substitution |
| C–O (Alcohols, Ethers) | 50 – 90 | Variable | Oxygenated aliphatic carbon count |
| Alkenes (C=) | 100 – 150 | CH: +ve, C: null | Olefinic carbon count & substitution |
| Aromatics / Heteroaromatics | 110 – 160 | CH: +ve, C: null | Aromatic ring system identification |
| Carbonyls (C=O) | 160 – 220 | Null | Ketone, aldehyde, ester, amide, acid count |
| Nitriles (C≡N) | 115 – 125 | Null | Cyano group identification |
Detailed Experimental Protocols
Protocol 1: Sample Preparation for Initial NMR Profiling
Objective: To prepare a purified natural product sample for high-quality ¹H and ¹³C NMR analysis. Materials: See Scientist's Toolkit. Procedure:
- Weighing: Transfer 1–5 mg of purified, dried compound into a clean, tared vial. Accurate mass recording is critical for concentration calculations.
- Solvent Selection: Add 0.5–0.7 mL of appropriate deuterated solvent (e.g., CDCl₃, DMSO-d₆, CD₃OD). Choice is based on compound solubility and chemical inertness. For broad screening, DMSO-d₆ is often preferred due to its high solvating power.
- Dissolution: Vortex or ultrasonicate the mixture until complete dissolution is achieved. A clear, particulate-free solution is essential.
- Tube Transfer: Using a Pasteur pipette, transfer the solution to a clean, high-quality NMR tube (e.g., 5 mm outer diameter). Avoid introducing air bubbles.
- Capping: Securely cap the NMR tube. Label the tube and cap with a unique sample identifier.
Protocol 2: Acquisition of ¹H NMR Spectrum
Objective: To obtain a high signal-to-noise (S/N) ¹H NMR spectrum for proton counting, integration, and chemical shift/multiplicity analysis. Instrument Setup (Bruker Avance NEO 500 MHz Example):
- Load Sample & Lock: Insert tube, auto-tune, and lock to the deuterium signal of the solvent.
- Shim: Perform automated gradient shimming (command:
topshim). - Pulse Program: Select
zg(standard ¹H acquisition). - Parameters:
- Spectral Width (SW): 20 ppm (centered on residual solvent peak).
- Number of Scans (NS): 16–64.
- Relaxation Delay (D1): 1–2 seconds (≥ 5 * T1 for quantitative integration).
- Acquisition Time (AQ): ~3-4 seconds.
- Receiver Gain (RG): Set automatically or manually to avoid clipping.
- Acquisition: Run the experiment (command:
zg). - Processing:
- Fourier Transform (command:
efp). - Phase Correction (command:
apk). - Baseline Correction (command:
abs). - Referencing: Set the residual solvent peak to its known chemical shift (e.g., CHCl₃ in CDCl₃ to 7.26 ppm).
- Fourier Transform (command:
Protocol 3: Acquisition of ¹³C NMR & DEPT-135 Spectra
Objective: To obtain a ¹³C NMR spectrum for counting carbon types and a DEPT-135 spectrum for distinguishing CH₃, CH₂, and CH groups. Part A: ¹³C NMR Acquisition
- Pulse Program: Select
zgpg30(inverse-gated decoupling to suppress NOE for semi-quantitative analysis). - Parameters:
- Spectral Width (SW): 240 ppm.
- Number of Scans (NS): 1024 – 4096 (due to low natural abundance).
- Relaxation Delay (D1): 2 seconds.
- Acquisition Time (AQ): ~1.3 seconds.
- Acquisition & Processing: Run experiment. Process with exponential multiplication (LB = 1-2 Hz) to enhance S/N before FT. Part B: DEPT-135 Acquisition
- Pulse Program: Select
dept135. - Parameters: Similar to ¹³C, but NS typically 256-1024.
- Processing: Process similarly to ¹³C. CH₃ and CH signals appear positive, CH₂ negative, and quaternary carbons are absent.
Workflow & Logical Pathway Diagrams
Title: Initial NMR Profiling Workflow
Title: NMR Data Informs Targeted 2D Experiments
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Initial NMR Profiling
| Item / Reagent | Function & Critical Specifications | Typical Vendor/Example |
|---|---|---|
| Deuterated NMR Solvents (CDCl₃, DMSO-d₆, CD₃OD) | Provides a deuterium lock signal for the spectrometer; must be >99.8% D, with low water content. | Cambridge Isotope Laboratories (CIL), Eurisotop |
| High-Precision NMR Tubes (5 mm) | Holds sample; quality affects spectral resolution (e.g., coaxiality, wall uniformity). | Norell (Standard Series), Bruker (SampleJet tubes) |
| NMR Tube Caps | Seals tube, prevents solvent evaporation and contamination. | WGS-5BL Cap (for 5mm tubes) |
| Micro-syringe / Capillary Pipettes | For precise transfer of small-volume, expensive deuterated solvents. | Hamilton Company |
| Analytical Balance (µg precision) | Accurately weighs sub-5mg samples for concentration determination. | Mettler Toledo XPR microbalance |
| NMR Processing Software | For phasing, baseline correction, integration, and peak picking. | MestReNova, TopSpin (Bruker), VnmrJ (Varian/Agilent) |
| Chemical Shift Reference Standards | For internal calibration (e.g., TMS, residual solvent peaks). | Tetramethylsilane (TMS) in deuterated solvent |
Application Notes
In the integrated structural elucidation workflow for natural products, High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) provides the molecular formula. The subsequent and most critical step is the deterministic assembly of the carbon-hydrogen framework, which is achieved through a suite of two-dimensional NMR experiments. This phase, termed the "Connectivity Puzzle," leverages through-bond correlations to map proton and carbon networks unambiguously. COSY identifies vicinal and geminal proton couplings, HSQC defines direct one-bond C-H connections, and HMBC reveals long-range couplings (typically 2-3 bonds), crucially linking protonated and non-protonated carbons. Together, they transform a list of NMR chemical shifts into a coherent, atom-by-atom structural map, enabling researchers to differentiate between isomers and establish complex glycosylation patterns or polycyclic systems common in bioactive natural products.
Experimental Protocols
Protocol 1: COSY (Correlation Spectroscopy) Experiment
Purpose: To identify scalar couplings (³JHH, ²JHH) between protons. Method:
- Sample: 5-10 mg of purified natural product dissolved in 0.6 mL of deuterated solvent (e.g., CDCl₃, DMSO-d₆).
- NMR Tube: Use a standard 5 mm NMR tube. Ensure sample is homogeneous and free of particulates.
- Instrument Setup:
- Place sample in a spectrometer (≥ 500 MHz for proton recommended).
- Lock, tune, match, and shim the sample.
- Calibrate the 90° pulse width for ¹H.
- Acquisition Parameters:
- Pulse Sequence: cosygpqf (gradient-selected, double-quantum filtered COSY).
- Spectral Width: 12-15 ppm in both dimensions (F2, F1).
- Data Points: 2K in F2 (acquisition); 256-512 increments in F1.
- Number of Scans: 4-8 per increment.
- Relaxation Delay: 1.0-1.5 seconds.
- Processing: Apply sine-bell or Qsine window functions in both dimensions. Zero-fill to 1K in F1. Perform Fourier transform and phase correction.
Protocol 2: HSQC (Heteronuclear Single Quantum Coherence) Experiment
Purpose: To correlate directly bonded protons and carbons (¹JCH). Method:
- Sample: As per Protocol 1.
- Instrument Setup: Optimize for ¹H and ¹³C channels.
- Acquisition Parameters:
- Pulse Sequence: hsqcedetgpsisp2.2 (phase-sensitive, gradient-selected, decoupled during acquisition).
- Spectral Width:
- F2 (¹H): 12-15 ppm.
- F1 (¹³C): 180-220 ppm.
- Data Points: 2K in F2; 128-256 increments in F1.
- ¹JCH Coupling Constant: Set to ~145 Hz.
- Number of Scans: 8-16 per increment (due to low ¹³C natural abundance).
- Processing: Use linear prediction in F1. Apply window function (e.g., Qsine). Zero-fill and Fourier transform. Set ¹³C referencing using solvent signal.
Protocol 3: HMBC (Heteronuclear Multiple Bond Correlation) Experiment
Purpose: To detect long-range couplings between protons and carbons (²,³JCH, typically 2-8 Hz). Method:
- Sample: As per Protocol 1.
- Instrument Setup: As per Protocol 2.
- Acquisition Parameters:
- Pulse Sequence: hmbcgplpndqf (gradient-selected, low-pass J-filter to suppress ¹JCH signals).
- Spectral Width: As per HSQC.
- Data Points: 2K in F2; 200-256 increments in F1.
- Long-Range Coupling Delay: Optimize for 8 Hz (62.5 ms delay).
- Number of Scans: 16-32 per increment.
- Processing: Similar to HSQC but typically presented in magnitude mode. Critical to adjust threshold to display weak correlations.
The following table summarizes the core information provided by each 2D NMR experiment, essential for solving the connectivity puzzle.
Table 1: Key Parameters and Data from Essential 2D NMR Experiments
| Experiment | Correlation Type | Typical Coupling Constant (J) | Key Information Provided | Optimal Sample Quantity* |
|---|---|---|---|---|
| COSY | ¹H - ¹H | ²JHH: 10-15 Hz; ³JHH: 5-8 Hz | Proton-proton connectivity within 2-3 bonds (spin systems). | 5-10 mg |
| HSQC | ¹H - ¹³C (Direct) | ¹JCH: 110-170 Hz | Direct attachment of protons to specific carbons. Distinguishes CH₃/CH₂/CH from quaternary C. | 2-5 mg |
| HMBC | ¹H - ¹³C (Long-Range) | ²,³JCH: 2-8 Hz | Connectivity over 2-3 bonds. Links protonated carbons to quaternary carbons and functional groups (e.g., C=O). | 5-15 mg |
*Quantities are estimated for a natural product with MW ~500 Da on a 500 MHz spectrometer with a cryoprobe.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for 2D NMR-Based Structure Elucidation
| Item | Function & Importance |
|---|---|
| Deuterated NMR Solvents (e.g., CDCl₃, DMSO-d₆, Methanol-d₄) | Provides a deuterium lock signal for spectrometer stability and minimizes intense solvent signals in the ¹H spectrum. |
| High-Purity NMR Tubes (5 mm, 7" length) | Precision tubes ensure consistent sample spinning and spectral line shape. |
| Micro-weighing Balances (0.01 mg sensitivity) | Accurate quantification of sub-milligram quantities of precious natural product isolates. |
| Cryogenically Cooled Probes (Cryoprobes) | Increases sensitivity by 4x or more, drastically reducing experiment time and sample requirement. |
| Structure Elucidation Software (e.g., MestReNova, ACD/Labs) | Enables processing, visualization, and manual/automated assignment of complex 2D NMR data sets. |
| Reference Compounds (e.g., TMS, residual solvent peaks) | Critical for accurate chemical shift calibration and reporting. |
Visualization: NMR Connectivity Workflow
Diagram 1: 2D NMR-Based Structural Assembly Workflow (82 chars)
Diagram 2: NMR Correlation Map on a 3-Carbon System (74 chars)
Within the comprehensive structure elucidation workflow for natural products—relying on HR-ESIMS for molecular formula and NMR for planar structure—determining relative and absolute configuration remains the final, critical hurdle. Stereochemistry dictates biological activity, making its accurate assignment essential for drug development. This protocol details the integrated application of NMR-based experiments (NOESY/ROESY, J-coupling analysis) and computational chemistry to solve stereochemical challenges.
Core Methodologies and Application Notes
NOESY & ROESY for Through-Space Correlations
Principle: Nuclear Overhauser Effect Spectroscopy (NOESY) and Rotating-frame Overhauser Effect Spectroscopy (ROESY) detect through-space dipolar couplings between nuclei (typically <5 Å). NOESY is ideal for mid-sized molecules, while ROESY is crucial for small molecules where NOE is weak or zero due to fast tumbling.
Protocol: NOESY/ROESY Acquisition
- Sample Preparation: Dissolve 5-20 mg of purified natural product in 0.6 mL of deuterated solvent (e.g., CDCl₃, DMSO-d₆). Filter through a micro-filter if necessary.
- Instrument Setup: Set probe temperature to 298 K. Calibrate 90° pulse widths.
- Parameter Settings:
- Spectral Width: Match to ¹H spectrum.
- Mixing Time (τₘ): Critical for quantitative analysis. Perform a series of experiments with τₘ from 200 ms to 800 ms to establish the initial linear build-up rate. For routine screening, 500 ms is typical.
- Relaxation Delay (d1): ≥ 1.5 s.
- Number of Scans: 8-16 per t1 increment.
- t1 Increments: 256-512 for adequate digital resolution in F1.
- Processing: Apply apodization (sine-bell or QSINE) in both dimensions. Zero-fill to 1K x 1K matrix. Phase correct symmetrically.
J-Coupling Constant Analysis for Dihedral Angles
Principle: The Karplus equation relates vicinal proton-proton coupling constants (³JHH) to their dihedral angle, providing direct conformational information.
Protocol: Measuring ³JHH from 1D or 2D NMR
- High-Resolution 1D ¹H NMR: Acquire spectrum with sufficient digital resolution (<0.5 Hz/pt after zero-filling). Fit multiplets using iterative Lorentzian line-fitting software.
- Phase-Sensitive COSY or TOCSY: Use to identify coupled spin systems and extract coupling constants from cross-peak fine structure in high-resolution acquired spectra (at least 4K points in F2).
- Angle Calculation: Use a modified Karplus equation (e.g., Haasnoot-Leeuw-Altona) to convert measured ³JHH into dihedral angle constraints. Typical values: ³JHH ~ 8-10 Hz for 180° (anti), 0-4 Hz for ~90°, and 2-5 Hz for 60° (gauche).
Computational Conformational Analysis
Principle: Molecular Mechanics (MM) and Density Functional Theory (DFT) calculations generate low-energy conformer ensembles, predict NMR parameters (chemical shift, J-coupling), and calculate theoretical NOE intensities for comparison with experiment.
Protocol: Integrated Computational Workflow
- Conformer Generation: Use software (e.g., CONFLEX, MacroModel, RDKit) to generate a broad set of conformers via systematic or stochastic search (MMFF94s or MM2 force field).
- Geometry Optimization & Re-ranking: Optimize all unique conformers (energy window: 10 kcal/mol) using DFT (e.g., B3LYP/6-31G(d) level). Calculate Boltzmann populations.
- NMR Prediction:
- Chemical Shifts: Calculate isotropic magnetic shielding tensors (GIAO method). Apply linear regression to referenced calculated shielding to predict ¹³C and ¹H shifts. Compare via DP4 probability analysis.
- Spin-Spin Couplings: Calculate reduced coupling constants via DFT, then convert to ³JHH.
- Theoretical NOE/ROE: Using the optimized conformer ensemble and Boltzmann weights, calculate interatomic distances and subsequently back-calculate expected NOE intensities (using the isolated spin pair approximation or complete relaxation matrix approach).
Table 1: Quantitative Comparison of Stereochemical Determination Methods
| Method | Key Parameter Measured | Typical Measurement Range | Information Provided | Key Limitation |
|---|---|---|---|---|
| NOESY | Cross-peak volume (V) | Interatomic distance: 1.8 - 5.0 Å | Through-space proximity, relative configuration | Signal nulling for small MW (~<500 Da) |
| ROESY | Cross-peak volume (V) | Interatomic distance: 1.8 - 4.0 Å | Through-space proximity for all MW sizes | Spin-lock artifacts, offset dependence |
| ³JHH Analysis | Scalar coupling (J) | 0 - 14 Hz | Dihedral angle (via Karplus equation) | Conformational averaging ambiguity |
| DFT Chemical Shift | Calculated δ (ppm) | -- | DP4 probability for configurational assignment | Computationally intensive; solvent effects |
| Calculated ECD | Δε (L·mol⁻¹·cm⁻¹) | UV range (e.g., 200-400 nm) | Absolute configuration (vs. experimental CD) | Requires suitable chromophore |
Integrated Experimental-Computational Workflow
Title: Integrated Stereochemistry Determination Workflow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Computational Tools for Stereochemical Analysis
| Item | Function & Specification | Example/Supplier |
|---|---|---|
| Deuterated NMR Solvents | Provide field-frequency lock and minimize solvent ¹H signal. Must be anhydrous. | Cambridge Isotope Laboratories (CIL): CDCl₃, DMSO-d₆, Methanol-d₄ |
| Chiral Derivatizing Agents | Convert enantiomers to diastereomers for NMR analysis (e.g., MTPA, MPA). | Mosher’s Acid (α-Methoxy-α-trifluoromethylphenylacetic acid), Sigma-Aldrich |
| NMR Tube Filter | Removes particulate matter from sample to improve lineshape. | Shigemi microcells or in-line micro-filters |
| Computational Software (MM) | Performs conformational searching and initial energy minimization. | Schrodinger MacroModel, CONFLEX, Open Babel |
| Quantum Chemistry Suite (DFT) | Performs high-level geometry optimization and NMR parameter calculation. | Gaussian 16, ORCA, NWChem |
| NMR Prediction & DP4 Software | Processes computed data and compares statistically with experiment. | MestReNova, ACD/Labs, SpecInfo, in-house DP4 scripts |
| ECD/ORD Database | Compares experimental chiroptical data for absolute configuration. | SpecDis, BioTools for ECD, ORD spectra matching |
Application Notes
In the integrated workflow for natural product structure elucidation, the correlation of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) fragmentation patterns with Nuclear Magnetic Resonance (NMR)-derived substructures represents a critical validation step. This synergy transforms discrete data points into a coherent structural hypothesis. HR-ESIMS provides precise molecular formulae and fragment ions that suggest connectivity breaks, while NMR (¹H, ¹³C, HSQC, HMBC, COSY) offers definitive proof of atom connectivity and stereochemistry within molecular fragments. The integration platform validates proposed substructures by ensuring that MS-derived fragments logically correspond to NMR-identified spin systems and long-range couplings, significantly reducing ambiguity and accelerating the dereplication and discovery of novel entities.
Table 1: Representative Correlation Data for a Model Natural Product (E.g., Flavonoid Glycoside)
| Data Type | Parameter | Value / Observation | Validation Outcome |
|---|---|---|---|
| HR-ESIMS | Precursor Ion [M+H]+ | m/z 449.1078 (C21H21O11) | Confirms molecular formula. |
| MS/MS Fragmentation | Key Fragment Ions | m/z 287.0556 (Aglycone), 161.0238 (Hexose) | Suggests glycosidic cleavage. |
| ¹H NMR | Anomeric Proton (Sugar) | δ 5.12 ppm (d, J=7.2 Hz) | Confirms β-linked glucose. |
| ¹³C NMR | Anomeric Carbon | δ 100.8 ppm | Correlates with MS sugar fragment. |
| HMBC Correlation | Anomeric H (δ 5.12) to Aglycone C | Correlates to δ 134.5 ppm (Aglycone C-2) | Validates glycosidic linkage site. |
| Integrated Result | Validated Substructure | Flavonoid-O-β-D-glucopyranoside | Hypothesis confirmed. |
Experimental Protocols
Protocol 1: Generation of MS/MS Fragmentation Data for Correlation
Objective: To produce diagnostic fragment ions from a purified natural product for substructure hypothesis generation. Materials: Purified compound, LC-MS/MS system (Q-TOF or Orbitrap). Procedure:
- Sample Preparation: Dissolve 0.1-1.0 mg of purified compound in LC-MS grade methanol to a concentration of ~10 µg/mL.
- LC-MS/MS Setup: Use a C18 column. Employ a gradient elution (e.g., 5-95% acetonitrile in water, 0.1% formic acid over 20 min).
- HR-ESIMS Parameters:
- Ionization Mode: Positive and/or negative ESI.
- Source Voltage: 3.5 kV.
- Capillary Temp: 300°C.
- Scan Range: m/z 100-1500.
- Resolution: >30,000.
- MS/MS Acquisition:
- Select the precursor [M+H]+ or [M-H]- ion with an isolation width of 1-2 m/z.
- Apply stepped normalized collision energy (e.g., 20, 35, 50 eV) to generate a range of fragments.
- Acquire fragments at high resolution (>15,000).
- Data Analysis: Using software (e.g., Xcalibur, MassLynx), list all fragment ions with exact mass. Calculate possible elemental compositions for each. Propose neutral losses and potential substructures.
Protocol 2: NMR Experiments for Substructure Elucidation
Objective: To acquire NMR data for the same sample to define connectivities and validate MS-proposed substructures. Materials: 0.5-2.0 mg purified compound, deuterated solvent (e.g., DMSO-d6, CD3OD), 500 MHz NMR spectrometer. Procedure:
- Sample Preparation: Dissolve compound in 0.5 mL of deuterated solvent. Transfer to a 5 mm NMR tube.
- 1D NMR Acquisition:
- ¹H NMR: Perform 16-64 scans. Reference to residual solvent peak.
- ¹³C NMR (DEPT-135): Perform 1024+ scans to distinguish CH3/CH (positive) from CH2 (negative) carbons.
- 2D NMR Acquisition:
- COSY: Identify ¹H-¹H vicinal coupling networks (spin systems).
- HSQC: Correlate all ¹H nuclei directly bonded to ¹³C nuclei (1JCH).
- HMBC: Acquire with long-range coupling constant (~8 Hz) to detect 2-3 bond ¹H-¹³C correlations, crucial for connecting substructures across heteroatoms or quaternary carbons.
- Data Analysis: Assemble substructures (e.g., sugar units, aromatic rings, isoprene chains) from COSY/HSQC data. Use HMBC to link these substructures, noting key connecting correlations.
Protocol 3: Data Integration and Hypothesis Validation Workflow
Objective: To systematically correlate MS fragments with NMR substructures. Procedure:
- Tabulate Data: Create a table listing key MS fragment ions and their proposed elemental formulae/neutral losses.
- Map NMR Substructures: Draw chemical substructures definitively identified by NMR (e.g., a glucose unit, a methoxyflavone system).
- In Silico Fragmentation: For each NMR-derived substructure, predict expected MS cleavages (e.g., cleavage at glycosidic bond yields aglycone and sugar fragments).
- Cross-Reference: Match observed MS fragments (from Protocol 1) with predicted fragments from NMR substructures.
- Direct Match: An observed fragment's mass matches the exact mass of an NMR substructure (e.g., m/z 161.0238 matches C6H10O5 - dehydrated hexose). Validation achieved.
- Consistency Check: Ensure all major MS fragments can be rationally explained by cleavages within or between the assembled NMR substructures. Fragments that cannot be explained may indicate a need to re-evaluate the structural assembly.
- Final Validation: The combined MS/NMR data must yield a single, consistent molecular structure that satisfies all observations.
Visualizations
Title: Integrated MS/NMR Structure Elucidation Workflow
Title: Correlating a Glycoside MS/MS Pattern with NMR Data
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions & Materials
| Item | Function in Integration Protocol |
|---|---|
| LC-MS Grade Solvents (MeOH, ACN, H2O with 0.1% FA) | Ensure high sensitivity, minimal background, and stable ionization in HR-ESIMS. |
| Deuterated NMR Solvents (DMSO-d6, CD3OD, CDCl3) | Provide locking signal for NMR spectrometer, allow for proper referencing of chemical shifts. |
| High-Purity Silica Gel / C18 Resin | For final purification of natural product prior to integrated analysis to avoid contaminants. |
| NMR Reference Standards (TMS, solvent residual peaks) | Essential for precise calibration of ¹H and ¹³C chemical shift scales. |
| Collision Gas (N2 or Ar) for MS/MS | Inert gas used in collision cell to induce controlled fragmentation of precursor ions. |
| Data Processing Software (e.g., MestReNova, ACD/Labs, Xcalibur) | For processing, analyzing, and visualizing complex NMR and MS datasets side-by-side. |
| Structure Drawing Software (e.g., ChemDraw) | To graphically propose and refine structural hypotheses based on integrated data. |
This document serves as detailed Application Notes and Protocols, framed within a broader thesis on the integrated use of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the structure elucidation of novel natural products. The target compound, "Alkaloid MPA-2024," was isolated from the marine sponge Mycale phyllophila collected from the Celebes Sea. This case study exemplifies a systematic approach to solving complex chemical structures, which is fundamental for researchers, scientists, and drug development professionals in identifying lead compounds with potential bioactivity.
Initial Profiling and Isolation
The crude extract, obtained via methanol dichloromethane (1:1) sonication, showed promising cytotoxicity against non-small cell lung cancer (NSCLC) A549 cells (IC₅₀ 12.5 µg/mL). Bioassay-guided fractionation using vacuum liquid chromatography (VLC) and subsequent semi-preparative HPLC yielded 3.2 mg of a pale-yellow, amorphous solid of Alkaloid MPA-2024.
Table 1: Isolation Scheme and Yields
| Step | Technique | Stationary Phase | Mobile Phase | Fraction | Yield (mg) |
|---|---|---|---|---|---|
| 1 | Extraction | - | MeOH:DCM (1:1) | Crude Extract | 5,200 |
| 2 | VLC | Silica gel | n-Hexane → 100% EtOAc | Active Fr. (F7) | 45 |
| 3 | HPLC | C18 | MeCN:H₂O (0.1% FA), 30:70 → 60:40 | Alkaloid MPA-2024 | 3.2 |
Structural Elucidation Workflow
The structure elucidation followed a convergent methodology integrating spectroscopic data.
Title: Structure Elucidation Workflow for Marine Alkaloid
Key Experimental Protocols
Protocol 4.1: HR-ESIMS Analysis for Molecular Formula Determination
Objective: To obtain the exact mass and isotopic pattern for elemental composition assignment. Method: The sample was dissolved in LC-MS grade methanol to a concentration of ~10 ng/µL.
- The solution was directly infused at 5 µL/min into a Q-TOF mass spectrometer equipped with an ESI source.
- Spectra were acquired in positive ion mode over an m/z range of 100–1200.
- Calibration was performed using sodium formate cluster ions.
- The [M+H]⁺ ion was selected, and the exact mass was processed using software (e.g., MassLynx, Xcalibur) with an elemental composition calculator. Constraints were set: C 0-50, H 0-100, N 0-10, O 0-15, with a mass error tolerance of < 3 ppm. Key Result: Observed [M+H]⁺ at m/z 405.2382.
Table 2: HR-ESIMS Data for Alkaloid MPA-2024
| Ion Species | Observed m/z | Calculated m/z | Δ (ppm) | Molecular Formula | DBE |
|---|---|---|---|---|---|
| [M+H]⁺ | 405.2382 | 405.2384 | -0.5 | C₂₄H₃₃N₂O₃ | 10 |
| [M+Na]⁺ | 427.2201 | 427.2203 | -0.5 | C₂₄H₃₂N₂O₃Na | 10 |
Protocol 4.2: Comprehensive 1D and 2D NMR Spectroscopy
Objective: To establish the carbon skeleton and proton connectivity. Sample Preparation: 3.0 mg of compound was dissolved in 0.6 mL of deuterated methanol (CD₃OD) and transferred to a 5 mm NMR tube. Instrument: 700 MHz NMR spectrometer equipped with a cryoprobe. Acquired Experiments:
- ¹H NMR: 64 scans, spectral width 20 ppm.
- ¹³C NMR (BB-Decoupled): 2048 scans, spectral width 240 ppm.
- DEPT-135: Distinguished CH₃/CH (positive) from CH₂ (negative) carbons.
- COSY: Identified vicinal (³JHH) proton coupling networks.
- HSQC: Correlated directly bonded ¹H-¹³C nuclei (¹JCH).
- HMBC: Detected long-range ¹H-¹³C correlations (²,³JCH), crucial for connecting molecular fragments.
Table 3: Key ¹H and ¹³C NMR Data (CD₃OD, 700 MHz)
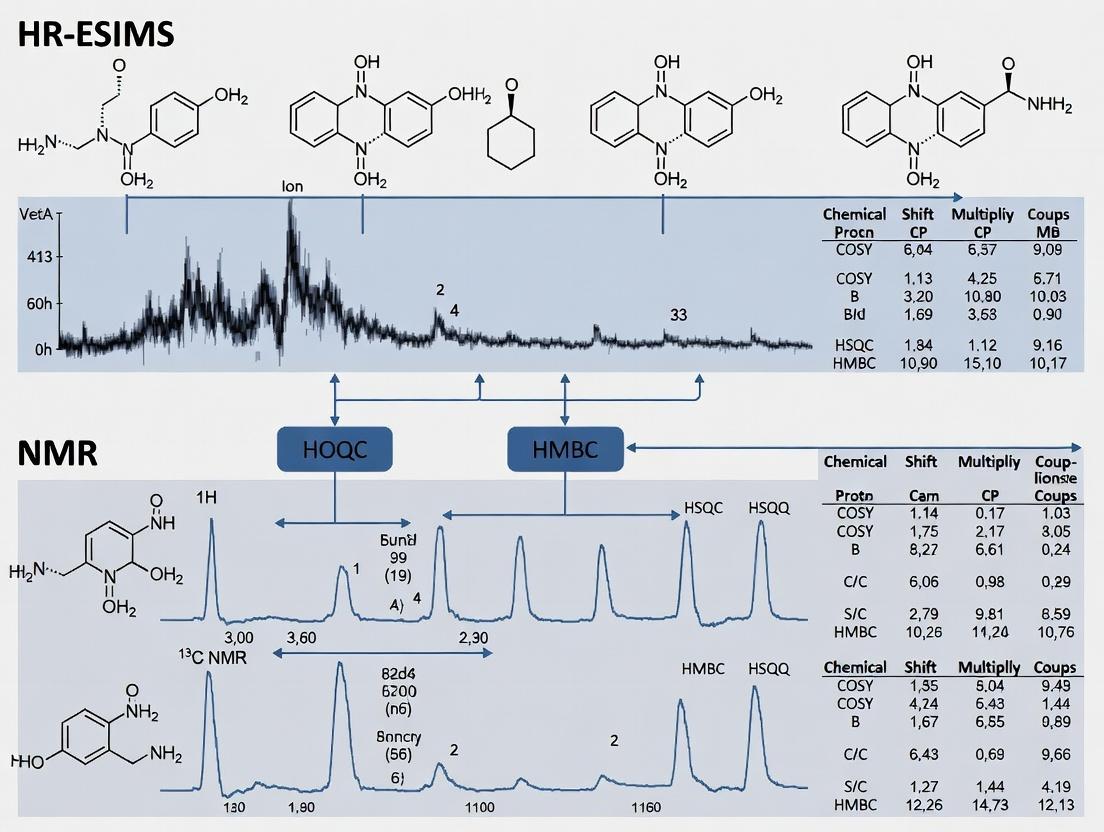
| Position | δC, type (DEPT) | δH, mult. (J in Hz) | Key HMBC Correlations (H→C) |
|---|---|---|---|
| 2 | 165.1, C | - | - |
| 3 | 118.7, CH | 6.89, s | C-2, C-4a, C-8a |
| 4a | 132.5, C | - | - |
| 6 | 48.2, CH₂ | 3.12, m | C-5, C-7, C-8a |
| 7 | 28.5, CH₂ | 2.45, m; 2.35, m | C-5, C-6, C-8, C-8a |
| 8 | 55.1, CH | 3.95, m | C-4a, C-6, C-7 |
| 8a | 78.3, C | - | - |
| N-CH₃ | 42.5, CH₃ | 2.98, s | C-2, C-3, C-8a |
| 1' | 172.5, C | - | - |
| 2' | 35.8, CH₂ | 2.55, t (7.5) | C-1', C-3' |
| 3' | 26.0, CH₂ | 1.65, m | C-1', C-2', C-4' |
Protocol 4.3: Computational DP4+ Probability Analysis
Objective: To statistically evaluate and rank candidate structures generated from spectroscopic data. Method:
- Candidate Generation: Multiple isomeric structures consistent with C₂₄H₃₂N₂O₃ were drawn using ChemDraw.
- Conformational Search: For each candidate, a molecular mechanics (MMFF94) conformational search was performed.
- NMR Calculation: Low-energy conformers (Boltzmann population > 1%) were optimized at the B3LYP/6-31G(d) level, and ¹³C NMR chemical shifts were calculated using the GIAO method with the PCM solvent model for methanol.
- DP4+ Analysis: The calculated chemical shifts for each candidate were compared to the experimental values using a published Python script. The DP4+ probability gives the percent likelihood that each candidate is correct, incorporating both ¹H and ¹³C data uncertainties.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for Marine Natural Product Elucidation
| Item | Function & Brief Explanation |
|---|---|
| Deuterated NMR Solvents (CD₃OD, CDCl₃, DMSO-d₆) | Provides the lock signal for NMR spectrometers and allows for the observation of solute signals without interference from protic solvents. |
| LC-MS Grade Solvents (MeOH, MeCN, H₂O with 0.1% Formic Acid) | Ultra-pure solvents minimize background noise and ion suppression in HR-ESIMS, ensuring accurate mass measurement. |
| Semi-Preparative HPLC Columns (C18, 5µm, 10 x 250 mm) | Essential for the final purification step to isolate milligram quantities of pure compound from complex biological matrices. |
| Cryoprobes (for NMR) | Significantly increases sensitivity (by 4x or more) by cooling the detector coils, enabling data acquisition on sub-milligram samples. |
| Silica Gel & TLC Plates (60 Å, 40-63 µm) | Standard media for normal-phase chromatographic separation and rapid monitoring of fractions. |
| Computational Chemistry Software (Gaussian, ACD/Labs, MestReNova) | Used for quantum mechanical NMR calculations, spectrum prediction, processing, and database management of spectroscopic data. |
Data Integration and Final Structure Assignment
The molecular formula C₂₄H₃₂N₂O₃ (DBE=10) from HR-ESIMS suggested a highly unsaturated system. ¹H NMR indicated signals for an olefinic proton (δH 6.89, s), an N-methyl group (δH 2.98, s), an oxymethine (δH 3.95, m), and aliphatic methylenes. ¹³C and DEPT spectra confirmed 24 carbons: 4 methyls, 5 methylenes, 8 methines (one olefinic, one oxymethine), and 7 quaternary carbons (including one carbonyl at δC 172.5 and one at δC 165.1). The HMBC correlation from the N-CH₃ protons to C-2 (δC 165.1), C-3 (δC 118.7), and C-8a (δC 78.3) established a 1,2-disubstituted pyrrolidine core fused to a lactam. The COSY chain H₂-2'/H₂-3'/H₂-4'... connected to the HMBC correlation from H₂-2' to the carbonyl C-1' (δC 172.5), which in turn showed an HMBC to the oxymethine H-8, linking a fatty acyl chain to the core. DP4+ analysis of three top candidates gave a 98.7% probability for the structure shown below.
Title: Computational Structure Validation Pathway
Final Structure: Alkaloid MPA-2024 was determined to be (8R)-N-methyl-8-(tetradecanoyloxy)-1,2,3,5,6,8a-hexahydropyrrolo[2,1-b]quinazolin-4-one, a new pyrroloquinazoline alkaloid.
Table 5: DP4+ Probability Analysis Results
| Candidate Structure | DP4+ Probability (%) (¹³C + ¹H) | Remarks |
|---|---|---|
| Structure A (Proposed) | 98.7 | All experimental data satisfied; stereochemistry at C-8 assigned by ROESY. |
| Structure B (Isomer) | 1.2 | Poor match for oxymethine and olefinic carbon shifts. |
| Structure C (Isomer) | 0.1 | Inconsistent lactam carbonyl chemical shift. |
Solving the Puzzle: Troubleshooting Common Challenges in HR-ESIMS and NMR Analysis
Application Notes for Natural Product Structure Elucidation
Within the integrated workflow of HR-ESIMS and NMR for natural product research, HR-ESIMS provides critical molecular formula and fragment ion data. However, analytical pitfalls including adduct formation, ion suppression, and poor ionization can compromise data integrity, leading to misidentification or missed detection of key metabolites. This document outlines the causes and provides validated protocols to mitigate these issues.
Adduct Formation
Adducts are non-covalent associations between the analyte ion and other ions/molecules (e.g., Na+, K+, NH4+, formate, acetate). While sometimes useful, they can complicate spectra and obscure the [M+H]+ ion.
Causes:
- Sample preparation using sodium or potassium salts.
- Glassware contamination.
- Mobile phase additives (e.g., ammonium formate/acetate).
- Instrumental carryover.
Solutions & Protocol: Protocol 1.1: Minimizing and Standardizing Adduct Formation
- Reagents: LC-MS grade water and organic modifiers (MeCN, MeOH), Optima-grade formic acid (0.1%), ammonium hydroxide (0.1%), ammonium acetate/ammonium formate (1-10 mM).
- Sample Prep: Avoid alkali metal salts. Use polymeric or low-adsorption plastic vials/tips instead of glass. For solid-phase extraction (SPE), condition with MS-compatible solvents.
- LC Conditions: Use mobile phase additives consistently. For positive mode, 0.1% formic acid promotes [M+H]+. For negative mode, 0.1% ammonium hydroxide or 1-5 mM ammonium acetate promotes [M-H]- or [M+acetate]-.
- Data Acquisition: Acquire in both positive and negative modes to profile all adducts. Use "Find by Formula" or "Adduct Ion Search" algorithms in data processing software (e.g., Bruker DataAnalysis, Thermo Compound Discoverer).
Table 1: Common ESI Adducts and Their Mass Differences
| Adduct Type | Mass Difference (Da) | Typical Polarity | Common Source |
|---|---|---|---|
| [M+H]+ | +1.00728 | Positive | Acidic Modifier |
| [M+Na]+ | +22.98922 | Positive | Glassware, Salts |
| [M+K]+ | +38.96316 | Positive | Salts |
| [M+NH4]+ | +18.03383 | Positive | Ammonium Salts |
| [M-H]- | -1.00728 | Negative | Basic Modifier |
| [M+FA-H]- | +44.99820 | Negative | Formic Acid/Formate |
| [M+Ac-H]- | +59.01385 | Negative | Acetic Acid/Acetate |
Visualization: Common ESI Adduct Formation Pathways
Diagram Title: Common ESI Adduct Formation Pathways
Ion Suppression
Ion suppression results from competition for charge and droplet surface during the ESI process, reducing the ionization efficiency of analytes, particularly in complex natural product extracts.
Causes:
- High concentrations of non-volatile salts (phosphates, sulfates).
- Co-eluting matrix components (polyphenols, carbohydrates, lipids, detergents).
- High concentration of easily ionizable compounds.
Solutions & Protocol: Protocol 2.1: Assessing and Overcoming Ion Suppression
- Sample Cleanup: Employ SPE (C18, HLB, SiOH phases) or liquid-liquid partitioning to remove non-volatiles and interfering matrices.
- Chromatographic Separation: Optimize LC methods to separate analytes from matrix interferences. Use longer gradients, different stationary phases (e.g., HILIC for polar compounds), or UHPLC for higher resolution.
- Post-column Infusion Test: To map suppression zones.
- A. Prepare a standard solution of a target analyte (e.g., 1 µg/mL in MeOH).
- B. Infuse this solution post-column via a T-union at a constant rate (e.g., 5 µL/min) using a syringe pump.
- C. Inject the sample matrix (e.g., crude extract) and run the LC-MS method.
- D. Monitor the ion trace of the infused standard. A drop in signal indicates a suppression region in the chromatogram.
- Internal Standards: Use stable isotope-labeled internal standards (SIL-IS) for each analyte. If unavailable, use structural analogs added post-extraction to correct for suppression.
Table 2: Ion Suppression Mitigation Strategies
| Strategy | Method | Key Benefit | Limitation |
|---|---|---|---|
| Sample Cleanup | SPE, LLE | Removes non-volatile salts, lipids | May lose some analytes |
| Chromatography | UHPLC, HILIC, Longer Gradients | Separates analyte from suppressor | Increased run time |
| Dilution | Pre-injection Dilution | Reduces matrix concentration | May drop analyte below LOD |
| Standardization | SIL Internal Standards | Precisely corrects for suppression | Expensive, not always available |
Visualization: Ion Suppression Mechanism and Mitigation Workflow
Diagram Title: Ion Suppression Mechanism and Mitigation Workflow
Poor Ionization
Some natural products (e.g., non-polar terpenes, carotenoids, sterols) ionize inefficiently by standard ESI, leading to weak or absent signals.
Causes:
- Low proton affinity or low electron affinity.
- Lack of readily ionizable functional groups.
- Instability in charged form.
Solutions & Protocol: Protocol 3.1: Enhancing Ionization for "Hard-to-Ionize" Compounds
- Derivatization: Introduce an easily ionizable moiety.
- For Positive Mode: React with Girard's Reagent T (introduces quaternary ammonium group) or dansyl chloride.
- For Negative Mode: Derivatize with hydroxylamine to form oximes.
- Alternative Ionization Sources: Use Atmospheric Pressure Chemical Ionization (APCI) or Atmospheric Pressure Photoionization (APPI). APCI is superior for less polar, thermally stable compounds. APPI is ideal for aromatic and polyaromatic compounds.
- Adduct-Enhanced Ionization: Deliberately use additives that form stable adducts.
- For Saponins/Sugars: Add LiCl to form [M+Li]+ adducts, which provide informative fragmentation.
- For Non-polar Compounds: Add AgNO3 to form [M+Ag]+ adducts with alkenes/alkynes.
- Infusion Solvent Optimization: Increase solvent volatility (use MeCN > MeOH) and add dopants (e.g., 0.01% toluene for APPI).
Table 3: Ionization Enhancement Techniques
| Technique | Typical Application | Mechanism | Recommended Additive/Reagent |
|---|---|---|---|
| Derivatization | Terpenes, Sterols, Alcohols | Introduces permanent charge | Girard's Reagent T, Dansyl chloride |
| APCI | Non-polar, Thermally Stable Lipids | Gas-phase chemical ionization | Standard volatile buffers |
| APPI | Aromatic Compounds, Carotenoids | Photon-induced ionization | Toluene (dopant) |
| Metal Adduction | Sugars, Alkenes, Dienes | Stable metal ion coordination | LiCl, AgNO₃, NaI |
The Scientist's Toolkit: Key Reagent Solutions
| Reagent/ Material | Function in HR-ESIMS Pitfall Mitigation |
|---|---|
| Ammonium Formate (Optima grade) | Volatile buffer for mobile phase; promotes [M+H]+/[M-H]- or formate adducts. |
| Formic Acid (LC-MS grade) | Common acidic modifier to promote protonation in positive mode. |
| SPE Cartridges (C18, HLB) | For sample cleanup to remove ion-suppressing salts and matrix. |
| Stable Isotope-Labeled Internal Standards (SIL-IS) | Gold standard for correcting ion suppression and matrix effects quantitatively. |
| Girard's Reagent T | Derivatizing agent to introduce a charged quaternary ammonium group into non-polar ketones/aldehydes. |
| Lithium Chloride (LiCl) | Additive to promote stable [M+Li]+ adducts for enhanced ionization and informative fragmentation of glycosides. |
| Post-column Infusion T-union & Syringe Pump | Hardware for performing the ion suppression assessment experiment. |
| APCI or APPI Source | Alternative ionization source for compounds with poor ESI efficiency. |
Integrated Troubleshooting Protocol
Protocol 4.1: Systematic HR-ESIMS Analysis of a Novel Natural Product Extract
- Initial Profiling: Analyze under standard conditions (e.g., C18 column, water/MeCN + 0.1% formic acid gradient, ± polarity).
- Adduct Identification: Use software to annotate all peaks with possible adducts and neutral losses. Check for consistency across samples.
- Suppression Check: Perform post-column infusion test (Protocol 2.1) if signal is low or erratic. Apply cleanup or chromatographic optimization.
- Ionization Assessment: If target compounds (known from NMR or bioassay) are not observed, switch to APCI/APPI or implement derivatization (Protocol 3.1).
- Data Integration: Cross-reference clean HR-ESIMS molecular formula data with 1D/2D NMR data for confident structure elucidation.
By systematically addressing these pitfalls, the complementary power of HR-ESIMS and NMR in the structural analysis pipeline is fully realized, leading to more confident and efficient characterization of novel natural products.
In the broader thesis on utilizing High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) for natural product structure elucidation, NMR spectroscopy is indispensable for defining carbon frameworks and stereochemistry. However, key obstacles—signal overlap, dynamic exchange, and interfering solvent/impurity peaks—routinely impede unambiguous analysis. This document provides targeted application notes and protocols to overcome these challenges, thereby accelerating drug discovery from natural sources.
Addressing Signal Overlap
Signal overlap in 1D 1H-NMR spectra obscures coupling constants and integration, critical for structural assignment.
Quantitative Summary of 2D NMR Techniques for Resolving Overlap
| 2D Technique | Nuclei Correlated | Primary Utility in Resolving Overlap | Typical Experiment Time (at 600 MHz) |
|---|---|---|---|
| COSY | 1H-1H | Identifies scalar-coupled networks | 5-15 minutes |
| TOCSY | 1H-1H | Reveals entire spin systems | 15-45 minutes |
| HSQC | 1H-13C | Directly pairs H with its bonded C | 30-60 minutes |
| HMBC | 1H-13C | Correlates H to long-range C (2-3 bonds) | 1-2 hours |
| 1H-15N HSQC | 1H-15N | Key for alkaloids, peptides | 2-4 hours |
Protocol 1.1: Acquiring a Phase-Sensitive HSQC for Optimal Resolution Objective: Obtain high-resolution 1H-13C heteronuclear single quantum coherence data. Materials: ~10-20 mg of purified natural product in 0.6 mL deuterated solvent (e.g., DMSO-d6, CD3OD).
- Sample Preparation: Dissolve sample. Ensure sample is free of particulate matter by centrifugation.
- NMR Setup: Load sample into spectrometer (≥500 MHz recommended). Lock, shim, and tune/probe.
- Parameter Setup:
- Pulse Program:
hsqcetgpsisp2.2(Bruker) or equivalent phase-sensitive, sensitivity-enhanced sequence. - Spectral Width (F2, 1H): 12-15 ppm centered on residual solvent peak.
- Spectral Width (F1, 13C): 220 ppm (for full chemical shift range).
- TD (F2): 2048; TD (F1): 256.
- Scans per Increment: 4-8.
- D1 (Relaxation Delay): 1.0-1.5 s.
- Set non-uniform sampling (NUS) to 50% for time savings if available.
- Pulse Program:
- Acquisition: Run experiment.
- Processing: Apply sine-bell window functions in both dimensions. Zero-filling in F1 to 1024. Fourier transform and phase correct.
Diagram: 2D NMR Strategy for Overlap Resolution
Title: 2D NMR Strategy for Overlap Resolution
Managing Dynamic Exchange
Conformational dynamics and proton exchange can lead to signal broadening or disappearance.
Protocol 2.1: Variable Temperature (VT) NMR to Probe Exchange Objective: Characterize dynamic processes by monitoring chemical shift and linewidth changes with temperature.
- Calibration: Use a standard (e.g., 80% ethylene glycol in DMSO-d6) to calibrate the VT unit’s temperature accuracy.
- Initial Acquisition: Acquire a standard 1H spectrum at 298 K.
- VT Series: Acquire 1H spectra across a temperature range (e.g., from 278 K to 318 K in 5-10 K increments).
- Analysis: Plot chemical shift (δ) vs. Temperature (K) for exchanging protons. A linear change suggests fast exchange; coalescence and separation indicate intermediate exchange. Calculate exchange rates and free energy barriers using the Gutowsky-Holm equation.
Protocol 2.2: Solvent Exchange for Labile Protons Objective: Identify exchangeable protons (e.g., OH, NH).
- Dry Sample Acquisition: Acquire a 1H spectrum of the sample in a dry, aprotic deuterated solvent (e.g., CDCl3, DMSO-d6).
- Solvent Addition: Add 1-2 drops of D2O directly to the NMR tube, vortex gently.
- Re-acquisition: Acquire a new 1H spectrum immediately.
- Analysis: Compare spectra. Signals from exchangeable protons (OH, NH, COOH) will be significantly reduced or absent in the D2O-shaken spectrum.
Diagram: Dynamic Exchange Analysis Workflow
Title: Dynamic Exchange Analysis Workflow
Suppressing Solvent and Impurity Peaks
Residual solvent and impurity signals can overwhelm key analyte signals.
Quantitative Efficacy of Solvent Suppression Techniques
| Technique | Principle | Best For | Key Limitation |
|---|---|---|---|
| Presaturation | Continuous weak RF at solvent frequency | Stable, simple solvents (H2O, CHCl3) | Irreversible saturation, nearby signals affected |
| Excitation Sculpting (e.g., WATERGATE) | Gradient-tailored bipolar pulse pairs | Excellent for H2O suppression in aqueous samples | Limited bandwidth; may suppress analyte signals with similar δ |
| WET | Combined shaped pulses and gradients | Simultaneous suppression of multiple solvents (e.g., CH3OH & H2O) | Requires precise pulse calibration |
Protocol 3.1: WATERGATE for Aqueous Samples Objective: Acquire a high-quality 1H NMR spectrum of a natural product in H2O/D2O mixture without the water peak. Materials: Sample in 90% H2O/10% D2O or D2O with residual H2O.
- Pulse Program: Select
zgesgp(Bruker) or equivalent 1D sequence with WATERGATE suppression. - Parameter Setup:
- Set the transmitter offset (O1P) to the exact frequency of the water signal (e.g., ~4.7 ppm).
- Spectral Width: 20 ppm.
- TD: 65536 points for high digital resolution.
- NS: 128.
- D1: 2.0 s.
- Acquisition & Processing: Run and process normally.
The Scientist's Toolkit: Key Reagents & Materials
| Item | Function & Rationale |
|---|---|
| Deuterated Solvents (DMSO-d6, CDCl3, CD3OD) | Provides lock signal and minimizes large 1H solvent signals. Choice affects compound solubility and chemical shift. |
| NMR Tube (5 mm, 7-inch) | High-quality, matched tubes ensure optimal shimming and spectral line shape. |
| Shift Reagents (e.g., Eu(fod)3) | Paramagnetic lanthanide complexes induce predictable chemical shift changes to resolve overlap. |
| D2O (99.9% D) | For solvent exchange experiments to identify labile protons. |
| Non-Uniform Sampling (NUS) Software | Dramatically reduces experiment time for 2D/3D NMR while maintaining resolution. |
| Spectral Database (e.g., AntiBase, Mnova) | For rapid comparison of experimental NMR data with known natural products. |
Diagram: Solvent/Impurity Suppression Decision Tree
Title: Solvent Suppression Decision Tree
Application Notes and Protocols
Within the broader thesis on integrating High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) for natural product structure elucidation, the most critical challenge is the analysis of sub-milligram, often microgram, quantities of pure compound. This document details optimized protocols for such sample-limited scenarios, leveraging cryogenic probe technology.
Table 1: Optimized NMR Parameters for Microgram Quantities using Cryoprobes
| Parameter | Conventional Probe (5 mm, 50-100 µg) | Cryoprobe (1.7-3 mm, 1-10 µg) | Rationale |
|---|---|---|---|
| Sample Volume | 500-600 µL | 30-120 µL | Minimizes dilution, matches active volume of microcoil/cryoprobe. |
| Concentration | 1-10 mM | 0.1-1 mM (for 10 µg) | Directly enables data acquisition on <10 µg samples. |
| Experiment Time (¹H-13C HSQC) | 30-60 min | 5-15 min | Enhanced signal-to-noise (S/N) from cryogenically cooled electronics reduces time 4-5 fold. |
| Number of Scans (¹³C NMR) | 1024-2048 | 256-512 | Cryoprobe S/N gain of ~4x allows for reduced scans, preserving sample. |
| Spectral Width (¹H, ppm) | Reduced to 8-10 ppm | Reduced to 8-10 ppm | Limits acquisition time, reduces noise. |
| Temperature | 298 K | 298 K (or lower) | Lower temperature can increase viscosity & reduce tumbling, benefiting NOE/ROE. |
Table 2: Optimized HR-ESIMS Parameters for Trace Natural Products
| Parameter | Standard Setting | Microgram-Optimized Setting | Purpose |
|---|---|---|---|
| Sample Injection | Loop injection, ~10 µL | Direct infusion via syringe pump at 1-3 µL/min | Prevents loss on loop walls, ensures stable signal. |
| Cone Voltage | 30-50 V | 20-40 V | Reduced to minimize in-source fragmentation of precious sample. |
| Desolvation Temp | 250-400°C | 150-250°C | Lower temperature for thermally labile natural products. |
| Acquisition Time | 1-2 min | 3-5 min | Longer time averaging for low-intensity signals from trace amounts. |
| Collision Energy (MS/MS) | Ramped (15-45 eV) | Stepped, starting at 10 eV | Fine-tuned to obtain fragment ions from limited precursor ions. |
Detailed Experimental Protocols
Protocol A: Integrated Sample Handling for HR-ESIMS and NMR
- Sample Preparation: Dissolve the purified microgram sample (5-50 µg) in a minimum volume (10-30 µL) of deuterated NMR solvent (e.g., CD₃OD, DMSO-d₆).
- NMR Analysis First: Transfer the entire sample to a 1.7 mm or 3 mm NMR tube using a micro-volume syringe. Acquire quick ¹D ¹H NMR and non-uniformly sampled (NUS) ¹H-13C gHSQC.
- Sample Recovery: Using a syringe, carefully withdraw the NMR sample from the tube and transfer it to a low-volume, conical HPLC vial insert.
- HR-ESIMS Analysis: Dilute the recovered sample with a minimal volume of LC-MS grade methanol (1:1) to ~60 µL. Use a syringe pump for direct infusion. Acquire full-scan HRMS followed by data-dependent MS/MS.
Protocol B: Optimized ¹H-13C gHSQC on a Cryoprobe (3 mm)
- Instrument: NMR spectrometer equipped with a triple-resonance (¹H, ¹³C, ¹⁵N) cryoprobe.
- Pulse Sequence: gHSQC with adiabatic pulses for ¹³C inversion.
- Parameters:
- Spectral Width (¹H): 10 ppm (centered on solvent)
- Spectral Width (¹³C): 165 ppm (220 to 55 ppm)
- Number of Complex Points (t₂): 1024
- Number of Increments (t₁): 128 (Use NUS to acquire 25-30%)
- Scans per Increment: 4-8
- Recovery Delay (d1): 1.2 s
- Total Experimental Time: ~12 minutes (with NUS).
Protocol C: HR-ESIMS for Microgram Samples via Direct Infusion
- Instrument: Q-TOF or Orbitrap mass spectrometer with ESI source.
- Sample Introduction: Hamilton syringe (25 µL) mounted on syringe pump.
- Flow Rate: 2 µL/min.
- Source Conditions:
- Capillary Voltage: 2.8 kV (positive) / 2.5 kV (negative)
- Source Temperature: 100°C
- Desolvation Temperature: 150°C
- Cone Voltage: 25 V
- Desolvation Gas Flow: 300 L/hr
- Acquisition: Profile mode, 0.5 sec scan time, 50-1500 m/z range for 3 minutes.
Visualization of Workflows and Relationships
Workflow for Microgram Sample Analysis
Technologies Overcoming Sample Limitation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Microgram-Scale Structure Elucidation
| Item | Function & Importance |
|---|---|
| 1.7 mm or 3 mm NMR Tubes | Matches the active volume of cryoprobes and microcoils, preventing signal dilution. |
| Deuterated Solvents (99.8% D+) | Highest isotopic purity reduces interfering ¹H signals, critical for low-concentration samples. |
| Micro-Syringes (e.g., 10, 25 µL) | For precise, quantitative transfer and recovery of precious sample solutions without hold-up. |
| Conical HPLC Vial Inserts (e.g., 100 µL) | Allows sample recovery from NMR tube and efficient transfer to MS via syringe pump. |
| Syringe Pump | Enables stable, low-flow direct infusion for HR-ESIMS, maximizing data quality from trace samples. |
| Non-Uniform Sampling (NUS) Software | Reduces NMR experiment time by 2-4x while maintaining resolution, preserving sample stability. |
The elucidation of novel bioactive compounds from complex natural product extracts presents a formidable analytical challenge. This document, framed within a broader thesis on HR-ESIMS and NMR for natural product structure elucidation, details integrated Application Notes and Protocols. The synergistic combination of Liquid Chromatography-High Resolution Mass Spectrometry-Mass Spectrometry (LC-HRMS-MS) and Nuclear Magnetic Resonance (NMR) spectroscopy forms the cornerstone of modern dereplication and discovery pipelines. These protocols are designed to efficiently separate, detect, tentatively identify, and confirm the structures of metabolites, accelerating the drug development process by minimizing redundant rediscovery of known compounds.
Application Notes
Integrated LC-HRMS-MS and NMR Dereplication Workflow
The sequential application of LC-HRMS-MS and NMR maximizes throughput and certainty. LC-HRMS-MS provides rapid, sensitive separation and fragmentation data for tentative identification against databases. Subsequent, targeted NMR analysis on isolated or partially purified fractions delivers definitive structural confirmation, including stereochemistry and substitution patterns unreachable by MS alone.
Key Quantitative Parameters for Instrumentation
Optimal performance relies on calibrated instrumentation. The following table summarizes critical specifications.
Table 1: Key Instrument Performance Parameters for Dereplication
| Instrument | Parameter | Target Specification | Purpose in Dereplication |
|---|---|---|---|
| LC-HRMS-MS | Mass Accuracy | < 2 ppm (internally calibrated) | Exact mass for molecular formula determination. |
| LC-HRMS-MS | Resolution (FWHM) | > 60,000 at m/z 200 | Separation of isobaric ions and isotopic fine structure. |
| LC-HRMS-MS | MS/MS Scan Speed | > 12 Hz (DDA mode) | Adequate data points across narrow LC peaks. |
| NMR | Magnetic Field Strength | ≥ 600 MHz (for 1H) | Enhanced sensitivity and dispersion for complex mixtures. |
| NMR | Cryoprobe Technology | Required | Signal-to-noise increase for limited samples. |
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Reagents
| Item | Function/Application |
|---|---|
| UHPLC-grade Solvents (MeCN, MeOH, Water) | Provide high-purity, low-UV-absorbance mobile phases for optimal LC-MS separation and sensitivity. |
| Formic Acid / Ammonium Formate | Common volatile modifiers for mobile phases to control ionization efficiency in ESI positive/negative modes. |
| Deuterated NMR Solvents (DMSO-d6, CD3OD, D2O) | Provide a locking signal for the NMR spectrometer and minimize interfering solvent signals in 1H spectra. |
| Solid Phase Extraction (SPE) Cartridges (C18, Diol) | For rapid fraction clean-up or pre-fractionation prior to targeted NMR analysis. |
| Database Subscriptions (e.g., AntiBase, GNPS, Reaxys) | Essential for comparing experimental HRMS and MS/MS data with known compound libraries. |
| Quantitative NMR (qNMR) Standard (e.g., 1,4-Bis(trimethylsilyl)benzene) | Enables accurate concentration determination of isolated compounds directly in NMR tube. |
Experimental Protocols
Protocol A: LC-HRMS-MS Data Acquisition for Crude Extracts
Objective: To generate high-quality HRMS and data-dependent MS/MS spectra for all major and minor components in a complex natural product extract.
Materials: Crude extract dissolved in appropriate LC-MS compatible solvent (e.g., 1 mg/mL in MeOH), UHPLC system coupled to Q-TOF or Orbitrap mass spectrometer.
Method:
- Chromatography:
- Column: Reversed-phase C18 column (e.g., 2.1 x 100 mm, 1.7 µm).
- Gradient: 5% to 100% MeCN in H2O (both with 0.1% formic acid) over 18 min, hold at 100% for 3 min.
- Flow Rate: 0.4 mL/min.
- Injection Volume: 2-5 µL.
- HRMS Parameters (ESI Positive/Negative Mode):
- Mass Range: m/z 100-1500.
- Resolution: > 60,000.
- Spray Voltage: ±3.5 kV.
- Capillary Temp: 320°C.
- Sheath/Aux Gas: Optimize for stable spray.
- Data-Dependent MS/MS Acquisition:
- Top-N: Isolate and fragment top 5 most intense ions per cycle.
- Isolation Window: 1.2 m/z.
- Fragmentation: HCD or CID at normalized collision energies stepped (e.g., 20, 40, 60 eV).
- Dynamic Exclusion: 15 s to ensure coverage of co-eluting minor ions.
- Data Processing:
- Use vendor software (e.g., Compound Discoverer, MZmine) to deconvolute peaks, align features, and generate lists of [M+H]+/[M-H]- ions with associated MS/MS spectra.
- Export .mgf or .msp files for database searching.
Protocol B: NMR-Based Dereplication of Pre-fractionated Material
Objective: To obtain structural confirmation and identify novel scaffolds in active fractions following LC-HRMS-MS screening.
Materials: Semi-purified fraction (50-200 µg), deuterated solvent (e.g., CD3OD), 1.7 mm or 3 mm NMR tube.
Method:
- Sample Preparation:
- Transfer dried fraction to a microtube.
- Dissolve in 30-60 µL of deuterated solvent.
- Transfer to a 1.7 mm or 3 mm NMR tube using a micro-syringe.
- 1D ¹H NMR Acquisition:
- Probe: Cryogenically cooled inverse detection probe.
- Pulse Sequence: Standard ¹H experiment with water suppression (e.g., zgpr).
- Parameters: Spectral width 20 ppm, center 8 ppm, 128-256 transients, acquisition time ~2 sec.
- Process with 0.3 Hz line broadening.
- 2D NMR Acquisition (for key fractions):
- 1H-1H COSY: Identifies scalar coupling networks (vicinal protons).
- 1H-13C HSQC: Identifies direct ¹H-13C correlations (protonated carbons).
- 1H-13C HMBC: Identifies long-range ¹H-13C correlations (2-3 bonds), crucial for assembling fragments and quaternary carbons.
- Data Analysis and Dereplication:
- Compare acquired ¹H NMR chemical shifts, coupling constants, and 2D patterns with in-house or commercial NMR databases (e.g., HiFSA-based).
- Integrate with HRMS-derived molecular formula to narrow candidate structures.
- Note: For fractions of pure compounds (>95%), full structure elucidation via extended 2D NMR (ADEQUATE, ROESY) should be performed.
Visualizations
Integrated Dereplication Workflow
LC-HRMS-MS Data Acquisition Logic
Within the broader thesis of employing High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the structure elucidation of novel natural products, the integration of specialized software tools is indispensable. The increasing complexity of isolated compounds and the sheer volume of data necessitate automated approaches to enhance accuracy, accelerate discovery, and reduce researcher bias. This application note details protocols for leveraging computational tools in the critical steps of molecular formula determination from HR-ESIMS data and for the prediction/verification of NMR spectra.
Application Note: Automated Formula Prediction from HR-ESIMS Data
The accurate determination of the molecular formula is the foundational step in structure elucidation. HR-ESIMS provides the exact mass of the molecular ion ([M+H]⁺, [M+Na]⁺, [M-H]⁻, etc.). Software tools use this exact mass, isotopic fidelity, and heuristic rules to generate a ranked list of candidate formulas.
Key Software Tools & Capabilities (Current as of 2024)
| Software/Tool | Primary Function | Key Algorithm/Feature | Typical Input | Output |
|---|---|---|---|---|
| Bruker DataAnalysis | Formula generation for LC-MS data | SmartFormula algorithm using mass accuracy, isotope pattern (Sigma), and MS/MS information | Exact mass, ion type, potential elements & limits | Ranked list of molecular formulas with score & isotope fit |
| Thermo Fisher Compound Discoverer | Untargeted analysis & formula prediction | Integrated formula prediction node using accurate mass and isotopic pattern | .raw file, mass tolerance, elemental composition settings | Annotated features with proposed formulas |
| mzMine 3 (Open Source) | Feature detection & formula prediction | Customizable pipeline with >5 prediction modules (e.g., SIRIUS, Chemistry Development Kit) | LC-MS data file, mass lists | Aligned features with formula assignments |
| SIRIUS 5 (Standalone) | Molecular formula identification (MFI) | Combines isotope pattern analysis with fragmentation tree-based scoring (CSI:FingerID) | MS1 and MS/MS data, mass accuracy | Top-ranked formula candidates with confidence scores |
Detailed Protocol: Molecular Formula Determination Using SIRIUS
Objective: To determine the most probable molecular formula for a novel natural product from HR-ESIMS data.
Materials & Reagents:
- Purified natural product sample (≥ 95% purity by LC-MS)
- Appropriate MS-grade solvent (MeOH, ACN, H₂O)
- HR-ESIMS instrument (e.g., Q-TOF, Orbitrap)
Procedure:
- Sample Preparation & Data Acquisition:
- Dissolve the purified compound to ~1 µg/µL in suitable solvent.
- Acquire high-resolution mass spectra in both positive and negative ionization modes.
- Record MS/MS spectra at multiple collision energies to obtain fragmentation data.
- Ensure mass accuracy is calibrated to within 3 ppm.
Data Preprocessing:
- Convert raw instrument files (.d, .raw) to open formats (.mzML, .mzXML) using tools like MSConvert (ProteoWizard).
- Extract the exact m/z value for the [M+H]⁺ or [M-H]⁻ ion and its isotopic peak intensities.
SIRIUS Analysis:
- Launch SIRIUS and create a new project.
- Import the .mzML file containing the MS1 and MS/MS data for your compound.
- Parameter Settings:
- Mass Accuracy: Set to 3.0 ppm (or your instrument's validated accuracy).
- Ionization Mode: Select [M+H]⁺ or [M-H]⁻ as appropriate.
- Candidate Elements: Define the search space: C (0-100), H (0-200), O (0-50), N (0-20). Add common natural product elements like S, P, Cl, Na, K as needed based on context.
- Filters: Apply typical heuristic rules (e.g., N rule, HC ratio, element ratio checks).
- Execute the computation. SIRIUS will calculate and rank formula candidates based on isotopic pattern score (using the Bayesian approach) and the fragmentation tree score from MS/MS data.
Result Interpretation:
- The candidate with the highest SIRIUS score (sum of isotope and tree score) is the top prediction.
- Cross-verify the top-ranked formula by checking its isotopic pattern fit (the simulated vs. measured pattern).
- Confirm plausibility within the context of known natural product chemistry.
Application Note: NMR Prediction and Verification
Once a molecular formula is established, candidate structures are proposed. NMR prediction software calculates the expected chemical shifts and coupling constants for a given structure, enabling direct comparison with experimental data for verification or for guiding total synthesis efforts.
Key Software Tools & Capabilities
| Software/Tool | Core Function | Prediction Method | Typical Input | Output |
|---|---|---|---|---|
| ACD/Labs NMR Suite | ¹H, ¹³C, multi-NMR prediction & verification | HOSE codes, incremental, and neural network algorithms | Chemical structure (drawing or SMILES) | Predicted chemical shifts, coupling constants, spectra simulation |
| MestReNova (Mnova) | NMR processing, prediction, and verification | Combined algorithm (HOSE, neural networks, DFT interface) | Processed FID and chemical structure | Overlay of predicted vs. experimental spectra, DP4-like probability |
| Gaussian 16 (w/ NMR) | First-principles shift calculation | Density Functional Theory (DFT) – GIAO method | Optimized 3D molecular geometry | Quantum-mechanically calculated absolute shieldings converted to shifts |
| ChemDraw Professional | Basic chemical shift prediction | Incremental and HOSE code algorithms | Drawn 2D structure | Estimated ¹H and ¹³C chemical shifts |
Detailed Protocol: Structure Verification Using ACD/Labs NMR Predictors
Objective: To verify a proposed chemical structure by comparing its software-predicted NMR spectrum with the experimentally acquired spectrum.
Materials & Reagents:
- Proposed chemical structure (e.g., as a *.mol file)
- Experimentally acquired ¹H and ¹³C NMR spectra (processed)
- NMR solvent used (e.g., CDCl₃, DMSO-d6)
Procedure:
- Structure Preparation:
- Draw the proposed candidate structure in ACD/ChemSketch or import a .mol file.
- Ensure stereochemistry is correctly assigned if known.
Prediction Parameter Setup:
- In the ACD/NMR Predictor module, select the nuclei for prediction (¹H and ¹³C).
- Select Prediction Algorithm:
- For rapid verification, use the "HOSE code" and "Incremental" methods.
- For higher accuracy, especially with novel scaffolds, employ the "Neural Network" algorithm (requires training set selection).
- Specify the NMR solvent (e.g., CDCl₃) to account for solvent effects.
- Define the output format (peak list, simulated spectrum).
Execution and Data Generation:
- Run the prediction. The software will generate a table of predicted chemical shifts (δ in ppm), multiplicities, and coupling constants (J in Hz).
- Optionally, generate a simulated NMR spectrum from this data.
Verification and Analysis:
- Import your experimental spectrum into the software.
- Use the "NMR Processor/Verifier" tool to overlay the predicted and experimental spectra.
- Critical Analysis Steps:
- Check for global agreement in the chemical shift range.
- Compare the pattern of signals (multiplicity, integration) in key regions (e.g., aromatic, olefinic, aliphatic).
- Calculate the mean absolute error (MAE) between predicted and experimental shifts. An MAE < 0.3 ppm for ¹H and < 5 ppm for ¹³C generally indicates a good match, but context is critical.
- Systematically investigate any significant outliers (>0.5 ppm for ¹H, >10 ppm for ¹³C) – they may indicate incorrect stereochemistry or regiochemistry.
The Scientist's Toolkit: Research Reagent & Software Solutions
| Item | Category | Function & Relevance |
|---|---|---|
| HR-ESIMS System (e.g., Orbitrap Exploris) | Instrument | Provides high-mass-accuracy (<3 ppm) and high-resolution (>60,000) data essential for formula prediction. |
| NMR Spectrometer (400 MHz+) | Instrument | Acquires ¹H, ¹³C, and 2D NMR data for structural verification against predictions. |
| Deuterated NMR Solvents (CDCl₃, DMSO-d₆) | Reagent | Provides a stable, deuterated lock signal for NMR experiments and influences predicted chemical shifts. |
| SIRIUS 5 Software | Software | Integrates isotope pattern and MS/MS fragmentation tree analysis for superior formula prediction confidence. |
| ACD/Labs NMR Suite | Software | Industry-standard for empirical and neural network-based NMR prediction and spectrum verification. |
| Gaussian 16 with GIAO | Software | Performs quantum mechanical DFT calculations for the most accurate NMR chemical shift predictions of complex or novel scaffolds. |
| mzMine 3 | Software | Open-source platform for preprocessing high-throughput MS data before formula prediction in downstream tools. |
Visualized Workflows
Software-Aided Structure Elucidation Workflow
NMR Prediction Algorithm Trade-offs
Best Practices for Data Acquisition and Processing to Maximize Information Yield
Within the context of natural product structure elucidation using High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy, maximizing information yield is paramount. This Application Note details integrated protocols for data acquisition and processing, ensuring efficient translation of raw analytical data into unambiguous chemical structures. The systematic approach minimizes resource waste and accelerates the drug discovery pipeline.
Application Notes
Strategic Sample Preparation & Prioritization
Effective data acquisition begins prior to instrument analysis. A tiered approach prioritizes samples based on preliminary bioactivity or chemotaxonomic data, ensuring that high-value samples receive comprehensive analytical resources. For natural product extracts, prefractionation via solid-phase extraction or low-resolution LC-MS is recommended to reduce complexity before advanced analysis.
Synergistic HR-ESIMS and NMR Data Acquisition
HR-ESIMS provides precise molecular formula and fragment ion data, while NMR offers definitive connectivity and stereochemistry. Acquiring data from these techniques in a complementary, iterative fashion maximizes structural information.
Table 1: Key Data Acquisition Parameters for Maximum Information Yield
| Technique | Critical Parameter | Optimal Setting (Typical) | Information Gained |
|---|---|---|---|
| HR-ESIMS | Resolution | >60,000 FWHM | Accurate mass for elemental composition |
| Mass Accuracy | < 2 ppm | Confident formula assignment | |
| Scan Mode | Positive/Negative switching | Broad metabolite detection | |
| Fragmentation (MSⁿ) | Data-Dependent Acquisition (DDA) or MRM | Structural connectivity via fragment ions | |
| NMR (¹H) | Field Strength | ≥ 600 MHz | Enhanced dispersion and sensitivity |
| Solvent | Deuterated, matched to LCMS eluent | Enables hyphenated LC-MS-NMR | |
| Experiment | 1D with water suppression | Concentration, purity, integrals | |
| NMR (2D) | Key Experiments | ¹H-¹³C HSQC, HMBC, COSY, NOESY/ROESY | C-H connectivity, long-range couplings, stereochemistry |
Integrated Data Processing Workflow
Raw data must be processed systematically to extract all latent information. Use vendor-neutral software (e.g., MZmine, MNova, ACD/Spectrus) to correlate MS and NMR datasets for a unified compound analysis.
Experimental Protocol 1: Tiered HR-ESIMS Data Acquisition for Crude Extracts
- Instrument Calibration: Calibrate mass spectrometer daily using standard reference mixture (e.g., sodium formate) in relevant ionization mode.
- LC-HR-ESIMS Analysis:
- Column: C18 (100 x 2.1 mm, 1.7 µm).
- Gradient: 5-100% MeCN in H₂O (both with 0.1% formic acid) over 18 min.
- Flow rate: 0.3 mL/min.
- Acquisition: Full scan (m/z 100-1500) at 70,000 resolution, followed by Top-5 DDA MS/MS scans at 17,500 resolution, stepped NCE (20, 40, 60).
- Data Processing:
- Perform peak picking with 5 ppm mass tolerance.
- Apply blank subtraction to remove background.
- Deconvolute [M+H]⁺/[M-H]⁻ adducts to neutral mass.
- Calculate elemental compositions (< 2 ppm, ring double equivalent constraints).
- Annotate known compounds via database search (e.g., GNPS, AntiBase).
Experimental Protocol 2: Complementary NMR Analysis for Pure Compounds or Key Fractions
- Sample Preparation: Transfer purified compound (≥ 0.1 mg) to NMR tube. Dissolve in 600 µL deuterated solvent (e.g., CD₃OD, DMSO-d₆). Use 3 mm tubes for mass-limited samples.
- 1D ¹H NMR: Acquire with sufficient scans for S/N > 100:1. Use presaturation for solvent suppression. Process with exponential multiplication (lb=0.3 Hz), automatic phasing, and baseline correction.
- 2D NMR Suite: Acquire in sequence:
- ¹H-¹³C HSQC: Set optimal ¹JCH (145 Hz). Key for direct C-H bonds.
- ¹H-¹³C HMBC: Optimize for long-range coupling (~8 Hz). Critical for quaternary carbon and connectivity through heteroatoms.
- ¹H-¹H COSY: Identify vicinal proton couplings.
- NOESY/ROESY (if needed): Mixing time 500 ms. Determine relative configuration.
- Data Processing & Integration: Process 2D data with Gaussian apodization. Analyze data in conjunction with HR-ESIMS-derived molecular formula. Use spin-system analysis to build structural fragments.
Visualized Workflows
Diagram 1: Integrated MS/NMR Structure Elucidation Workflow (96 chars)
Diagram 2: Logical NMR Experiment Sequence (85 chars)
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 2: Essential Materials for HR-ESIMS & NP Structure Elucidation
| Item | Function & Specification | Critical Note |
|---|---|---|
| Deuterated NMR Solvents (CD₃OD, DMSO-d₆, CDCl₃) | Provide deuterium lock for stable NMR field; must be >99.8% D. | Match solvent to LC-MS eluent composition for LC-NMR hyphenation. |
| MS Calibration Solution (e.g., NaTFA, Agilent Tune Mix) | Enables sub-ppm mass accuracy for confident formula assignment. | Calibrate in both ionization modes used. |
| C18 UHPLC Columns (1.7-2.6 µm, 2.1 mm id) | High-efficiency chromatographic separation of complex extracts. | Use guard column to extend lifespan. |
| Solid-Phase Extraction (SPE) Cartridges (C18, Diol, Cyano) | Pre-fractionate crude extracts to reduce complexity for NMR. | Tiered elution (H₂O, MeOH, EtOAc) groups compounds by polarity. |
| 3 mm NMR Tubes (Shigemi type) | Minimize required sample volume for mass-limited natural products. | Essential for samples < 0.1 mg. |
| Data Processing Software (e.g., MestReNova, ACD/Spectrus, MZmine) | Integrates and correlates multi-technique data in a single project. | Vendor-neutral formats (JCAMP, mzML) ensure long-term accessibility. |
| Reference Databases (AntiBase, GNPS, SciFinder) | Annotate known compounds rapidly to avoid rediscovery. | Critical for dereplication early in workflow. |
Adherence to these best practices in acquisition and processing creates a virtuous cycle where high-quality, complementary HR-ESIMS and NMR data streams are efficiently transformed into structural knowledge. This systematic, iterative approach is fundamental to advancing natural product research and accelerating the identification of novel drug leads.
Beyond a Single Answer: Validation Strategies and Comparative Analysis of Analytical Techniques
Within natural product research, the unambiguous elucidation of chemical structure is the critical gateway to understanding bioactivity, enabling synthesis, and guiding drug development. Reliance on a single analytical technique is a recognized source of error, leading to misassignment and costly downstream consequences. This application note, framed within a broader thesis on integrated spectroscopic approaches, argues that the synergistic combination of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy forms a non-negotiable, orthogonal dataset. This partnership delivers the "gold standard" of proof by providing complementary molecular descriptors: exact mass and elemental composition from HR-ESIMS, and atomic connectivity, stereochemistry, and functional group information from NMR.
The Complementary Data Landscape: HR-ESIMS vs. NMR
The following table summarizes the core quantitative and qualitative data provided by each orthogonal technique, illustrating their complementary nature.
Table 1: Complementary Data from HR-ESIMS and NMR Spectroscopy
| Parameter | HR-ESIMS | Multidimensional NMR (¹H, ¹³C, 2D) |
|---|---|---|
| Primary Output | Exact mass (m/z) | Chemical shift (δ, ppm) |
| Key Quantitative Data | Measured m/z (e.g., 455.2387), Calculated m/z, Mass error (Δ ppm) | Integration (proton count), Coupling constant (J, Hz) |
| Structural Information | Molecular formula (from exact mass), Fragment ions (substructure) | Atom connectivity (COSY, HMBC), Carbon hybridization (DEPT), Spatial proximity (NOESY/ROESY) |
| Detection Limit | Very high sensitivity (fmol-pmol) | Lower sensitivity (nmol-μmol range) |
| Sample Throughput | High | Moderate to Low |
| Critical Role | Formula assignment, purity assessment, detecting minor impurities | Unambiguous constitution and relative configuration |
Detailed Experimental Protocols
Protocol 1: HR-ESIMS Analysis for Natural Product Extracts
Objective: To obtain the exact molecular mass and propose molecular formulas for purified compounds or major components in a crude extract.
Materials & Sample Prep:
- Sample: Purified compound (10-100 μM in 50-100 μL) or crude fraction dissolved in suitable solvent.
- Solvent: LC-MS grade MeOH or ACN, often with 0.1% formic acid (positive ion mode) or ammonium hydroxide (negative ion mode).
- Calibrant: Use appropriate ESI low/high concentration tuning mix (e.g., from Agilent, Waters, Thermo) for immediate external calibration or as a co-injected internal standard.
Instrument Parameters (Generic):
- Ionization Mode: ESI positive or negative.
- Mass Range: 50-2000 m/z.
- Resolution: >30,000 (FWHM at specified m/z).
- Source Temperature: 120-150°C.
- Desolvation Temperature: 250-350°C.
- Cone/Sheath Gas Flow: Optimized for stable spray.
- Lock Mass/Reference Compound: Leucine enkephalin (556.2771 [M+H]+) or other suitable standard for internal mass correction.
Procedure:
- Direct infusion: Introduce sample via syringe pump at 5-10 μL/min.
- Data Acquisition: Acquire data over 1-2 minutes, averaging multiple scans.
- Data Processing: Use instrument software to centroid raw data. Apply internal/external calibration.
- Formula Calculation: Input measured [M+H]+ or [M-H]- ion exact mass into formula calculator software. Set constraints: elements likely present (C, H, O, N, etc.), unsaturation index, and isotopic pattern matching (using M+1, M+2 intensities).
- Validation: Accept proposed formulas with mass error < 3 ppm and a good fit to the isotopic distribution.
Protocol 2: Comprehensive NMR Analysis for Structure Elucidation
Objective: To determine the planar structure and relative stereochemistry of a purified natural product.
Materials & Sample Prep:
- Sample: 0.5-2 mg of highly purified compound.
- Deuterated Solvent: CDCl₃, CD₃OD, DMSO-d₆, etc., selected based on compound solubility. Use 500-600 μL in a 5 mm NMR tube.
- Reference Standard: Add 0.1% v/v TMS (δ 0.00 ppm for ¹H/¹³C) or use residual solvent peak (e.g., CHCl₃ in CDCl₃ at δ 7.26 ppm for ¹H).
1D NMR Acquisition (¹H, ¹³C, DEPT-135):
- ¹H NMR: 16-64 scans, spectral width 0-12 ppm, relaxation delay (D1) 1-2 sec.
- ¹³C NMR: 1024-4096 scans, spectral width 0-220 ppm, D1 = 2 sec, use broadband decoupling.
- DEPT-135: Differentiates CH₃/CH (positive phase) from CH₂ (negative phase) and quaternary C (no signal).
2D NMR Acquisition (Critical for Connectivity):
- COSY (Correlation Spectroscopy): Identifies scalar-coupled protons (typically ²JHH, ³JHH).
- HSQC (Heteronuclear Single Quantum Coherence): Direct ¹H-¹³C one-bond correlations (¹JCH). Essential for assigning protons to their directly attached carbons.
- HMBC (Heteronuclear Multiple Bond Correlation): Long-range ¹H-¹³C correlations (typically ²JCH, ³JCH). Crucial for connecting structural fragments through quaternary carbons and carbonyls.
- NOESY/ROESY (Nuclear Overhauser Effect Spectroscopy): Provides through-space proximity information (<5 Å) for determining relative stereochemistry and conformation.
Workflow:
- Acquire ¹H NMR to assess purity and proton count (integration).
- Acquire ¹³C and DEPT-135 to determine number and type of carbons.
- Acquire HSQC to create a direct C-H correlation map.
- Acquire COSY to establish proton networks.
- Acquire HMBC to connect proton networks via long-range couplings to carbons.
- Acquire NOESY/ROESY to derive spatial relationships.
- Iterative Assignment: Use all spectra in concert to assign all proton and carbon signals and propose/refine the structure.
Visualizing the Integrated Workflow
The logical flow from sample to confirmed structure, emphasizing the non-negotiable integration of both techniques, is depicted below.
Diagram Title: Integrated HR-ESIMS & NMR Structure Elucidation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Combined HR-ESIMS/NMR Analysis
| Item | Function & Importance |
|---|---|
| Deuterated NMR Solvents (CDCl₃, CD₃OD, DMSO-d₆) | Provides the lock signal for stable NMR magnetic field; allows for spectral referencing without interfering proton signals. |
| Internal Mass Calibrants (e.g., Leu-Enkephalin, Ultramark 1621) | Enables real-time internal mass calibration during HR-ESIMS acquisition, ensuring sustained sub-ppm mass accuracy. |
| LC-MS Grade Solvents & Volatile Buffers (MeOH, ACN, H₂O, 0.1% FA) | Minimizes ion suppression and background noise in HR-ESIMS; ensures compatibility with ESI source and column chemistry. |
| NMR Reference Standards (Tetramethylsilane - TMS) | Provides an absolute chemical shift reference point (δ 0.00 ppm) for both ¹H and ¹³C nuclei. |
| Specialized NMR Tubes (5 mm, 400-600 MHz matched) | High-quality tubes ensure optimal magnetic field homogeneity, which is critical for achieving high-resolution NMR spectra. |
| Structure Elucidation Software (e.g., MestReNova, ACD/Labs, ChenDraw) | Essential platform for processing, analyzing, and correlating multi-technique datasets (NMR, MS) and drawing chemical structures. |
Within the context of a thesis on natural product structure elucidation, the selection of analytical techniques is paramount. High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy are cornerstone technologies. This article provides detailed application notes and protocols, comparing their specific utilities for answering discrete structural questions, framed for the researcher in drug development.
Core Quantitative Comparison: HR-ESIMS vs. NMR
Table 1: Fundamental Parameters for Technique Comparison
| Parameter | HR-ESIMS | NMR (¹H, 500 MHz) |
|---|---|---|
| Sample Requirement | ~1-100 pg (for pure compounds) | ~0.1-5 mg |
| Analysis Time | ~1-5 minutes per run | 5 minutes to several hours |
| Key Output | Exact mass (m/z), elemental composition | Connectivity, stereochemistry, functional groups |
| Mass Accuracy | < 5 ppm (routinely), often < 1 ppm | Not Applicable |
| Sensitivity | High (femtomole to attomole) | Moderate to low (millimolar concentrations) |
| Quantitative Capability | Semi-quantitative (requires standards) | Quantitative (integrals) |
| Throughput | High | Low to moderate |
Table 2: Suitability for Specific Structural Questions
| Structural Question | HR-ESIMS Suitability (Score: 1-5) | NMR Suitability (Score: 1-5) | Primary Technique |
|---|---|---|---|
| Determining Molecular Formula | 5 (Definitive) | 2 (Indirect, via fragments) | HR-ESIMS |
| Identifying Knowns via Database Match | 5 | 3 | HR-ESIMS |
| Establishing Carbon Connectivity | 1 | 5 | NMR |
| Determining Double Bond Location | 3 (via MS/MS fragments) | 5 (via J-coupling, NOE) | NMR |
| Identifying Functional Groups | 2 (via diagnostic ions) | 5 (via chemical shift) | NMR |
| Assigning Stereochemistry | 1 (limited) | 5 | NMR |
| Detecting Minor Impurities | 5 (high sensitivity) | 3 (if not resolved) | HR-ESIMS |
| Observing Labile Protons (e.g., OH) | 1 | 5 (with exchange) | NMR |
| Following Reaction Pathways | 5 (rapid, sensitive) | 4 (if concentration sufficient) | HR-ESIMS |
Detailed Application Notes & Protocols
Application Note: Rapid Dereplication of Crude Extracts
Objective: To quickly identify known natural products in a crude extract, filtering out previously characterized compounds. Primary Technique: HR-ESIMS. Workflow Protocol:
- Sample Preparation: Dissolve 1 mg of crude natural product extract in 1 mL of LC-MS grade MeOH. Filter through a 0.22 µm PTFE syringe filter.
- LC-HR-ESIMS Analysis:
- Column: C18 reversed-phase (2.1 x 100 mm, 1.7 µm).
- Mobile Phase: A: H2O (0.1% Formic Acid), B: MeCN (0.1% Formic Acid).
- Gradient: 5% B to 100% B over 20 min.
- MS Parameters (ESI+): Capillary Voltage: 3.0 kV; Cone Voltage: 40 V; Source Temp: 120°C; Desolvation Temp: 350°C. Acquire full-scan data from m/z 150-2000 with resolution > 30,000.
- Data Processing: Extract exact masses for all major peaks. Search against in-house or public databases (e.g., DNP, AntiBase) within a 5 ppm mass tolerance.
- Confirmation: For putative matches, compare isotopic patterns and acquire MS/MS spectra. Note: Final confirmation of a known compound requires co-injection with an authentic standard or comparative NMR analysis.
Protocol: Determining Relative Stereochemistry via NMR
Objective: To establish the relative configuration of a complex natural product with multiple chiral centers. Primary Technique: NMR (¹H, COSY, HSQC, HMBC, ROESY). Detailed Protocol:
- Sample Preparation: Weigh 2-3 mg of pure compound into a 1.7 mm NMR tube. Dissolve in 30 µL of deuterated solvent (e.g., CD3OD). Ensure sample is homogeneous.
- ¹H NMR Acquisition (500 MHz):
- Use a standard 1D pulse sequence (zg30).
- Set spectral width to 20 ppm, acquisition time to 4 seconds, relaxation delay (D1) to 2 seconds.
- Collect 64 transients. Process with exponential multiplication (lb=0.3 Hz).
- 2D NMR Experiments:
- COSY: Identify scalar (J) coupling networks. Use standard cosygpmfpph pulse sequence.
- HSQC: Assign all protonated carbons. Use hsqcetgp with adiabatic pulses on 13C.
- HMBC: Correlate protons to long-range coupled carbons (²,³JCH). Use hmbcgplpndqf, optimize for 8 Hz long-range coupling constant.
- ROESY (Critical for Stereochemistry): Acquire a phase-sensitive ROESY (roeasyph) with a spin-lock mixing time of 400 ms. Through-space correlations (NOEs) observed here are key for determining relative configuration of protons within ~5 Å.
- Stereochemical Analysis:
- Construct a molecular model (e.g., using computational software).
- Integrate key ROESY cross-peaks. Protons held in close proximity in space (e.g., diaxial protons on a ring) will show strong NOEs, while those trans-diaxial or distant will show weak or no NOE.
- Apply the Karplus equation (using ³JHH coupling constants from 1D/2D data) to determine dihedral angles, further constraining stereochemistry.
Visualization of Workflows
Title: HR-ESIMS & NMR Synergistic Workflow
Title: Structural Question Decision Tree
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for HR-ESIMS & NMR Analysis of Natural Products
| Item | Function/Benefit | Typical Vendor/Example |
|---|---|---|
| LC-MS Grade Solvents | Ultra-purity minimizes background ions and enhances MS sensitivity and reproducibility. | Fisher Chemical, Honeywell |
| Deuterated NMR Solvents | Provides the lock signal for stable NMR acquisition; allows for solvent signal suppression. | Cambridge Isotope Laboratories (e.g., DMSO-d6, CD3OD) |
| Reverse-Phase C18 UHPLC Columns | High-efficiency separation of complex natural product mixtures prior to ESIMS analysis. | Waters ACQUITY, Phenomenex Kinetex |
| PTFE Syringe Filters (0.22 µm) | Removes particulate matter from samples, protecting LC columns and NMR tubes. | Millipore Millex |
| Capillary NMR Tubes (1.7 mm) | Enables high-quality NMR data acquisition with sub-milligram sample quantities. | Bruker SampleJet |
| Internal Mass Calibrants (for ESI) | Provides real-time, accurate mass calibration during HR-ESIMS analysis. | e.g., Sodium formate, Agilent ESI-L Tuning Mix |
| NMR Reference Standards | Provides chemical shift reference (e.g., TMS at 0 ppm) for accurate spectral assignment. | Tetramethylsilane (TMS), DSS |
| Structure Elucidation Software | Aids in processing 2D NMR data, predicting NMR shifts, and managing structural hypotheses. | MestReNova, ACD/Labs, ChenDraw |
Within a thesis focused on HR-ESIMS and NMR as the primary engines for natural product structure elucidation, the assignment of absolute configuration and the resolution of complex stereochemical puzzles often require complementary techniques. This document provides application notes and detailed protocols for Electronic/Vibrational Circular Dichroism (ECD/VCD), X-ray Crystallography, and Chemical Derivatization, guiding researchers on their strategic deployment.
Application Notes & Decision Framework
When to Choose ECD/VCD
ECD and VCD are chiroptical spectroscopic methods used to determine absolute configuration. ECD measures the differential absorption of left- and right-circularly polarized light in the UV-Vis region, sensitive to chromophores. VCD operates in the IR region, probing vibrational transitions, and is applicable to a wider range of molecules without specific chromophores.
Application Scenario:
- ECD: Ideal for natural products with a known or derivatized chromophore (e.g., conjugated dienes, aromatic systems, carbonyls). Use when only ~0.1-1 mg of compound is available. Effective for rigid, semi-rigid systems, or flexible molecules for which stable conformers can be computationally modeled.
- VCD: Suitable for virtually all organic molecules, including those without UV chromophores (e.g., terpenes, alkaloids). Requires ~0.5-5 mg. Particularly powerful for molecules with multiple stereocenters where NMR coupling constants are ambiguous.
Limitations: Both require close comparison with computationally simulated spectra (TD-DFT for ECD; DFT for VCD). Results are model-dependent and can be confounded by solvent effects, conformational flexibility, and impurities.
When to Choose X-ray Crystallography
X-ray crystallography provides unambiguous determination of the complete molecular structure, including absolute configuration if anomalous scattering atoms (e.g., S, Cl, Br, or heavy atom derivatives) are present.
Application Scenario:
- The gold standard when a single crystal of suitable quality (~10-100 µm in all dimensions) can be obtained from ~0.1-1 mg of compound.
- Mandatory for novel, complex polycyclic frameworks with unprecedented connectivity or multiple, densely packed stereocenters.
- Essential for confirming structures of metal-organic complexes or where other techniques yield ambiguous results.
Limitations: The primary barrier is obtaining a diffraction-quality crystal. This can be time-consuming and sometimes impossible for oily or amorphous compounds. The molecule must be stable in the crystalline state.
When to Choose Chemical Derivatization
Chemical derivatization involves covalently modifying the natural product to either (a) introduce a chromophore for ECD, (b) introduce a heavy atom for X-ray analysis, or (c) simplify stereochemical analysis by converting problematic functional groups (e.g., alcohols to esters for NMR analysis via Mosher's method).
Application Scenario:
- For Absolute Configuration: Apply Mosher’s method (¹H NMR analysis of (R)- and (S)-MTPA esters) to determine the absolute configuration of secondary alcohols. Requires ~0.5-1 mg per derivative.
- For Crystallography: Synthesize a heavy-atom containing derivative (e.g., bromobenzoate) to facilitate crystal structure solution.
- For ECD: Create a chromophoric derivative (e.g., para-dimethylaminobenzoate ester) for ECD analysis of aliphatic alcohols.
Limitations: Requires additional synthetic steps, consumes more material, and assumes the derivatization proceeds with known stereochemistry. Not suitable for compounds unstable to reaction conditions.
Quantitative Comparison & Decision Table
Table 1: Comparison of Complementary Techniques for Stereochemical Analysis
| Feature | ECD | VCD | X-ray Crystallography | Chemical Derivatization (e.g., Mosher's) |
|---|---|---|---|---|
| Sample Need | 0.1-1 mg | 0.5-5 mg | 0.1-1 mg (for crystal) | 0.5-2 mg (per derivative) |
| Key Requirement | UV Chromophore | Computational model | Single, quality crystal | Reactive functional group |
| Timeframe | Days to weeks | Weeks | Days to months | Days to weeks |
| Information Gained | Absolute configuration | Absolute configuration | Full 3D structure | Relative/absolute configuration |
| Unambiguity | Model-dependent | Model-dependent | Unambiguous (with anomalous scatterers) | High (if reaction known) |
| Primary Cost | Computation/Instrument | Computation/Instrument | Instrument/Personnel | Reagents/Personnel |
Detailed Experimental Protocols
Protocol for ECD Analysis and Computational Comparison
Objective: Determine absolute configuration via experimental ECD and Time-Dependent Density Functional Theory (TD-DFT) simulation.
Materials:
- HR-ESIMS/NMR-characterized compound (0.1-1 mg).
- Spectroscopic-grade solvent (e.g., acetonitrile, methanol).
- Quartz ECD cuvette (path length 0.1-1 mm).
- ECD spectrometer.
- Computational software (e.g., Gaussian, ORCA).
Procedure:
- Sample Preparation: Accurately weigh compound. Dissolve in appropriate solvent to an absorbance of <2 in the region of interest. Filter (0.2 µm PTFE) if necessary.
- Experimental ECD: Acquire ECD spectrum (typically 190-400 nm). Parameters: bandwidth 1 nm, step size 0.5 nm, scan speed 100 nm/min, 3-5 accumulations. Subtract solvent baseline.
- Computational Modeling: a. Use DFT (e.g., B3LYP/6-31+G(d,p)) to optimize the geometry of proposed stereoisomer(s). b. Perform conformational search to identify low-energy conformers (>1% population). c. At a higher theory level (e.g., CAM-B3LYP/def2-TZVP), calculate excited states and rotational strengths for each conformer. d. Boltzman-weight and sum the spectra of all conformers. Apply a Gaussian broadening function (σ ~0.3 eV).
- Comparison: Compare the sign and shape of key Cotton effects in the experimental and simulated spectra. Match confirms the tested absolute configuration.
Protocol for Heavy-Atom Derivatization for X-ray
Objective: Synthesize a 4-bromobenzoate ester to facilitate crystal structure solution.
Materials:
- Natural product alcohol (1-2 mg).
- 4-Bromobenzoyl chloride, anhydrous pyridine, dichloromethane (DCM).
- Standard micro-scale workup materials (TLC plates, silica, etc.).
Procedure:
- Under inert atmosphere, dissolve the alcohol (~1 mg) in anhydrous DCM (100 µL) in a micro-vial.
- Add anhydrous pyridine (2 µL, excess) followed by 4-bromobenzoyl chloride (1.5 molar equiv) in DCM (10 µL).
- Stir the reaction at room temperature, monitoring by TLC (hexanes/EtOAc).
- Upon completion (typically 2-12 h), quench with a drop of methanol.
- Directly purify the mixture using micro-scale silica gel preparative TLC.
- Elute the derivative band, concentrate, and subject to crystallization trials (e.g., vapor diffusion of pentane into a DCM solution).
Protocol for Mosher Ester Analysis for Absolute Configuration
Objective: Determine the absolute configuration of a secondary alcohol via ¹H NMR analysis of its (R)- and (S)-α-methoxy-α-(trifluoromethyl)phenylacetyl (MTPA) esters.
Materials:
- Chiral secondary alcohol (0.3-0.5 mg per derivative).
- (R)- and (S)-MTPA chloride.
- Anhydrous pyridine, deuterated chloroform (CDCl₃), NMR tube.
Procedure:
- Divide the alcohol sample into two equal portions in separate 1 mL micro-vials.
- To each vial, add anhydrous CDCl₃ (50 µL) and anhydrous pyridine (5 µL).
- To one vial, add (R)-MTPA-Cl (2 µL, ~1.2 equiv). To the other, add (S)-MTPA-Cl (2 µL).
- Cap vials, stir at room temperature overnight.
- Directly transfer each reaction mixture to a 1.7 mm NMR tube. Acquire high-field ¹H NMR spectra (500 MHz or higher).
- Analysis: For each ester, assign the signals for the protons near the stereocenter (e.g., L1, L2, L3 groups). Calculate the chemical shift difference (Δδ = δS – δR) for each proton. A positive Δδ for a proton indicates it lies in the quadrant of the MTPA plane where it is shielded by the phenyl group, allowing assignment of absolute configuration via the established model.
Visualized Workflows & Relationships
Title: Decision Flow for Complementary Stereochemical Techniques
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Complementary Structure Elucidation
| Item | Function & Application |
|---|---|
| (R)- & (S)-MTPA Chloride | Chiral derivatizing agents for determining absolute configuration of alcohols, amines via ¹H NMR chemical shift differences. |
| 4-Bromobenzoyl Chloride | Heavy-atom derivatizing agent. Introduces bromine to facilitate X-ray crystallographic phase determination for alcohol-containing NPs. |
| para-Dimethylaminobenzoyl Chloride | Chromophoric derivatizing agent. Introduces a strong UV chromophore into aliphatic alcohols for subsequent ECD analysis. |
| Anhydrous Pyridine | Base and acylation catalyst for micro-scale derivatization reactions in inert atmosphere. |
| Deuterated Chloroform (CDCl₃) | Standard NMR solvent for derivatization reactions and direct acquisition of ¹H NMR spectra of Mosher esters. |
| Spectroscopic-Grade Solvents (MeCN, MeOH) | Essential for acquiring high-quality, artifact-free UV, ECD, and VCD spectra. |
| Micro-scale Reaction Vials (1-2 mL) | For conducting derivatization reactions on sub-milligram quantities of precious natural products. |
| 0.2 µm PTFE Syringe Filters | For filtering NMR/ECD samples to remove particulate matter that causes light scattering or degraded spectra. |
| Short Pathlength Quartz Cuvettes (0.1-1 mm) | For ECD measurements of small sample quantities, allowing higher effective concentration without saturation. |
1. Introduction Within a thesis on the integrated application of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) for natural product structure elucidation, database benchmarking emerges as a critical, time-saving prelude to full structural analysis. Dereplication—the early identification of known compounds—prevents redundant isolation and characterization, allowing researchers to focus resources on novel entities. This protocol details the systematic use of spectral libraries for HRMS and NMR data to achieve rapid confirmation and dereplication.
2. Core Spectral Databases: A Quantitative Overview The efficacy of dereplication is directly tied to the breadth and quality of the reference databases employed. Below is a comparison of key proprietary and public resources.
Table 1: Key HRMS and NMR Databases for Natural Product Research
| Database Name | Type | Typical Size/Coverage (Approx.) | Primary Use in Dereplication |
|---|---|---|---|
| AntiBase / AntiBase 2023 | HRMS (EI, ESI), NMR (1D, 2D) | ~55,000 microbial & natural products | Comprehensive LC-HRMS/MS & NMR library for microbial metabolites. |
| DNP (Dictionary of Natural Products) | Structural & Physicochemical | > 275,000 compounds | Authoritative source for searching by mass, formula, substructure. |
| GNPS (Global Natural Products Social Molecular Networking) | Public HRMS/MS | Millions of community MS/MS spectra | Molecular networking & spectral matching against public data. |
| HMDB (Human Metabolome Database) | Public HRMS/MS, NMR | > 200,000 metabolites | Useful for dereplication of common bioactive metabolites. |
| Spektraris (Bruker) | NMR | Customizable library for 1D/2D NMR | Direct spectral comparison and scoring within NMR software. |
| mzCloud | HRMS/MS | High-quality curated MS/MS trees | Advanced spectral interpretation and fragmenter matching. |
3. Integrated Dereplication Workflow Protocol
Protocol 3.1: Integrated HRMS and NMR Dereplication Objective: To rapidly identify a purified natural product compound (1-5 mg) and determine if it is novel or known. Materials: Purified compound, LC-HRMS system (Q-TOF, Orbitrap), NMR spectrometer (≥ 400 MHz), database access (e.g., AntiBase, DNP, GNPS).
Procedure: A. HR-ESIMS Analysis & Database Query
- Sample Preparation: Dissolve compound in appropriate LC-MS grade solvent (e.g., MeOH, ACN) at ~1 µg/µL.
- HR-ESIMS Acquisition: Acquire high-resolution mass spectra in both positive and negative ionization modes. Key parameters:
- Mass Range: 100-2000 m/z
- Resolution: > 30,000 FWHM
- Source Voltage: 3.5-4.5 kV (ESI+)
- Accurate mass calibration using standard calibrant (e.g., sodium formate).
- Data Processing:
- Extract accurate mass for [M+H]⁺, [M+Na]⁺, [M-H]⁻ ions.
- Calculate possible molecular formulas using instrument software (e.g., Elemental Composition) with constraints: C₂-₁₀₀ H₁-₂₀₀ O₀-₅₀ N₀-₂₀, etc., and a mass error tolerance of < 5 ppm.
- Database Benchmarking:
- Input the calculated molecular formula into structure databases (e.g., DNP, SciFinder). Record all possible structural hits.
- If HR-MS/MS data is available, perform spectral similarity search in MS/MS libraries (e.g., GNPS, mzCloud). A cosine score > 0.7 suggests a high-probability match.
B. NMR Analysis & Spectral Matching
- Sample Preparation: Dissolve 1-2 mg of compound in 0.6 mL of deuterated solvent (e.g., CD₃OD, DMSO-d₆). Transfer to a 5 mm NMR tube.
- 1D NMR Acquisition: Acquire ¹H NMR and standard decoupled ¹³C NMR spectra.
- ¹H NMR: 16-64 scans, spectral width 12-15 ppm.
- ¹³C NMR: 1024-4096 scans, spectral width 220-240 ppm.
- Preliminary Analysis: Calculate the unsaturation index from the molecular formula. Analyze ¹H NMR for coupling constants, integration, and characteristic signals (e.g., olefinic, aromatic, methyl singles).
- Database Benchmarking:
- Process NMR data (phasing, baseline correction, referencing).
- Import spectra into database software (e.g., Spektraris, ACD/Labs). Perform automated peak picking and chemical shift search against selected libraries.
- Manually compare experimental ¹H and ¹³C chemical shifts with those of the top candidates from the HRMS query. A mean deviation of < 0.1 ppm for ¹H and < 1-3 ppm for ¹³C is strongly indicative of a match.
- Confirmation: For the top database match, compare acquired 2D NMR data (e.g., HSQC, HMBC, COSY) with published data or predicted correlations for final confirmation.
4. The Scientist's Toolkit: Essential Reagents & Materials
Table 2: Key Research Reagent Solutions for HRMS & NMR Dereplication
| Item | Function & Specification |
|---|---|
| LC-MS Grade Solvents (MeOH, ACN, H₂O) | Minimize background ions and signal suppression during HR-ESIMS analysis. |
| Deuterated NMR Solvents (CD₃OD, DMSO-d₆, CDCl₃) | Provide a lock signal for spectrometer stability and minimize solvent interference in ¹H NMR. |
| Mass Calibration Standard (e.g., Agilent ESI-L Tuning Mix, Sodium Formate) | Enables sub-ppm mass accuracy calibration of the HRMS instrument pre-run. |
| NMR Reference Compound (e.g., TMS, DSS) | Provides a 0 ppm reference point for chemical shift calibration in NMR spectra. |
| Solid Phase Extraction (SPE) Cartridges (C18, Diol) | For rapid cleanup and desalting of crude extracts prior to LC-HRMS analysis. |
5. Visualization of the Integrated Dereplication Workflow
Title: Integrated HRMS and NMR Dereplication Decision Pathway
Title: Data Flow from Instrument to Database Report
Within the framework of natural product drug discovery, the definitive elucidation of molecular structure is paramount. This article, situated within a broader thesis on the integrated application of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) spectroscopy, establishes formal criteria for classifying structural assignments. Distinguishing between a proposed and a definitive structure is critical for prioritizing compounds for lead optimization, synthesis, and preclinical studies, thereby streamlining the drug development pipeline.
Core Criteria for Structural Assignment Confidence
The following table summarizes the key data types and their contribution to confidence levels.
Table 1: Criteria for Structural Assignment Confidence
| Data Category | Proposed Structure | Definitive Structure | Quantitative Benchmark (Typical) |
|---|---|---|---|
| HR-ESIMS | Molecular formula suggestion. | Exact mass confirmation of molecular ion and key fragments. | Mass accuracy < 5 ppm; isotopic pattern match (RMSD < 10%). |
| 1D NMR (¹H, ¹³C) | Partial skeletal information, number of protons/carbons. | Complete, unambiguous assignment of all chemical shifts. | ¹³C NMR signal-to-noise (S/N) > 20:1 for minor peaks. |
| 2D NMR Correlations | Limited set (e.g., COSY, HSQC only). | Exhaustive set (COSY, HSQC, HMBC, NOESY/ROESY). | HMBC long-range correlations justify all connectivities. |
| Stereochemistry | Undefined or partially defined (relative only). | Fully defined (absolute configuration). | DP4+ probability > 95%; CD/TDDFT fit; or X-ray crystallography. |
| Literature & Database | Partial match to known class. | No contradictory data; full differentiation from all known analogues. | Comprehensive search (e.g., SciFinder, AntiBase). |
| Corroboration | Single technique emphasis. | Concordance of all spectroscopic, computational, and/or synthetic data. | Synthetic confirmation of NMR and [α]D of the natural product. |
Application Notes and Experimental Protocols
Protocol 1: Integrated HR-ESIMS and NMR Workflow for Definitive Elucidation
Objective: To unambiguously determine the planar structure and stereochemistry of a novel natural product (NP-1). Materials: Purified NP-1 (>95% purity by analytical LCMS), deuterated NMR solvents (CD₃OD, DMSO-d₆), ESI-LTO-Orbitrap or Q-TOF mass spectrometer, 600 MHz NMR spectrometer with cryoprobe. Procedure:
- HR-ESIMS Analysis:
- Prepare a 10 µg/mL solution of NP-1 in LC-MS grade MeOH with 0.1% formic acid.
- Acquire data in positive and negative ion modes. Calibrate instrument daily using standard mixture.
- Collect full-scan MS (m/z 100-2000) and data-dependent MS/MS on the [M+H]⁺ and/or [M-H]⁻ ions.
- Data Analysis: Process using Xcalibur or similar. Confirm molecular formula from the accurate mass of the adduct ion (e.g., [M+Na]⁺) with mass error < 2 ppm. Analyze MS² fragments for characteristic substructures.
- NMR Experiments for Planar Structure:
- Dissolve 2-5 mg of NP-1 in 0.6 mL of appropriate deuterated solvent.
- Acquire ¹H NMR (8-64 scans), ¹³C NMR (APCT, >2000 scans for S/N), and the following 2D experiments:
- HSQC: 256 t1 increments, 1K data points in F2.
- COSY: 256 t1 increments.
- HMBC: Optimize for J = 8 Hz, 512 t1 increments.
- Data Analysis: Process with MestReNova or ACD/Labs. Assign all ¹H and ¹³C signals. Use HMBC long-range correlations (²,³JC,H) to establish all atom connectivities, closing all structural loops.
- Stereochemical Determination:
- Acquire NOESY (mixing time 600 ms) or ROESY experiments.
- Measure coupling constants from 1D ¹H NMR.
- Perform DP4+ probability analysis: Calculate NMR chemical shifts for all possible stereoisomers using Gaussian 16 at the mPW1PW91/6-31G(d) level. Input experimental shifts to determine the most probable isomer.
- Optional for absolute configuration: Acquire experimental Electronic Circular Dichroism (ECD) spectrum and compare to TDDFT-calculated spectra for candidate enantiomers.
Workflow for Definitive Structure Elucidation
Protocol 2: DP4+ Probability Calculation for Stereochemical Assignment
Objective: To compute the statistical probability for each candidate stereoisomer. Software: Gaussian 16, Python with RDKit and NumPy, DP4+ script. Procedure:
- Model Generation: Generate all possible stereoisomers of the proposed planar structure using RDKit.
- Conformational Search: For each isomer, perform a molecular mechanics (MMFF94) conformational search. Select all low-energy conformers within 10 kcal/mol of the global minimum.
- NMR Calculation: Optimize selected conformers using Gaussian 16 at the mPW1PW91/6-31G(d) level. Calculate GIAO NMR shielding tensors (for ¹H and ¹³C) at the same level of theory.
- DP4+ Analysis:
- Boltzmann-weight the calculated chemical shifts for each isomer based on conformer energies.
- Input the weighted average calculated shifts and the experimental shifts into the DP4+ Python script.
- The script outputs a probability percentage for each isomer. A probability >95% for one isomer is considered strong evidence for a definitive stereochemical assignment.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Definitive Structure Elucidation
| Item | Function in Context |
|---|---|
| Deuterated NMR Solvents (e.g., CD₃OD, DMSO-d₆) | Provides the lock signal for NMR spectrometer; allows for accurate, solvent-referenced chemical shift measurements. |
| NMR Chemical Shift Reference Standards (e.g., TMS, DSS) | Internal standard for calibrating chemical shift (δ) scale to 0 ppm. |
| LC-MS Grade Solvents & Volatile Buffers (e.g., Ammonium Formate) | Essential for clean HR-ESIMS analysis, minimizing adduct formation and background noise. |
| Cryogenically Cooled NMR Probe (Cryoprobe) | Increases sensitivity by 4x or more, enabling NMR data collection on sub-milligram samples, critical for scarce natural products. |
| DP4+ Computational Software Suite | Statistical tool for quantifying confidence in stereochemical assignments by comparing calculated vs. experimental NMR shifts. |
| ECD Spectrophotometer & Chiral HPLC Column | For experimental determination of absolute configuration via circular dichroism and enantiomeric purity assessment. |
Introduction Within the broader thesis on the integration of High-Resolution Electrospray Ionization Mass Spectrometry (HR-ESIMS) and Nuclear Magnetic Resonance (NMR) for natural product structure elucidation, a critical exercise involves comparing methodological approaches. This application note details the parallel elucidation of a common sesquiterpene lactone, parthenolide, emphasizing either spectroscopic (HR-ESIMS-driven) or spectrometric (NMR-driven) primary data streams, culminating in the same structural solution.
Experimental Protocols
Protocol 1: HR-ESIMS-First, NMR-Validation Workflow
- Sample Preparation: Dissolve 1 mg of purified parthenolide in 1 mL LC-MS grade methanol. Dilute to ~10 ng/µL for direct infusion.
- HR-ESIMS Analysis:
- Instrument: Q-TOF or Orbitrap mass spectrometer with ESI source.
- Parameters: Negative ion mode. Capillary voltage: 2.8 kV. Source temp: 150°C. Desolvation temp: 350°C. Cone voltage: 40 V. Scan range: m/z 50-1000.
- Calibration: Use sodium formate or external calibrant for mass accuracy < 5 ppm.
- Data Acquisition: Acquire spectrum for 1 minute. Use lock mass (e.g., leucine enkephalin) for internal calibration if available.
- Data Processing: Extract exact mass of [M+H]+ and [M+Na]+. Perform formula prediction (C15H20O3). Trigger targeted MS/MS on precursor m/z 249.1492 ([M+H]+) with collision energy ramped from 20-40 eV.
- NMR Validation:
- Prepare ~5 mg sample in 0.6 mL deuterated chloroform (CDCl3).
- Acquire 1H, 13C, HSQC, and HMBC spectra (see Protocol 2 for details).
- Use key HMBC correlations (e.g., H-13 to C-11, C-7, C-8) to confirm lactone and exomethylene placement suggested by MS/MS fragments.
Protocol 2: NMR-First, HR-ESIMS-Corroboration Workflow
- NMR Structure Assembly:
- Sample Preparation: Dissolve 10-15 mg of purified parthenolide in 0.6 mL CDCl3.
- Data Acquisition:
- 1H NMR (500 MHz): 128 scans.
- 13C NMR (125 MHz): 1024 scans, using inverse-gated decoupling for quantitative assessment.
- 2D NMR: HSQC (J-coupling 145 Hz), HMBC (long-range J-coupling 8 Hz), COSY.
- Structure Assembly: Assemble substructures from COSY/HSQC. Connect via HMBC long-range correlations. Key correlations: H3-14 to C-1, C-9, C-10; H3-15 to C-3, C-4, C-5; H-6 to C-8, C-12 (lactone carbonyl).
- HR-ESIMS Corroboration:
- Analyze a dilute aliquot from the NMR sample.
- Confirm molecular formula via exact mass of [M+H]+ (calc. for C15H21O3+: 249.1491). Confirm presence of key functional groups via diagnostic MS/MS fragments (e.g., loss of H2O, loss of C5H8 from the lactone moiety).
Data Presentation
Table 1: Comparative Key Data for Parthenolide Elucidation
| Structural Feature | HR-ESIMS-First Evidence (Primary) | NMR-First Evidence (Primary) |
|---|---|---|
| Molecular Formula | [M+H]+ at m/z 249.1492 (Δ 0.4 ppm). Formula prediction: C15H20O3. | 13C NMR count: 15 distinct carbons. HSQC: 5 methylene, 5 methine, 5 quaternary (incl. carbonyls). Supports C15H20O3. |
| α,β-Unsaturated Lactone | MS/MS fragment at m/z 231.1386 ([M+H-H2O]+) and m/z 203.1437 ([M+H-H2O-CO]+). | 13C signal at δ 170.1 (C-12). HMBC: H-13 to C-11 (δ 141.2), C-12. 1H NMR: H-13 as two broad singlets (~δ 6.2, 5.5 ppm). |
| Exocyclic Methylene | MS/MS fragment at m/z 191.1072 (neutral loss of C3H4O from lactone ring). | 1H NMR: H3-15 as a singlet (~δ 1.4 ppm). HMBC: H3-15 to C-3 (δ 79.5), C-4, C-5. |
| Epoxide Group | Not directly probed. Inferred from molecular formula and unsaturation count after other features. | Key 1H-1H COSY correlations between H-2, H-3. 13C chemical shifts of C-2/C-3 (~δ 55-60 ppm). |
Visualization
HR-ESIMS-Driven Workflow: From Mass to Structure
NMR-Driven Workflow: From Spin Systems to Formula
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item & Typical Solution/Supplier | Function in Elucidation |
|---|---|
| Deuterated NMR Solvents (e.g., CDCl3, DMSO-d6, Methanol-d4) | Provides NMR-inert solvent environment, allows for field frequency locking and internal chemical shift referencing. |
| LC-MS Grade Solvents (e.g., Methanol, Acetonitrile, Water) | Ultra-pure solvents for HR-ESIMS to minimize background ions and adduct formation, ensuring accurate mass measurement. |
| Mass Calibration Solutions (e.g., Sodium Formate, Agilent Tune Mix) | Provides known ion clusters for internal or external calibration of the mass spectrometer to achieve <5 ppm mass accuracy. |
| NMR Reference Standards (e.g., Tetramethylsilane (TMS), Residual solvent peak) | Provides a precise internal reference point (0 ppm) for chemical shift assignment in NMR spectroscopy. |
| Fraction Collection Tubes (e.g., Certified LC/MS vials) | Inert, low-binding containers for storing purified natural product fractions prior to combined HR-ESIMS/NMR analysis. |
Conclusion
The synergistic integration of HR-ESIMS and NMR spectroscopy represents the cornerstone of modern natural product structure elucidation, transforming complex analytical challenges into solvable puzzles. As detailed through foundational principles, practical workflows, troubleshooting guidance, and validation frameworks, this powerful combination provides an unmatched level of certainty in determining molecular formulas, connectivity, and stereochemistry. For biomedical and clinical research, the accurate structures revealed by these techniques are the critical starting point for understanding bioactivity, synthesizing analogs, and developing new therapeutics. Future directions point towards increased automation, more powerful computational integration for data analysis and prediction, and the application of these methods to ever-smaller sample sizes and more complex mixtures, such as those found in microbiomes. The continued evolution of both HR-ESIMS and NMR technology promises to further accelerate the discovery pipeline, unlocking the full potential of nature's chemical diversity for drug development and beyond.